Myelodysplastic syndromes (MDS) are a heterogeneous group of hematological malignancies characterized by dysplasias, ineffective hematopoiesis and risk of acute myeloid leukemia transformation. Approximately 90% of MDS patients present mutations in genes involved in various cell signaling pathways. Specialized DNA polymerases, such as POLN, POLI, POLK, POLQ, POLH, POLL and REV3L, insert a nucleotide opposite replication–blocking DNA lesions in an error-prone manner and, in this way, sometimes can actively promote the generation of mutation. For the best of our knowledge, has not been described the mutations of these genes in MDS. DNA target sequencing CDS regions of the REV3L gene was performed in a 58-year-old man diagnosed as High Risk Myelodysplastic Syndrome. The patient presented very low hemoglobin, increased number of blasts, karyotype:47,XY,+8[6]/47,XY,del(7)(q32),+8[7], no response to hypomethylating therapy (decitabine), all markers of poor prognosis. Target sequencing identified a mutation c.9253-6T>C REV3L (Substitution - intronic) with VAF (variant allele frequency)=16% considered pathogenic according to Functional Analysis through. Hidden Markov Models (FATHMM). This is the first evidence of REV3L mutation in MDS and, of utmost importance, associated with poor prognosis.
Myelodysplastic syndromes (MDS) are a heterogeneous group of hematological malignancies characterized by dysplasias, clonal and ineffective hematopoiesis, peripheral cytopenias and risk of progression to acute myeloid leukemia. Recently, studies using sequencing techniques such as next-generation sequencing (NGS) have demonstrated frequent mutations in approximately 90% of MDS patients in genes involved in various cell signaling pathways, including RNA-splicing machinery (U2AF1, SRSF2 and SF3B1), epigenetic regulators (EZH2, ASXL1), cohesin components, transcription factors (EVI1, RUNX1, GATA2), DNA damage response and signal transduction molecules (JAK2, KRAS, CBL). Mutations in TP53, NRAS, RUNX1 and IDH1/IDH2 have been reported to influence overall survival in multivariate analyses while SF3B1 is associated with favorable prognosis.1–3
DNA lesions may be spontaneous or induced by radiations or chemicals, causing genomic alterations such as nucleotide substitutions, amplifications, deletions, rearrangements or chromosomal loss. To ensure genome stability, cells use DNA damage response (DDR) to sense and repair damage.3 Despite the efficient action of repair systems that remove lesions in DNA in an error-free manner, some lesions can escape these mechanisms.3 Thus, a second response to damaged DNA, termed DNA damage tolerance (DDT), has evolved to promote replication through and beyond an altered template, leaving the damage to be repaired at a later time point, thus lowering the overall risk of replication fork collapse and genome instability4 One of the DDT mechanisms, the translesion DNA synthesis (TLS) is a DNA damage tolerance process which contributes to cell survival by bypassing the unrepaired DNA lesions.5 Specialized DNA polymerases (POLs), such as POLN, POLI, POLK, POLQ, POLH, POLL and REV3L insert a nucleotide opposite replication–blocking DNA lesions in an error-free or error-prone manner and, in this way, sometimes can actively promote the generation of mutations.4
Material and methodsWe performed a DNA target sequencing 108 amplicon regions of the REV3L gene from bone marrow sample of MDS patient at diagnosis. Amplicon coverage was 400 readings. The genome of interest used for alignment all of the sequencing reads was Homo sapiens (UCSC hg19) using the NextGENe software. Library preparation was performed according to Illumina Truseq Custom Amplicon (TSCA) standard procedure and run on MiSeq with the use of version 2.2 chemistry (Illumina, San Diego, CA, USA). The Q30 quality score bases was of 93.60%. For initial analysis, paired-end reads (175bp) were de-multiplexed into individual BAM files and aligned to UCSC Hg19 (GRCh37) by the on-board Illumina reporter software Miseq Reporter Software 2.4 with the use of the TruSeq Amplicon software. The single-nucleotide variations (SNVs) with ≥2% variant allele frequency (VAF) and more than 400 reads depth was considered significant. For secondary analysis, the variants candidates were loaded into a database NextGENe using BAM files for viewing of each variant. As tertiary analysis, we detected the variants in SNPs, mutations or variants of unknown significance using dbSNP, COSMIC and ESEMBL databases.
Case reportA 58-year-old man was referred for investigation of anemia. At physical examination, the patient was pale and presented petechiae. The hematology profile showed hemoglobin of 5.7g/dL, leucocytes of 1.200/mm3 and platelets of 22.000/mm3. The bone marrow smear presented dyseritropoiesis, dysgranulopoiesis and increased number of blasts (18%). Karyotype analysis of bone marrow cells showed clonal evolution: 47,XY,+8[6] and 47,XY,del(7)(q32),+8[7] (Figure 1). The patient was diagnosed as Refractory Anemia Excess Blasts II (RAEB II) according to World Health Organization (WHO)6 and IPSS-R (revised-International Prognostic Score Systems) was considered High Risk. The patient was treated with Decitabine (20mg/m2) without response and died due to sepsis after two-month follow up. DNA target sequencing CDS regions of the REV3L gene from bone marrow sample was performed at diagnosis.
We evaluated all the polymerases with TLS activity (POLN, POLI, POLK, POLQ, POLH, POLL and REV3L) by NGS and detected a mutation in REV3L gene. Reversionless 3-like (REV3L) is the catalytic subunit of DNA polymerase zeta (POLZ) complex formed by REV3 and an accessory subunit REV7.7,8 The encode protein plays an important role in the translesion synthesis (TLS). However, the less fidelity of the REV3L due to its absence of exonuclease activity, compared with the high-fidelity replicative DNA polymerases, makes its synthesis translesion error-prone that causes spontaneous mutations. REV3L maintains genomic integrity by inserting a substitute nucleotide in the opposite DNA adducts, which increases the mutation rate and contributes to carcinogenesis.7–9 Diseases associated with REV3L include Moebius Syndrome and Poland Syndrome.10 Additionally, recent studies have presented mutations in REV3L gene in myeloid neoplasms, but not yet in MDS.11
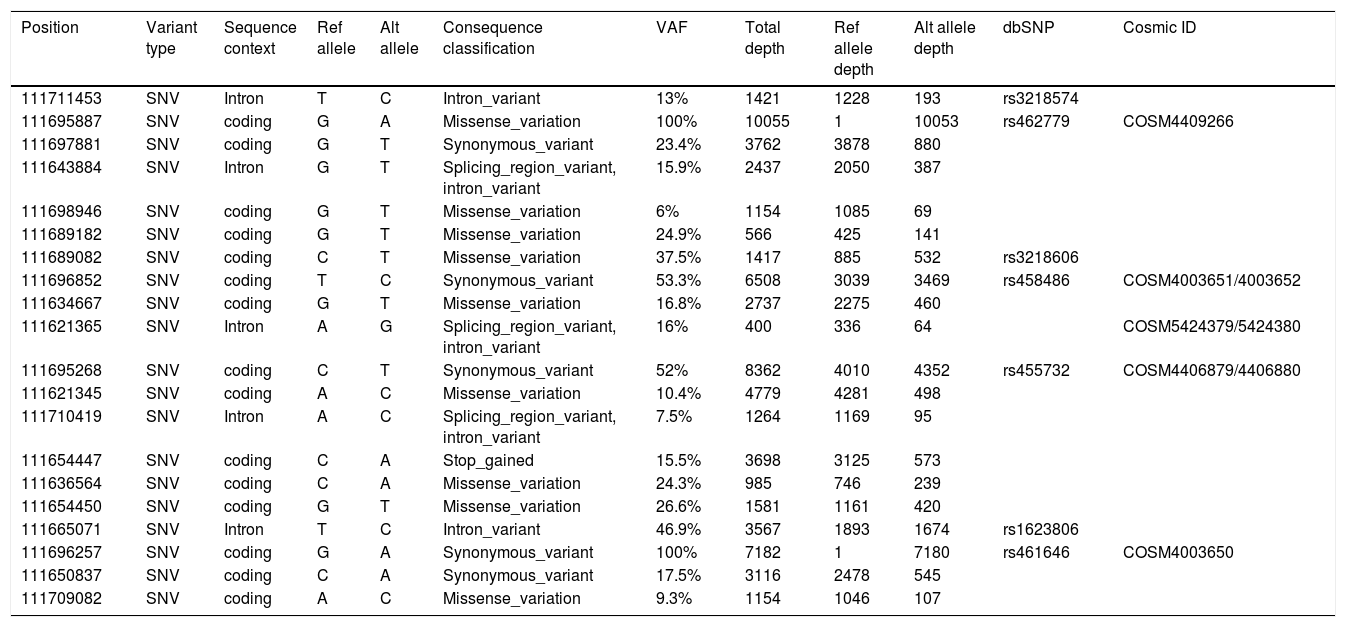
Sequencing analysis showed 20 single-nucleotide variants of which, 15 in coding regions and 5 in intronic regions (Table 1). Variation consequences were calculated following the guidelines at ESEMBL databases and Truseq Amplicon software in missense variations (09), Synonymous (05), Stop Gained (01), intronic variants (05) and, more important, sequencing showed the presence of mutation c.9253-6T>C REV3L (Substitution - intronic) with VAF (variant allele frequency)=16% which is considered pathogenic according to Functional Analysis through Hidden Markov Models (FATHMM) (Table 1). According to Catalogue of Somatic Mutations In Cancer (COSMIC), this mutation was described in a case of Acute Myeloid Leukemia with mutation ID COSM5424379 in project entitled ICGC (LAML-KR): Acute Myeloid Leukemia – KR.
Variant calling of somatic variants in the case herein reported.
| Position | Variant type | Sequence context | Ref allele | Alt allele | Consequence classification | VAF | Total depth | Ref allele depth | Alt allele depth | dbSNP | Cosmic ID |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 111711453 | SNV | Intron | T | C | Intron_variant | 13% | 1421 | 1228 | 193 | rs3218574 | |
| 111695887 | SNV | coding | G | A | Missense_variation | 100% | 10055 | 1 | 10053 | rs462779 | COSM4409266 |
| 111697881 | SNV | coding | G | T | Synonymous_variant | 23.4% | 3762 | 3878 | 880 | ||
| 111643884 | SNV | Intron | G | T | Splicing_region_variant, intron_variant | 15.9% | 2437 | 2050 | 387 | ||
| 111698946 | SNV | coding | G | T | Missense_variation | 6% | 1154 | 1085 | 69 | ||
| 111689182 | SNV | coding | G | T | Missense_variation | 24.9% | 566 | 425 | 141 | ||
| 111689082 | SNV | coding | C | T | Missense_variation | 37.5% | 1417 | 885 | 532 | rs3218606 | |
| 111696852 | SNV | coding | T | C | Synonymous_variant | 53.3% | 6508 | 3039 | 3469 | rs458486 | COSM4003651/4003652 |
| 111634667 | SNV | coding | G | T | Missense_variation | 16.8% | 2737 | 2275 | 460 | ||
| 111621365 | SNV | Intron | A | G | Splicing_region_variant, intron_variant | 16% | 400 | 336 | 64 | COSM5424379/5424380 | |
| 111695268 | SNV | coding | C | T | Synonymous_variant | 52% | 8362 | 4010 | 4352 | rs455732 | COSM4406879/4406880 |
| 111621345 | SNV | coding | A | C | Missense_variation | 10.4% | 4779 | 4281 | 498 | ||
| 111710419 | SNV | Intron | A | C | Splicing_region_variant, intron_variant | 7.5% | 1264 | 1169 | 95 | ||
| 111654447 | SNV | coding | C | A | Stop_gained | 15.5% | 3698 | 3125 | 573 | ||
| 111636564 | SNV | coding | C | A | Missense_variation | 24.3% | 985 | 746 | 239 | ||
| 111654450 | SNV | coding | G | T | Missense_variation | 26.6% | 1581 | 1161 | 420 | ||
| 111665071 | SNV | Intron | T | C | Intron_variant | 46.9% | 3567 | 1893 | 1674 | rs1623806 | |
| 111696257 | SNV | coding | G | A | Synonymous_variant | 100% | 7182 | 1 | 7180 | rs461646 | COSM4003650 |
| 111650837 | SNV | coding | C | A | Synonymous_variant | 17.5% | 3116 | 2478 | 545 | ||
| 111709082 | SNV | coding | A | C | Missense_variation | 9.3% | 1154 | 1046 | 107 |
POSITION: Position within reference chromosome; VARIANT TYPE: Type of variant including single nucleotide variant (SNV), insertion and deletion; SEQUENCE CONTEXT: Location of the variant based on annotations of the reference genome; REF ALLELE: The reference allele; ALT ALLELE: The altered allele; CONSEQUENCE CLASSIFICATION: Predicted transcript consequence; VAF: variant allele frequency; TOTAL DEPTH: Number of reads aligned at this position; REF ALLELE DEPTH: Number of reads containing the reference allele; ALT DEPTH: Number of reads containing the variant allele; dbSNP: Identifier in the Single Nucleotide Polymorphism Database (dbSNP), a free public archive for genetic variation within and across different species developed and hosted by the National Center for Biotechnology Information (NCBI); COSMIC: The numeric identifier for the variant in the Catalogue of Somatic Mutations in Cancer (COSMIC) database.
Here, we present for the first time the presence of single-nucleotide variants and mutations on REV3L gene in patient diagnosed as MDS. Of utmost importance, our report is the first study to detect mutation c.9253-6T>C REV3L in MDS. This finding suggests this mutation is an important step during acute myeloid leukemia transformation. Mutations in REV3L predispose to double strand breaks in DNA which are strongly associated with chromosomal abnormalities.12 Our case reported herein presented trisomy 8 and deletion 7q as clonal evolution, all markers of poor prognosis in MDS and genomic instability.
We suggest this mutation is associated with great genomic instability in MDS because REV3L has no exonuclease activity compared to high-fidelity replicative DNA polymerases, making its function as error-prone which causes spontaneous and DNA damage-induced mutagenesis as here presented with clonal evolution with deletion 7q. The lack of the TLS synthesis may provoke chromosome rearrangements and even chromosome loss and inactivation of the REV3L gene causes a high frequency of chromosomal breaks. Thus, REV3L is important for maintaining genome stability.
Author contributionRTGO, GBCR, IGFF, SMMM and RFP designed the study, provided patient materials and were responsible for collection and assembly of data. HLRJ, RTGO, DPB, GMC performed the molecular procedures and analyzed the data. All drafted and edited the manuscript. All authors have approved the final version of manuscript before publication.
FundingThis study was partially supported by the National Council for Scientific and Technological Development (CNPq) (grant number # 420501/2018-5 and # 424542/2016-1).
Conflict of interestThe authors declare no conflicts of interest.
Ethical approvalAll procedures were approved by the Ethics Committee of UFC (#1.292.509) and are in accordance with the 1964 Helsinki declaration and its later amendments. Informed consent was obtained from all individual participants included in the study.



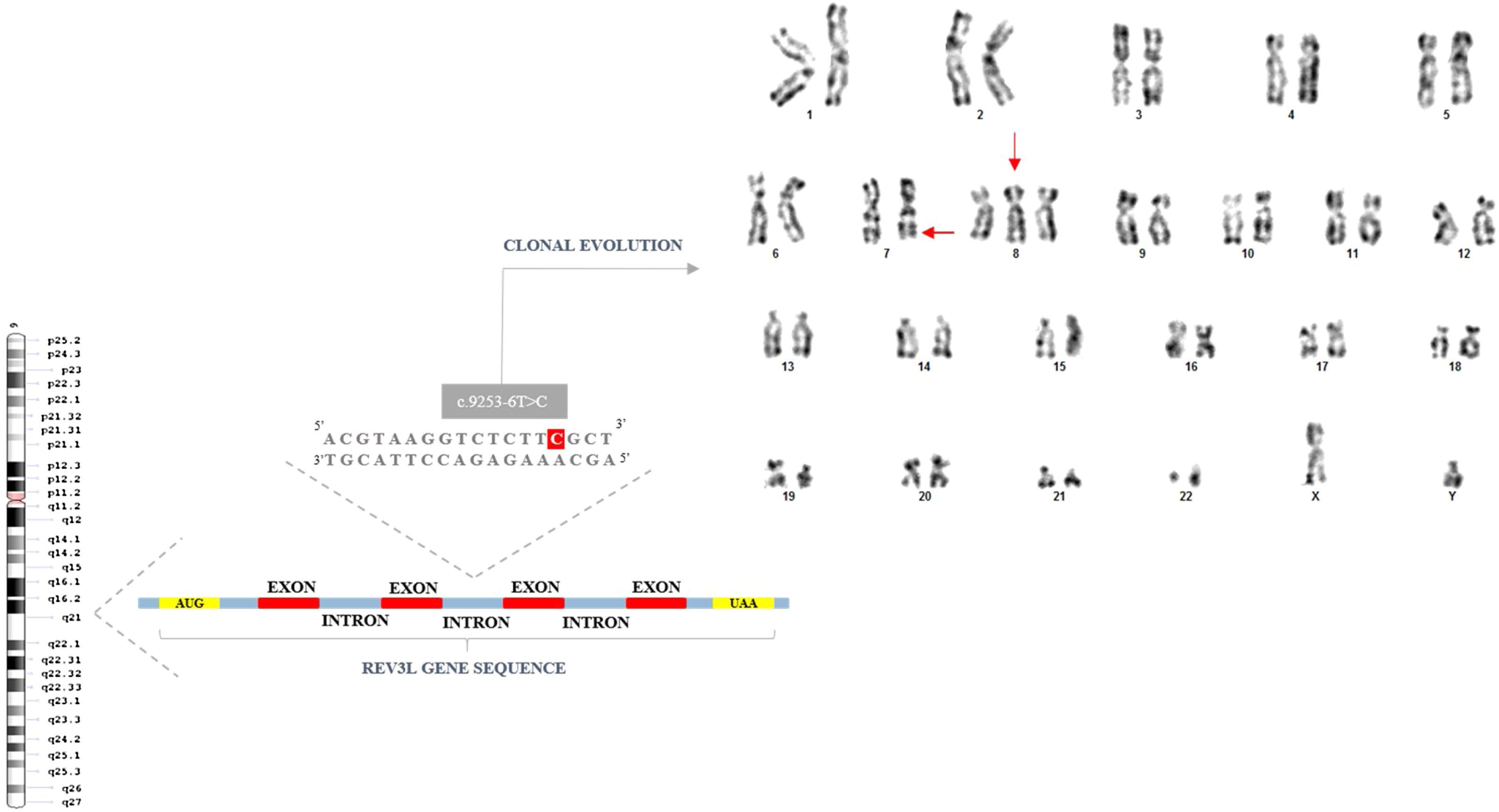
![Next-generation sequencing showing REV3L gene mutation in MDS patient with clonal evolution. karyotype: 47, XY,+8[6]/47,XY,del(7)(q32),+8[7]. Next-generation sequencing showing REV3L gene mutation in MDS patient with clonal evolution. karyotype: 47, XY,+8[6]/47,XY,del(7)(q32),+8[7].](https://static.elsevier.es/multimedia/25311379/0000004300000003/v1_202109150707/S253113792030078X/v1_202109150707/en/main.assets/thumbnail/gr1.jpeg?xkr=ue/ImdikoIMrsJoerZ+w93OM6WmS6o9DeZl+SVh74uo=)



