The administration of antenatal and postnatal Rh immunoglobulin prophylaxis significantly prevents Rh D alloimmunization during pregnancy. A dose of 20μg anti-D IgG protects against 1mL of D-positive red blood cells or 2mL of whole blood. Rh D-negative women if unsensitized receive antenatal anti-D prophylaxis of 300μg at 28 weeks of pregnancy and postnatal prophylaxis if the baby is Rh D positive. Initial trials concluded that postpartum immunoprophylaxis decreased the incidence of Rh D immunization from 12–13% to 1–2% which was further shown to be reduced to 0.1% thanks to antenatal prophylaxis.1 Rh immunoglobulin (RhIg) administration is quite safe during pregnancy although RhIg can cross the placenta and cause hemolysis of the red cells of D-positive fetuses. Unfortunately data is sparse on this serious adverse event. Herein we present an unusual case of hemolytic disease of newborn (HDN) with positive Direct Coombs’ Test (DCT) due to antenatal RhIg prophylaxis in an Rh-negative mother.
Case reportA 28-year-old primipara with blood group O Rh negative, delivered a male baby weighing 2.6kg at 37+2 weeks by emergency cesarean section in view of fetal bradycardia. During 30 weeks of pregnancy, her antibody screen was negative and she received a prophylactic dose of 300μg Rh immunoglobulin (RhIg, Rhogam). She had an otherwise uneventful pregnancy.
Since the mother was Rh D negative, a requisition for blood grouping, Rh typing and DCT of the baby was made. Blood group of the baby was O Rh D positive (tube technique). DCT was performed using column agglutination technology (Ortho clinical diagnostics, USA) and was found to be positive (3+) for polyspecific anti-human globulin (IgG+ C3d) having IgG specificity. Anti-D antibody was identified in the baby's serum (Resolve Panel A, Ortho Clinical Diagnostics, USA).
Antibodies were eluted from the baby's red cells using the acid elution technique (Diacidel, Bio-Rad, Germany). Antibody screening was negative for the supernatant of the last wash. The eluate was positive for anti-D. The mother's blood group was O Rh D negative while the father's blood group was O Rh D positive. Antibody screening and identification of mother's sample revealed the presence of anti-D. The anti-D titers of the eluate and the mother's sample were 1:4 and 1:8 respectively. The mother was given 300μg RhIg, 6h postpartum.
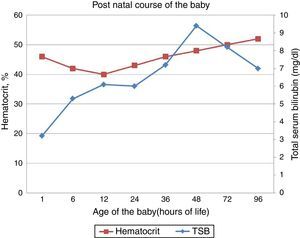
The baby's Activity, Pulse, Grimace, Appearance, Respiration (APGAR) scores were 8 and 9 at 1min and 5min respectively. At birth, cord total serum bilirubin (TSB) and hematocrit were 3.2mg/dL and 46% respectively. At 6h of life, TSB was 5.3mg/dL and it was noticed that the baby had jaundice. Reticulocytes were 6% (normal range 0.5–2%). glucose-6-phosphate dehydrogenase (G6PD) deficiency testing was negative. There was no evidence of sepsis. The child received phototherapy in view of hyperbilirubinemia as per our institute protocol. The postnatal evolution of the baby is shown in Figure 1. Antibody screening of the mother after six months was negative. Baby had no residual sequelae during subsequent visits.
DiscussionTSB concentrations above 5mg/dL on day one of life in a term neonate indicates pathologic jaundice. Hemolytic anemia should be considered in the setting of ABO/Rh incompatibility, jaundice within 24h, positive DCT, raised reticulocyte count, raised serum bilirubin, presence of pallor or evidence of hemolysis in peripheral blood smear. In this case, along with these features, antibody titers were low and six-months follow-up analysis showed negative antibody screening in mother favoring RhIg induced HDN. Such secondary hemolysis is transient. ABO incompatibility was ruled out. Rh alloimmunization has occurred even in first pregnancy leading to HDN due to early trimester fetomaternal hemorrhage or failure of RhIg. In such cases, anti-D will be persistent being detected in early trimester of second pregnancy.2 Rh Immunoglobulin generally disappears within 6–8 weeks of administration.
To the best of our knowledge, this rare case is the first report from India. Incidence of positive direct antiglobulin test (DAT) in babies born to Rh negative mothers ranges from 2 to 30%. When mothers were given two doses of anti D immunoglobulin antenatally, 20% of the Rh D positive babies showed positive DAT. In another study, two out of 1238 (0.16%) Rh positive newborns were affected by passive transfer of maternal RhIg. Cohen et al. in 2014 reported a case of passive transfer of RhIg inducing HDN.3 Also a high grade DAT is a strong predictor of phototherapy.4 Maayan-Metzger et al., in their retrospective analysis, concluded that there was no significant risk of hemolysis due to RhIg among preterm (28–34 weeks) Rh D positive babies born to Rh negative mothers and further prospective studies were required to confirm the correlation.5
To conclude, antenatal and postnatal RhIg prophylaxis for all Rh-negative mothers who are unsensitized is the standard practice in vogue. Blood grouping and DCT is mandatory for all newborns of Rh-negative mothers. Although RhIg administration to Rh D negative mothers is considered safe, positive DCT in those D positive neonates should be further evaluated to rule out the possibility of RhIg induced HDN. Follow-up of mother and baby will assist in differentiating hemolysis caused by Rh immunization from that of RhIg prophylaxis.6
Conflicts of interestThe authors declare no conflicts of interest.