Diversity in Classical Hematology Research
More infoIntrathecal chemotherapy is a mainstay component of acute lymphoblastic leukemia treatment. In Mexico, there is a considerable practice variability in aspects, such as the manner of preparation and the administration technique.
ObjectiveOur objective was to describe the different techniques used for the application of ITC and review the existing recommendations in the literature.
MethodA cross-sectional, nationwide survey study was conducted by an electronic questionnaire sent to hematologists and oncologists in Mexico. We collected demographic data, personal experience, intrathecal chemotherapy techniques, drug preparation and postprocedural conduct.
ResultsWe received 173 responses. Twenty percent had an anesthesiologist administering sedation and pain management. The platelet count considered safe was 50 × 109/L in 48% of the participants. In 77% (n = 133) of the cases, the conventional needle with stylet used was, 49% did not receive any added diluent in the intrathecal chemotherapy and only 42% were recommended to rest in a horizontal position for more than 30 min.
ConclusionWe identified a considerable variation in the administration of intrathecal chemotherapy across the hematologists in Mexico. We discuss the implications and opportunities in reducing the variation in our setting, highlighting the unmet need to establish guidelines that should be evaluated by the Mexican professional society to produce a position paper regarding practice standardization.
The central nervous system (CNS) is considered a sanctuary site for neoplastic cells in patients with acute lymphoblastic leukemia (ALL).1 However, most chemotherapeutic agents do not reach adequate levels in the brain, spinal cord, meninges and cerebrospinal fluid (CSF) and, thus, the administration of intrathecal therapy (ITC) through a lumbar puncture (LP) is needed. The LP is a procedure to access the subarachnoid space at the lumbar cistern level using a needle. It was described for the first time in 1891 by Quincke.2,3
Currently, high-dose methotrexate (HD-MTX) and ITC administration are mainstay components of ALL treatment regimens. It has progressively displaced prophylactic cranial irradiation used to prevent CNS relapse, given its similar efficacy, with fewer late adverse effects.2,4,5-8
Several factors associated with ITC administration contribute to optimal drug distribution throughout the CNS; for example, patient body position, type of spinal needles and stylet, dosing and dilution of drugs, which is essential for effective CNS therapy.9 The use of a correct technique for diagnostic and therapeutic LPs with ITC is one of the keystones of acute leukemia treatment and adequate technique is imperative, not only for ensuring treatment efficacy, but also for preventing traumatic lumbar puncture at diagnosis, leading to increased CNS relapse risk.10
Despite ITC administration being a frequent procedure, there is much practice variability in preparation (volume and type of dissolvent) and administration technique11. The extent of practice variation in Mexico has not been documented.
In this nationwide study, we aimed to describe the different techniques used to apply ITC and review the existing evidence and recommendations in the oncology literature.
MethodsWe carried out a cross-sectional, nationwide survey study. An electronic questionnaire via Google Forms was sent and accessed by cell phone, electronic tablet or computer. Our local ethics committee approved this study. The survey was emailed to 400 hematologists and oncologists from all over the country.
The survey collected demographic data (age, hospital and years of practice) and personal experience (number of ITCs performed per month and academic grade). The rest of the questions were designed as multiple choice and referred to the technique's fundamental aspects (analgesia, patient position, type of needle, bezel orientation and reintroduction of the stylet before needle extraction) and drug preparation, as well as the postprocedural conduct.
The survey was sent directly to physicians from January to April 2018. It was sent through the website platform of two pediatric hematology and/or oncology professional societies; first, through the Agrupación Mexicana para el Estudio de la Hematología (AMEH) and then, through the Agrupación Mexicana de Onco-hematología Pediátrica (AMOHP). Subsequently, a descriptive analysis of the answers and a literature review were performed.
ResultsDemographic dataWe obtained a total of 173 responses (43% response rate). Of these, 116 were collected through the AMEH distribution list and 57, through AMOHP.
We received responses from 26 of the 32 states of the Mexican Republic, 53% of which were from the center of the country, 40%, from northern states and only 7%, from the southern region. This distribution highlights the centralization of the care of patients with cancer in Mexico. Only 1.2% of those surveyed had undergone procedures exclusively in private institutions, 16.3%, in both private and public health institutions and 82.5%, only in public institutions, of which 28 (16%) were hematology/oncology fellows.
Sedation and analgesiaConcerning pain management and anxiety during the procedure, 30% (57) of the surveyed allowed parents to stay in the room when the procedure was performed in pediatric patients. Only 20% (n = 36) answered that they had had an anesthesiologist administering sedation and pain management. The options for anesthesia and sedation varied widely: local lidocaine in 37% (n = 64) of the cases, benzodiazepines in 7% (n = 12) and a combination of analgesic and sedative drugs in 6% (n = 10).
Platelet countThe platelet count that physicians considered safe to apply an ITC in the participants was > 50,000/μL in 48% (n = 84), > 20 × 109/L in another 24% (n = 42), and > 10 × 109/L in 6% (n = 11).
Needle and dilutionIn 77% (n = 133) of the cases, the needle used by practitioners was the conventional one with a stylet (Quincke Type) and, in 8.6% (n = 15), a needle without stylet was used during the procedure.
Forty percent of the doctors (n = 70) introduced the needle with the bezel in a parallel orientation to the spinal ligament longitudinal fibers; 57% (n = 99) reintroduced the stylet before removing the needle.
Ninety percent of the doctors (n = 155) used triple intrathecal therapy (methotrexate, cytarabine and hydrocortisone), 86% (n = 148) applied the three drugs in the same syringe and the remaining 14% (n = 24), in different syringes.
Regarding the diluents used during the procedure, 49% (n = 85) did not add any diluent, while the rest added 5 or 10 mL of sterile water or sodium chloride 0.9% solution to the chemotherapy mixture.
Resting timeAfter the procedure, only 42% of the surveyed doctors (n = 72) recommended resting in a horizontal position for more than 30 min.
DiscussionPractice standardization is the main strategy to improve care quality and reduce unnecessary or harmful variation.12 The extent of practice variation in intrathecal chemotherapy administration in children with lymphoblastic leukemia in Mexico had not been documented. This online survey is the first effort in our country to standardize ITC administration. It is important to note that more than half of the invited hematologists did not answer the questionnaire, which could bias our results. However, the most important centers of our country were well represented. The distribution of the physicians who responded to the survey highlights the centralization of the care of patients with cancer in Mexico.
We identified important variations and discussed the implications and opportunities to reduce them in our setting.
The effectiveness and prevention of undesirable adverse events of ITC depend primarily on a good application technique.13 Furthermore, knowing the behavioral practices in our setting is fundamental.
Regarding sedation, anesthesiologist participation would be desirable,8 but it represented only 20% (n = 34) of those surveyed in our series.
Maintaining a 50 × 109/L platelet count threshold may represent a real challenge during the induction phase for ALL patients. Only 48% of the surveyed doctors considered a threshold of 50 × 109/L to administer ITC, as established per guidelines.14 Howard et al., analyzed 5,223 LPs with no serious complications presented regardless of the platelet count; they suggest that prophylactic platelet transfusion is unnecessary for children with platelets >10 × 109/L. However, this platelet count is not standard practice.15 Only 6% (n = 10) of the surveyed doctors answered that they agree with this recommendation.
Atraumatic needles, such as Whitacre or Sprotte, characterized by a blunt-tipped needle with an opening on its side, are recommended.13 We only had 15% (n = 26) of the physicians following this recommendation. Guidelines suggest that inserting the needle in parallel to ligament fibers could reduce the likelihood of CSF leaks and decrease the risk of post lumbar puncture headaches and possible cerebral bleeding or thrombosis.13,16 Of the surveyed doctors, 70% (n = 121) direct the bezel to the patient's head without considering the dural fibers.
We recommend that intrathecal chemotherapy preparation be done in a mixing center by trained personnel, using the dose and volume specifications provided. As by Joint Commission recommendations, preprocedural time-out protocols help corroborate the correct dose and route of drug administration before proceeding to its application. In pediatric patients, the dose and dilution should be calculated based on age.17-19
The total volume of ITC administration is another point of debate. No trials have compared the impact of different volumes administered on the CNS distribution.17 Olmos-Jiménez et al. suggest using a predetermined volume, depending on the patient's age: for infants under a year: 6 ml; in 1 to 2-year toddlers: 8 ml; for 2 to 3-year old toddlers: 10 ml, and; for children over 3 years: 12 ml of normal saline solution as a diluent. Nevertheless, these recommendations lack scientific evidence to support them.13 In our survey, almost half of the physicians did not add any diluent to the final product. In recent years, and for different reasons, a greater number of alternatives to the drugs used in the application of intrathecal chemotherapy have appeared and each center must define its best options, according to a scrupulous registry of secondary reactions that occur with each of the different brands.20
The best time of administration is not completely clear. However, Yeh et al. demonstrated no clinical difference between early (1st day) or late (8th day) administration of the first triple ITC. They found an event-free survival of 75% vs. 72% (p = 0.26) and a CNS relapse rate of 4% vs. 4.1% (p = 0.96), when comparing delayed vs. early lumbar puncture.21-23 We consider that the late administration strategy would be better at academic centers, where physicians in training perform the procedure.
Optimal intraventricular distribution of ITC is limited by the slow and unidirectional CSF flow. Animal studies show that the position influences drug distribution. Przuntek and Breithaupt studied the resting time in the horizontal position after the ITC application and determined an ideal time of 60 min lying in a horizontal position. Only 42% (n = 73) of the surveyed followed this recommendation.24,25. Patients, family and nurses should be educated on the importance of patients remaining in bed at least 60 min after the procedure and prompt reporting of complications (such as seizures, acute paresthesias or other significant neurological complications).
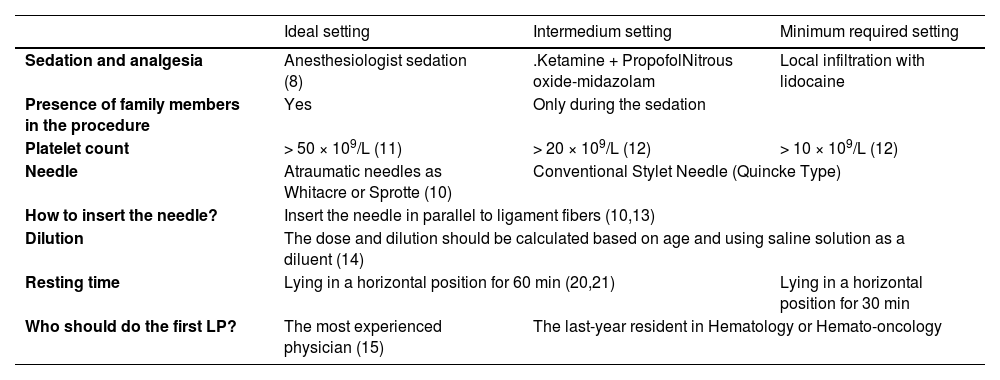
As in many Latin American countries, the Mexican health system has huge disparities in the access to financial resources and, for these reasons, the treatment of leukemia patients is heterogeneous; we suggest that each health professional evaluate these differences. In Table 1, we show data that could support the standardization of the ITC application, according to available scientific information.
Basic recommendations of intrathecal chemotherapy administration.
Ideal setting: Large cancer center with the necessary economic resources. Intermedium setting: School hospital that does not have all the economic resources or space to attend to their patients. Minimum required setting: Small clinic that does not have any more resources (8, 10 – 21).
According to our literature review, a good ITC technique, according to available resources at our center, should include anesthesiologist sedation, a platelet count > 10 × 109/L, the use of atraumatic needles inserted in parallel to ligament fibers and the use of saline solution as a diluent. The first LP should be performed under sedation, on the 8th day by the most experienced physician and a resting time of 60 min in a horizontal position should be followed.
In conclusion, we found a wide variability in technique and practical aspects of ITC administration in Mexico. Accordingly, more research studies are necessary in this area. It is essential to be updated on the safest techniques available to administer ITC to our patients. In this study, we highlight the unmet need to establish guidelines for ITC administration and consider the role of professional associations in building consensus on, and thus establishing, those practice standards.