The minimal residual disease (MRD) status plays a crucial role in the treatment of acute lymphoblastic leukemia (ALL) and is currently used in most therapeutic protocols to guide the appropriate therapeutic decision. Therefore, it is imperative that laboratories offer accurate and reliable results through well standardized technical processes by establishing rigorous operating procedures.
MethodOur goal is to propose a monoclonal antibody (MoAb) panel for MRD detection in ALL and provide recommendations intended for flow cytometry laboratories that work on 4-color flow cytometry platforms.
Results and conclusionThe document includes pre-analytical and analytical procedures, quality control assurance, technical procedures, as well as the information that needs to be included in the reports for clinicians.
The prognostic importance of minimal residual disease (MRD) status during the treatment of acute lymphoblastic leukemia (ALL) has been demonstrated over the last 20 years.1–10 The MRD results are crucial to risk stratification and they guide clinical decision-making for the best therapeutic option and therefore, improve patient outcomes.
Considering this reasoning, it is essential that laboratory approaches for the detection of MRD be robust, fast and as accurate as possible, with sufficient sensitivity and specificity to guarantee reliable results for clinical use. The multiparametric flow cytometry (MFC) meets these requirements. Strategies for the detection of MRD by MFC have been progressively improving in recent years, due to a more in-depth knowledge on the matter and an improvement in the accuracy of flow cytometers, as well as the availability of specific reagents for this purpose.
Because there are many flow cytometry laboratories in our country, many of them working with 4-color flow cytometry, this document is addressed to those laboratories. We assembled the panel of monoclonal antibodies (MoAbs) and the recommendations we propose here are in alignment with the literature data review and practice patterns among the Working Group members. The MoAbs used here were validated by testing marrow samples in normal subjects and ALL cases. For laboratories that work with 8-color flow cytometers, the group's recommendation is to use Euroflow strategies for MRD assessment.11
This current document is an update to this group’s previous report,12 which introduces new antibodies and combinations for MRD detection and aims to recommend standardized cytometric procedures for MRD detection in ALL to be used in laboratories throughout Brazil to obtain more accurate results for clinical use.
RecommendationsBelow are listed the recommended levels (essential, recommended and optional) for each pre-analytical, analytical and post-analytical method.
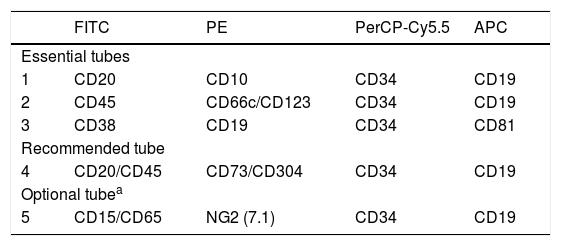
Panel of monoclonal antibodies proposed for MRD detection in ALL (essential)The MoAb panels designed herein for the detection of the B-cell precursor (BCP)-ALL and T-ALL MRD include markers that address the normal maturation of these cell lineages, added to other anomalous ones, to identify the leukemia-associated immunophenotypes (LAIP), such as asynchronous antigen expressions or cross-lineage markers. The proposed MoAb panels and their respective selected fluorochromes are presented in Tables 1 and 2, with the respective levels of recommendations.
Fluorochrome conjugated antibody panels for MRD detection in BCP-ALL by using 4-color multiparametric flow cytometry.
| FITC | PE | PerCP-Cy5.5 | APC | |
|---|---|---|---|---|
| Essential tubes | ||||
| 1 | CD20 | CD10 | CD34 | CD19 |
| 2 | CD45 | CD66c/CD123 | CD34 | CD19 |
| 3 | CD38 | CD19 | CD34 | CD81 |
| Recommended tube | ||||
| 4 | CD20/CD45 | CD73/CD304 | CD34 | CD19 |
| Optional tubea | ||||
| 5 | CD15/CD65 | NG2 (7.1) | CD34 | CD19 |
Abbreviations: FITC: fluorescein isothiocyanate; PE: phycoerythrin; PerCP-Cy5.5: peridinin chlorophyll protein/cyanin5; APC: allophycocyanin.
Fluorochrome conjugated antibody panels for MRD detection in T-ALL by using 4-color multiparametric flow cytometry.
| FITC | PE | PerCP-Cy5.5 | APC | |
|---|---|---|---|---|
| Essential tubes | ||||
| 1 | CD45 | SmCD3 | cyCD3 | CD7 |
| 2 | NuTdT | cyCD3 | CD5 | CD7 |
| 3 | CD7 | CD99 | cyCD3 | CD1 |
| Recommended tubesa | ||||
| 4 | CD7 | CD10+CD117 | cyCD3 | CD45RA |
| 5 | CD7 | cyCD3 | CD34 | CD13+CD33 |
| 6 | CD44 | CD7 | cyCD3 | CD56 |
Abbreviations: FITC: fluorescein isothiocyanate; PE: phycoerythrin; PerCP-Cy5.5: peridinin chlorophyll protein/cyanin5; APC: allophycocyanin.
To evaluate the maturation of the B-cell lineage, CD10, CD19, CD20, CD34, CD38, CD45 and CD81 were included in the panel, in which CD19 and CD34 were used as backbone markers. It is important to take into account that some of these markers undergo expression modulation, such as CD19 and CD20 (upregulation), CD10 and CD34 (downregulation), during induction therapy, due to the action of corticosteroids.13 Therefore, some LAIP markers were added to more clearly distinguish regenerative cells from residual leukemic cells after immunosuppression.
The cross-lineage markers CD66c, CD73, CD123 and CD304 were chosen on the basis of their prevalence at diagnosis and stability after treatment.11,14–19 The CD66c and CD123 are both associated with hyperdiploidy.14,15,19–21 Moreover, the CD66c is expressed in a large proportion of pediatric BCP-ALL, being significantly more frequent in the BCR-ABL1 positive cases than in the negative ones.15
The CD123 is normally expressed in CD34-negative BCP cells, but is often overexpressed in BCP-ALL, with a higher median fluorescence intensity (MFI) and percentage of positive cells, when compared to T-ALL.14,21 Patient ages are inversely correlated to the CD123 mean/median fluorescence intensity (MFI) in BCP-ALL.14 Although the CD123 is a characteristic marker of plasmacytoid dendritic cells (pDC), there is evidence that a subset of pDC can be developed from the common lymphoid progenitor (CLP).22 This subset of pDC has intermediate expression of the CD19 and also expresses the CD22 and CD81, similarly to B cells.22 These rare CD123+/CD19+ cells can be observed when more than 5 million events are acquired and should be distinguished from residual blast cells.
The CD73 and CD304 are usually negative in the normal BCP and are often overexpressed in BCP-ALL.17 The CD73 can be upregulated in leukemic B cells, but also in regenerative BCP. Therefore, misinterpretation must be attentively avoided. The CD304 can be expressed at low levels in pre-BI cells (CD34+), associated with the TEL-AML1 translocation.17 Although the CD304 expression remains positive in earlier post-therapy assessments, it may decrease in later evaluations.17,23
In addition, the CD15 and CD65 (cross-lineage markers) and NG2 (associated with MLL rearrangement)24 can be used as optional MRD markers for cases of pre-B cell ALL.20
Rational for T-ALL MRD markersT cell maturation markers are the nuclear terminal deoxynucleotidyl transferase (NuTdT), cytoplasmic CD3 (cyCD3), surface-membrane CD3 (SmCD3), CD5, CD7, CD1a, CD44, CD45RA and CD99.
The CD7 has a broad and usually intense expression in T-ALL cells and was included in all samples for detection of MRD. The SmCD3 is less frequently expressed in T-ALL. It is expressed in more mature T-ALL subsets, but cyCD3 expression occurs in most T-ALL cases.
CD1a, CD5 and CD45RA are proteins acquired on the T cell membrane during the maturation process. The CD1a is a hallmark of cortical T-ALL, while CD5 can be expressed in any T-ALL, but is often negative or present in <75% of early-T precursor-ALL (ETP-ALL) blast cells.25 The CD45RA is partially expressed in normal mature T cells, but it is observed only in more immature subtypes of T-ALL, which can be useful for detecting MRD.26
The CD99 is an antigen intensely expressed in the early stages of maturation of myeloid and lymphoid precursors, but there is no overexpression in cells outside the thymus.27 The CD99 is a MoAb useful in the detection of T-ALL MRD, due to its stability after treatment.27 The CD99 recognizes more immature T-ALL.20,27
Additionally, the immaturity markers CD10, CD34 and CD117 and the cross-lineage markers CD13 and CD33 are useful in the detection of ETP-ALL MRD.20,28
The CD44 is an optional marker for MRD detection. The CD44 upregulation may be involved in T-ALL leukemogenesis29 and it has been reported to be highly expressed in pediatric T-ALL.30,31
The CD56 is useful for excluding small subsets of NK cells that express cyCD3 from the T cell gate32, besides being a LAIP in the CD56+ T-ALL MRD detection.20,32
Of note, TdT, CD99, CD34 and CD10 can be found down-regulated during T-ALL induction therapy, while CD8 and CD45 may have a slight gain in MFI, compared to normal T cells. The CD2, CD3, CD4, CD5 and CD7 appear to be stable.33
Rationale for the selection of fluorochromesA successful reagent panel design strongly depends on the antibody clones and fluorochrome conjugates chosen for specific antigens.20,34
Fluorochromes selection in the panels must be in accordance with classic criteria: low density markers should be conjugated with bright fluorochromes, while highly expressed markers must be conjugated with dim ones. Another important tool for choosing fluorochromes is that they must have a restricted emission of fluorescence, which does not overlap into other channels (spillover).
Recommendations for technical proceduresStandard operating procedures (SOPs) (essential)This should be established by each laboratory and must be strictly followed, along with general quality control procedures, to ensure robust flow cytometry tests.35,36
Instrument settings (essential)The flow cytometer performance should be checked daily (or according to the manufacturer's recommendations) to ensure that the same parameters are always used to evaluate samples, allowing for an accurate analysis of the under-and over-expression of the markers, which is very important in MRD assessment.37,38 (See the detailed processes in the supplementary files.)
Quality control of monoclonal antibodies (essential)The MoAb titration process should provide a good resolution between the positive reference cell population and the negative population for each specific marker (stain index). It is necessary to test the stability of MoAb, because the storage and transportation conditions may affect its effectiveness, but also because lot-to-lot differences in fluorescence intensities can occur between consecutive batches of the same MoAb, even from the same manufacturer.39
Samples for MRD detection (essential)Bone marrow (BM) samples are preferable to peripheral blood (PB) for BCP-ALL MRD because the frequencies of BCP-ALL cells are from 1 to 3 logs higher in BM than in PB.40 In T-ALL, it can occur up to 1 log and therefore, PB can be used in this case.40 Quality of samples is essential for an adequate assessment of MRD. The BM samples collected in K3EDTA (7.5%) should be obtained from the first pull and the total volume should not exceed 2mL. Sample cellularity, hemodilution and clotting must be reported by the laboratory because these factors have great impact on the results.12 The storage should not exceed 24h upon collection, when prepared by the bulk lysis method.41 Otherwise, it can be stored for 48h at room temperature (RT) after collection. Samples must not be refrigerated, frozen, treated or stored with stabilized solutions, such as the TransFix (Cytomark) TM and Streck Cyto-ChexTM, because of the decrease in MFI, increase in background, affecting cell scatter and making it difficult to discriminate among the several cell subsets42
Sample preparation, staining and acquisition (essential)Conventional stain-lysis-wash sample preparation is described in supplemental files. We also recommend sample preparation and staining, as well as data acquisition using EuroFlow SOPs.38
To achieve greater sensitivity in MRD tests, sample bulk lysis protocol is preferentially recommended for sample preparation to achieve greater sensitivity in the MRD tests, reaching a sensitivity of 10−5.11 Bulk lysis procedures are available at https://www.euroflow.org/protocols.41
It should be emphasized that at least 1 million events must be acquired to achieve the sensitivity required for clinical use, which currently is 10−4, or 0.01%. This amount is feasible even in a 4-color flow cytometer. (See tests for validation of monoclonal antibodies.)
The staining processes must be subsequent to the bulk lysis and acquired on the same day. Otherwise, there is significant and progressive loss of cell viability.
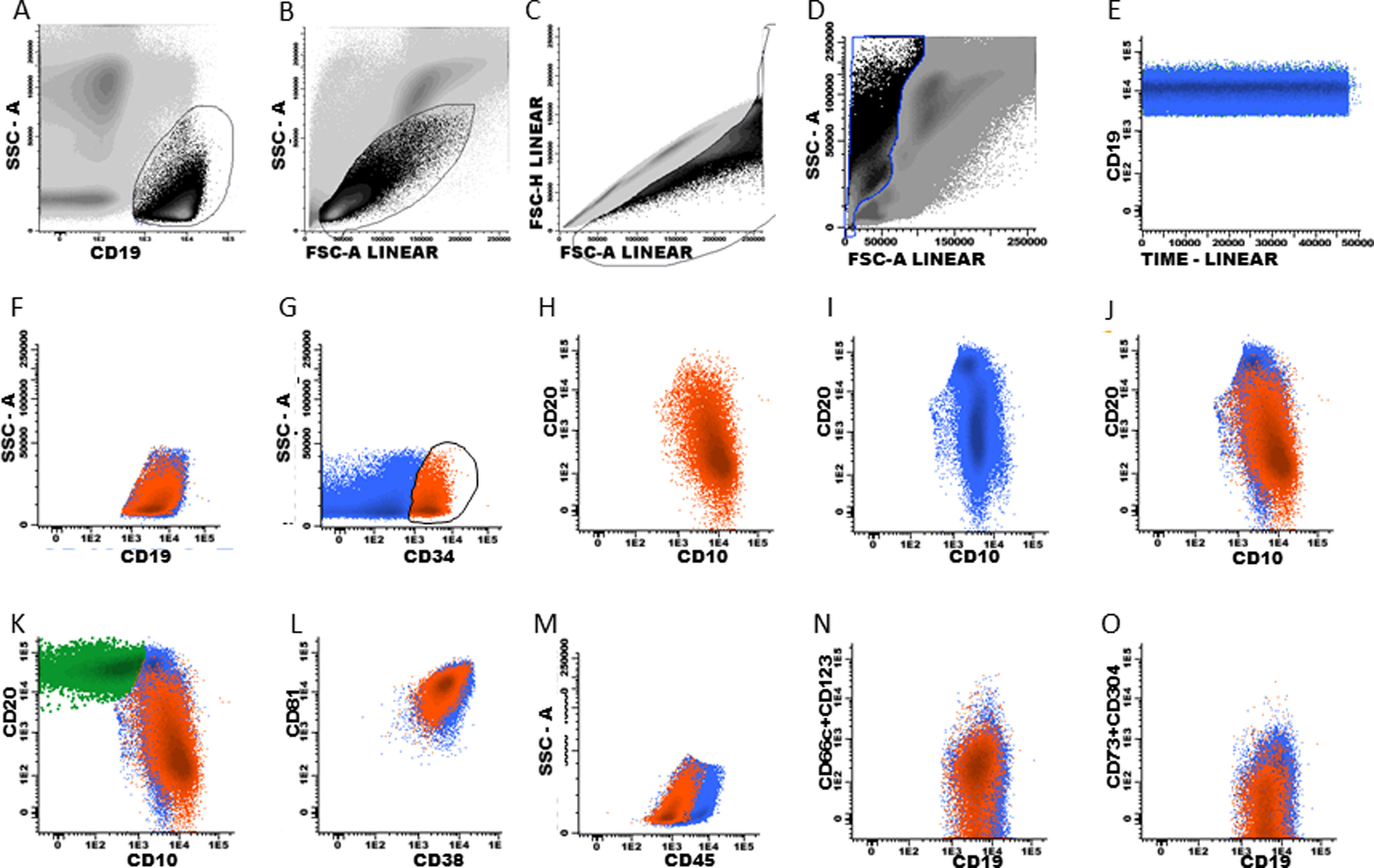
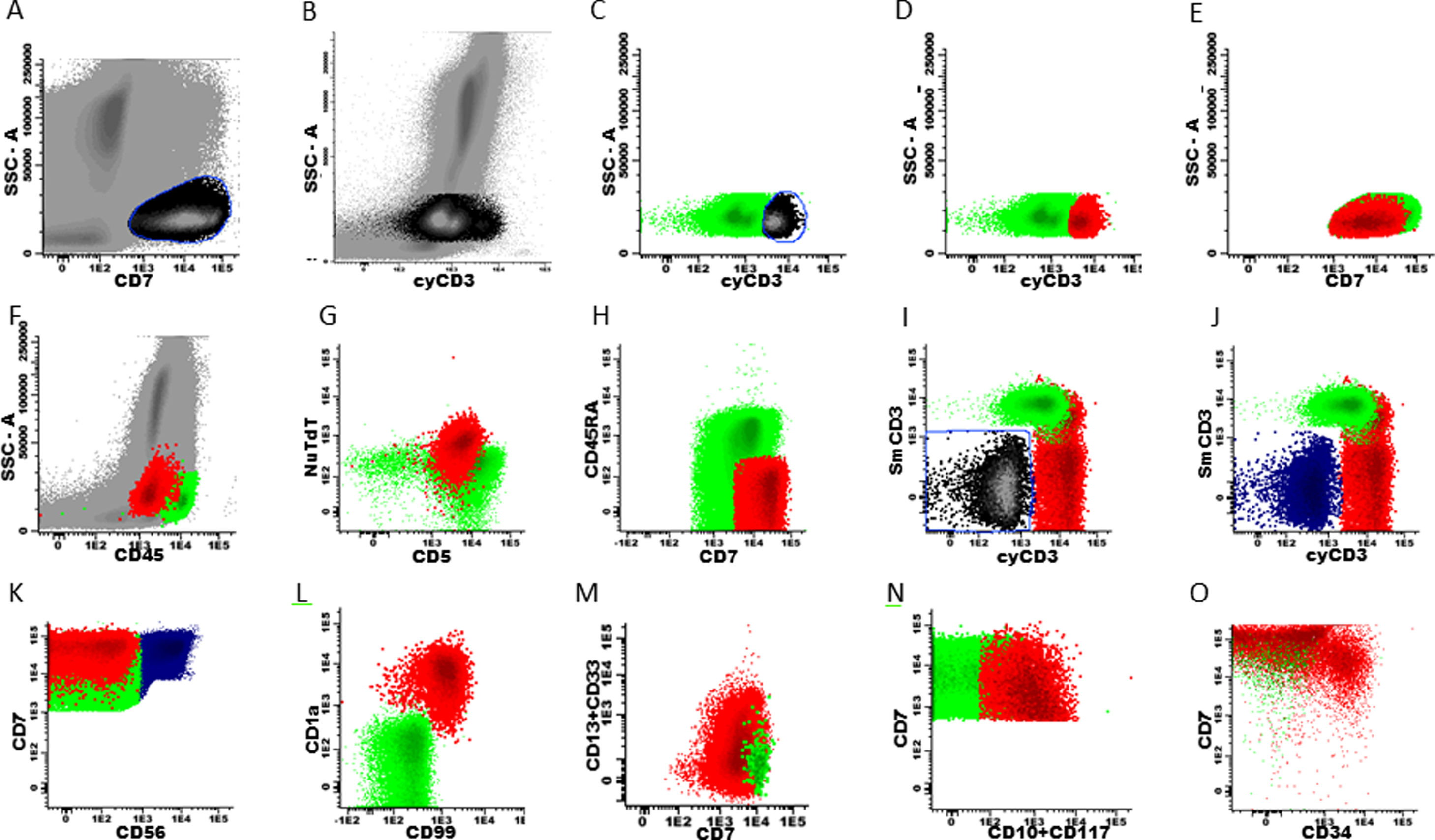
Data analysis: gating strategy for BCP-ALL and T-ALL (recommended)It must be emphasized that there are many gating strategies for ALL MRD and it depends on the expertise of each cytometry professional. Some suggestions that may be useful for the analysis of MRD in ALL are presented in flow cytometry figures (Fig. 1, 2 and 3) and in the supplementary material.
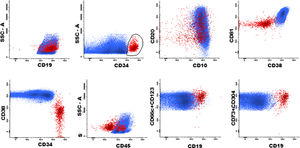
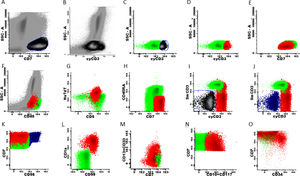
Marker expression from the T-ALL MRD panel, generated from different patient files. Initial gate in CD7positive cells x SSC (A and E) and sequentially in cyCD3 (B-D). Exclusion of NK cells (blue) from the T cell gate (I-K). Marker expression in normal T cells (green) and residual T-ALL cells (red) (D-O).
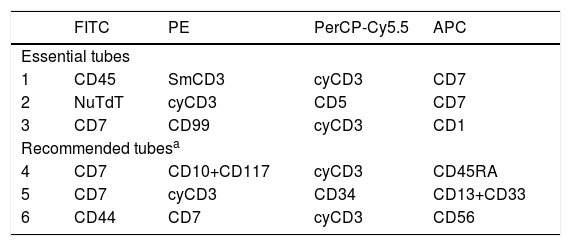
Using a traditional analysis, the initial gate is made on CD19+ cells versus the side scatter (SSC) plot (Fig. 1, A and F)); debris and doublets are excluded using two dot plots, FSC-H (in flow cytometer provided with this parameter) x FSC A, and FSC-A x SSC-A, in order to better quantify the analyzed sample cells (Fig. 1, B–D). A dot plot of time x CD19 must be included (Fig. 1E) to verify the stability of data acquisition (Fig. 1E).
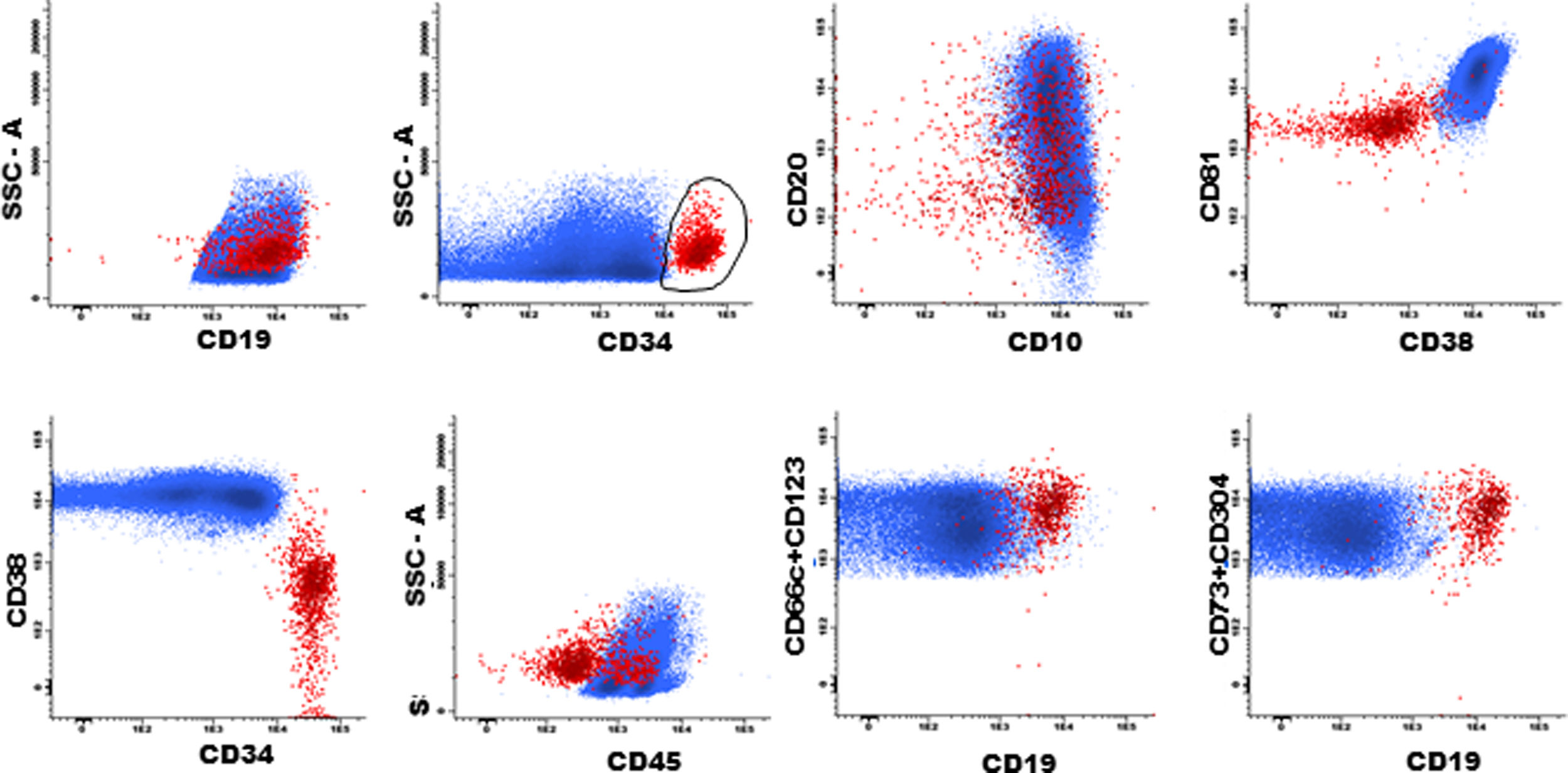
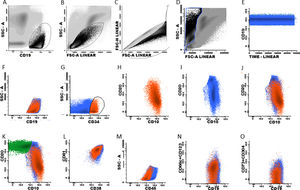
The next step is to verify that the intensity of each normal B cell marker expression, such as CD10, CD20, CD34, CD38, CD45 and CD81, is compatible with the stage of B cell maturation. Normal B cell precursors have partial expression of CD34 (Fig. 1G), heterogeneous expression of CD10 from bright (in CD34-positive cells) to moderate (Fig. 1, G–J), heterogeneous expression of the CD20 from negative to weak positive (Fig. 1, H–K), intermediate to high expression of the CD38, high expression of the CD81 (Fig. 1L), and weak expression of the CD45 (Fig. 1M). Fig. 1N and 1O show the negative expression of anomalous markers CD66c, CD123, CD73 and CD304 in normal B cells. Populations detected outside the normal pattern of maturation and asynchronisms of antigen expressions can be considered as positive MRD (Fig. 2). Of note, the normal pattern of regenerating BCP during different stages of treatment may be a "deviation from normal", and flow cytometry analysts must be familiar with, and aware of, these changes to properly analyze this type of sample.
Plasma cells and lymphoplasmacytic cells are excluded from the B cell gate by successive gates (Suppl. Fig. S-1, A–D). The next steps include the evaluation of the B cell maturation stages. Mature B cells have dim expression of CD38 and CD81 (Suppl. Fig. S-2, A and H) and bright CD20 expression, without CD10 and CD34 (Suppl. Fig. S-2, B, G and I). The marker expression patterns of the BCP were mentioned above (Suppl. Fig. S-2, C–K).
According to studies on B cell ontogeny: (i) B cell lineage-compromised progenitors express CD34 and CD38 before the upregulation of CD10 and CD19,43,44; (ii) lymphoid and early B cell progenitors do not express the CD19 and express the CD10 and CD34 and weak CD4545, and; (iii) B cell progenitors in fetal life do not express the CD10 and CD19 and have the potential for leukemogenesis.45 It is essential to include the evaluation of these subpopulations in the context of MRD. Supplemental Fig. S-3 shows the immunophenotype of the CD19-negative BCP.
Importantly, following anti-CD19 therapies, residual leukemic cells may have a CD19-negative immunophenotype and therefore, the MRD may not be detected.46,47 For these cases, alternative B cell markers, including the CD22 and CD24 are used to detect BCP-ALL MRD.48
Some patients with BCP-ALL may relapse with the lineage switch after CD19 therapies, especially those with KMT2A/MLL rearrangements, and flow cytometry professionals should be aware of this fact.49,50
For T-ALL MRDThe gating strategy for T-ALL can be initiated with both cyCD3 and CD7 positive cells, due to the higher frequency of expression of these markers in these leukemic cells, which gives them the position of backbone markers (Fig. 3, A–E). The same exclusion of debris and doublets must be made, like in BCP-ALL (Fig. 1B–D). Normal mature T cells express the SmCD3 and bright CD45, heterogeneous CD5 and partial CD45RA (Fig. 3, F–I). All other panel markers are not expressed in normal T cells of the bone marrow and peripheral blood, such as the NuTdT, cyCD3, CD1a, CD34, CD10, CD13, CD33, CD44, CD56, CD99 and CD117 (Fig. 3, G, J, L–O). The presence of at least two of these markers characterizes T-ALL residual cells and the sequential gates allow for the discovery of these anomalous populations. The NK cells are directly excluded from the T cell gate by the gating on CD56 positive cells, or as an alternative, on the CD7-positive and double negative cells for the cyCD3 and SmCD3 (Fig. 3, I–K).
MRD assessment request form (essential)It is important that laboratory professionals receive patient clinical data and information on previous immunophenotype results, from the diagnosis to the last MRD assessment. To achieve this, an exam request form containing the necessary information must be available to clinicians.
Reporting the MRD results to the clinician (essential)Recommendations for reporting results, according to the international flow cytometry societies (ICCS and ESCCA), are described in supplementary files. Essentially, the MRD should be reported as a percentage of the total nucleated cells in the bone marrow. The report must contain the number of events / cells acquired. In addition, complete information on the MRD detection and quantification levels should be reported to assess the depth of response to treatment. The MRD results should not only be reported as positive or negative.11,34,50–52 Furthermore, the report should inform if it was not possible to draw conclusions for any specific reason.
Tests for validation of monoclonal antibodiesThe performance of antibodies in the proposed combinations were tested to validate the MoAb panel designed herein.
Samples, material and methodsA total of 17 BM samples were collected in K3EDTA-containing tubes from patients, 10 with BCP-ALL (1 pre-B, 7 common and 2 mature), 4 with T-ALL (2 ETP, 1 cortical T-ALL and 1 pre-T), 2 with undetectable MRD (1 BCP-ALL and 1 T-ALL) and 1 non-leukemic. Results were acquired using 8-color FACSCanto II (BD)™ flow cytometers at two independent laboratories. A 4-color FACSCalibur (BD)™ was used to test the efficacy of the CellQuest-Pro (BD)™ software to acquire samples prepared by the bulk lysis process. The 4 color flow cytometer was capable of acquiring and recording 1–5 million sample events, when prepared with the bulk lysis protocol. The FACSDiva (BD) ™ software in the 8-color FACSCanto II (BD)™ FACSDiva was capable of acquiring more than 10 million events.
The MoAbs were titrated to achieve an appropriate staining index.
To verify the reproducibility of the antibody expression, the same samples were processed in parallel, using the previously validated 8-color panel, according to EuroFlow standard operating procedures (SOPs), and also using the proposed 4-color panel, complying with all the SOP recommendations described above. The MoAb clones, fluorochromes and titration are provided in Supplemental Table S-1.
The qualitative comparison of the MoAb expression patterns in normal and leukemic cell populations was performed between the paired samples. Differences between the ALL cell and its control unaffected counterpart MFI were assessed by applying the mean value of the leukemic B cell MFI with the normal mature B cell MFI, as well as that of the leukemic T cell with the normal mature T cell. These parameters were evaluated in each sample.
ResultsThe samples tested herein showed the same antibody expression in both experiments, using panels of 4 and 8 colors, demonstrating the effectiveness of the proposed MoAb panels. There was no loss of the marker expression, nor was there any spillover or non-specific labeling between the panel markers (data not shown). The median and range of the MFI intensity values of each MoAb are presented in Supplemental Table S2.
ConclusionsConsidering the importance of validating standardized flow cytometry protocols for MRD detection, this document may be useful for Brazilian laboratories as a guideline for this purpose. The rigorous use of these protocols can enable laboratories to provide reliable MRD results for clinical use. Although these procedures have been addressed for 4-color MFC settings, most of them also apply to ≥ 8-color MFC, which currently is the most recommended platform for MRD detection, given its superior sensitivity and accuracy, as well as for financial resource optimization. Therefore, the migration of the MFC from 4 to 8 colors should be stimulated due to the irrefutable clinical benefit for patients, although it requires further training. Furthermore, all these processes must be periodically reviewed, constantly incorporating new knowledge, for continuous technical development, always with the main objective of guaranteeing more precise results for a better clinical management of patients with ALL.
AuthorshipMRVIC: proposed and designed the study; MRVIC and MPB: performed MFC experiments, analyzed MFC data and wrote the manuscript; MRVIC: designed MFC analysis strategies; all authors: discussed the recommendations, reviewed and approved the manuscript.
Conflict of interestThe authors declare no conflict of interest
The group is thankful to Becton & Dickinson for kindly supplying several of the MoAbs validated by the laboratories.
We also thank Joana Espricigo Conte-Spilari, Camila Marques Bertolucci, Alef Rafael Severino, Diana AC Kluck, Raysa Rosseto Rodrigues, Regielly Cognialli and Vanessa Kukla for technical support.
SBTMO MRD working group: A Seber, A Wagner, AP Alegretti, AP Azambuja, BE Gomes, DMV Avelar, ES Costa, EX Souto, F Gevert, FM Furtado, G Pimenta, I Lorand-Metze, M Malvezzi, MRV Ikoma-Colturato, M Yamamoto, MP Beltrame, NLL Silva, NS Bacal, SICP Ferreira, V Pires.