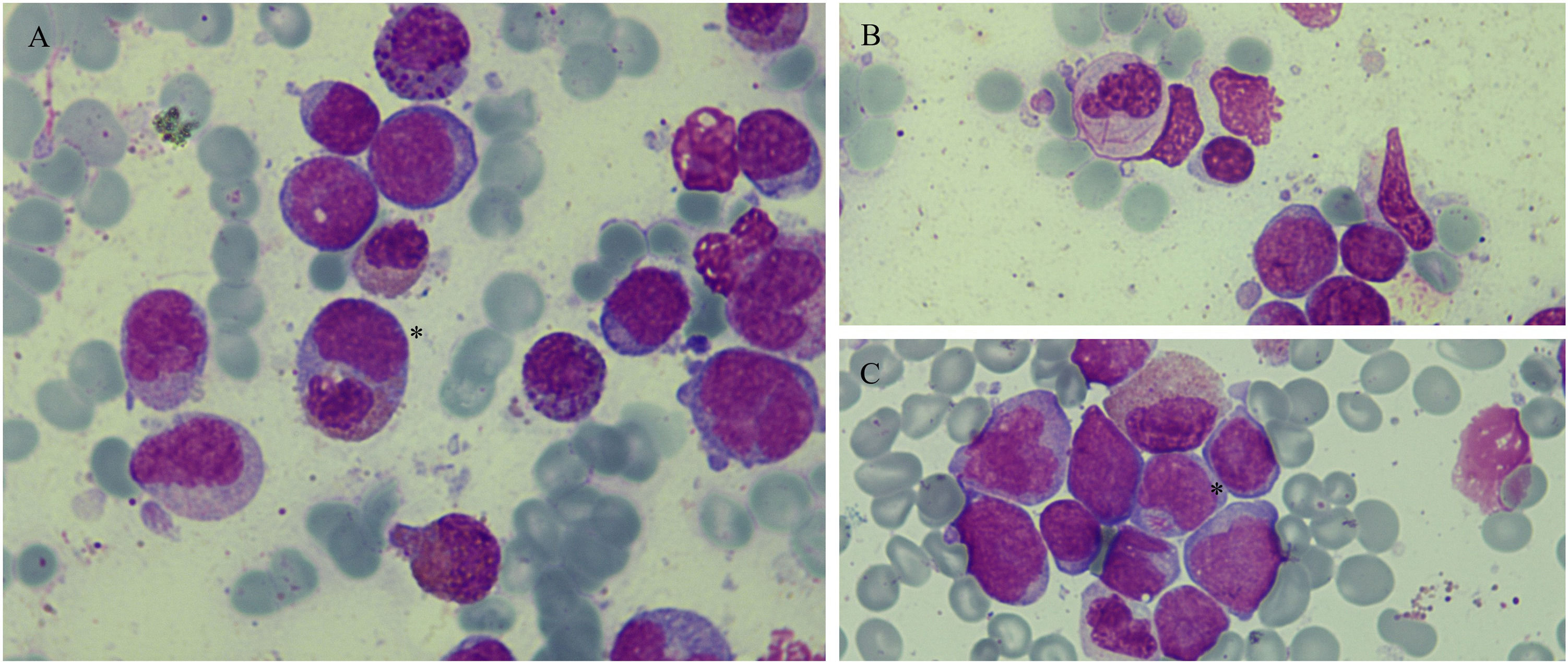
A 43-year-old man presented with malaise, odynophagia and palatine petequial hemorrhage. A full blood count showed: hemoglobin 137 g/L, platelets 50×109/L, and leukocytes 40.17×109/L with blasts in the blood film. Coagulation test results were within normal range. The bone marrow aspirate examination revealed a hypercellular marrow with the presence of 62% blast infiltration with heterogeneous morphology. Most blasts were big and agranular; but others were medium-size, some had cleaved nuclei; and less with bundles of Auer rods (faggot cells). Additionally eosinophils were increased, some with atypical dense and coarse granulation. Sporadically, an eosinophil phagocytosis by a blast was seen (Figure 1A). Some neutrophils presented faggot cell appearance (Figure 1B-C). The blasts were positive for myeloperoxidase and alpha-naphthyl acetate esterase. Flow cytometry identified 31% of blasts: CD34+, CD117+, HLADR+, CD13+, CD33++, CD123+, MPO+; and increase of monocytes (38%): CD64++, CD14+, CD33++, CD11b+, CD38+, HLADR+, CD36+, CD4bi+, MPO+. Molecular studies confirmed a CBF::MYH11 rearrangement. A next generation sequencing study revealed four different mutations in NRAS (c.182A>G; c.181C>A c.38G>A and c.34G>A). cKIT was not mutated.
Bone marrow smear showing infiltration by blasts (all figures May-Grünwald Giemsa staining; magnification: x1000). A: Phagocytosis of an eosinophil by a blast cell (*) and eosinophils with coarse basophilic granulation. B: Neutrophil faggot cell. C. Blast cell with monocytic appearance with bundles of Auer rods (*).
With diagnosis of acute myeloid leukemia (AML) with CBF::MYH11 rearrangement, the patient started induction treatment with idarubicin, cytarabine (“3+7”) and gemtuzumab ozogamicin (GO) and three ulterior consolidations based on high dose cytarabine and GO. Nine months after initial diagnosis he presented cytological and molecular relapse (without associated NRAS mutations in a next generation sequencing study) which was treated with fludarabine, cytarabine and idarubicin (FLAG-Ida) and consolidation with hematopoietic stem cell transplant (HSCT) (related donor). Eight months after the procedure he presented molecular relapse and received five cycles of venetoclax-azacitidine and a second HSCT (non-related donor). Currently he maintains complete response with undetectable CBF::MYH11.
Phagocytosis by blasts is an extremely atypical finding in acute myeloid leukaemia, with the erythrophagocytosis phenomenon being most frequently described in leukaemias with rearrangements involving the KAT6A gene or leukaemias with megakaryocytic or monocytic-histiocytic morphology.1,2 Eosinophil phagocytosis is rarer.
Although the presence of faggot blast cells is considered nearly a pathognomonic finding of acute promyelocytic leukaemia (APL), it has been reported in sporadic cases of core-binding factor (CBF) leukaemias. However, faggot-type cells in final stages of granulopoiesis is even a rarer finding. The presence of faggot neutrophils can be seen in APL during all-trans retinoic acid treatment and in newly diagnosed non-promyelocytic leukemia (most frequent CBF-AML followed by mixed phenotype). Some authors suggest that the presence of faggot neutrophils at AML onset could raise the suspicion of CBF-AML rather than APL.3,4