It is estimated that the human T lymphotropic virus type 1 (HTLV-1) infects approximately 20 million people worldwide. Associated diseases, however, manifest only in 5–10% of infected individuals.1 Studies have shown that HTLV-1 is endemic in Southern Japan, the Caribbean, South America, Melanesian islands, Papua New Guinea, the Middle East and Central and Southern Africa. In Brazil, HTLV-1 infection is considered a public health concern as the country figures among the endemic areas in the world with prevalence rates from 1% to 5%.1
Since the discovery of HTLV-1 in 1979, it has been clearly associated with hematologic disorders, specifically with adult T-cell leukemia/lymphoma (ATL), an aggressive neoplasm with poor prognosis.1 Although ATL has clinical forms with different manifestations, it is generally characterized as a clonal proliferation of CD4+ T cells containing randomly distributed HTLV-1 proviral integration sites. The onset of the disease usually occurs 20–30 years after viral infection and is primarily associated with vertical transmission, mainly through breastfeeding by a seropositive woman.1
Apart from ATL, other diseases have been consistently linked to HTLV-1 infection over the years. Tropical spastic paraparesis/HTLV-1-associated myelopathy (TSP/HAM) more frequently affects women, with onset ranging from years to decades after infection, at an average age of 40 years.1 With an insidious manifestation and chronic evolution, HAM/TSP takes years to progress from the onset of symptoms, such as weakness and spasticity of one or both legs and minor sensory changes, to wheelchair confinement and bowel/bladder incontinence.1 Other diseases such as uveitis, infective dermatitis, pulmonary and rheumatic disorders such as rheumatoid arthritis and Sjögren's syndrome, as well as other autoimmune conditions such as bronco-alveolar pneumonitis, autoimmune thyroiditis and endemic polymyositis, have also been associated with HTLV-1 infection.1,2 In addition, in the last two decades, HTLV-1 has been reported in association with other blood diseases, such as acute myeloid leukemia,3 idiopathic thrombocytopenic purpura4 and myelodysplastic syndrome.5
Case reportA 27-year-old woman was referred for a hematology consultation to investigate pancytopenia. At admission, she had spastic paraparesis, and bowel and bladder dysfunction, clinical signs consistent with the initial presentation of HAM/TSP. Her symptoms started as a progressive weakness of the legs when she was 22 years old. The patient was born in a known endemic area for HTLV-1 infection, located in the Northern Region of Minas Gerais State, Brazil. Although there was no history of blood transfusion, she reported that she was breastfed by a wet nurse. HTLV-1 testing was performed and the positive enzyme immunoassay results were confirmed by western blot. No alterations were found in cerebrospinal fluid analysis or a computed tomography (CT) scan of the lumbosacral spine. At admission, she presented with pancytopenia that had been investigated in another hospital in 1996 by bone marrow aspirate and biopsy; at that time, no abnormalities were found. These results could not be checked due to the lack of material.
Later in the same year, the patient was hospitalized for another investigation of the pancytopenia and metrorrhagia. During her hospitalization, the patient received 600mL of packed red cells and daily platelet transfusions. The absolute neutrophil count ranged from 1.310×109/L to 2.060×109/L (reference range [RR]=4.0×109/L to 11.0×109/L); platelet count varied from 3.74×109/L to 64.00×109/L (RR=150×109/L to 450×109/L) and hemoglobin levels were between 6.0g/dL and 8.4g/dL (RR=12g/dL to 14g/dL). The reticulocyte count was 3.7% (RR=0.5% to 1.5%) with the absolute red cell count of 2.35×103/μL. Abdominal ultrasound revealed liver at 4cm below the lower edge of the right costal margin and the spleen was classified as Boyd grade III.
Several tests were carried out to investigate the cause of the pancytopenia, including leishmaniasis, HIV, hepatitis B and C, syphilis and antinuclear antibody test, all with negative results. Tests for vitamin B12, folate, iron serum levels and other routine tests were also performed, with results within normal reference ranges. A bone marrow aspirate and biopsy showed increased cellularity, abnormal distribution of erythropoiesis, atypical megakaryocytes, immature granulocytes and evidence of reactive marrow fibrosis. Marrow iron stores were normal. The metrorrhagia was controlled with the administration of combined oral contraceptives.
Throughout 11 years, a non-progressive pancytopenia was the only persistent sign of a hematological disorder. A significant hepatosplenomegaly was noticed and confirmed by imaging in 2002. The liver and spleen were no longer palpable in the patient's first consultation in our outpatients’ clinic in 2009. An abdominal CT scan, performed in 2009, revealed mild splenomegaly with no other abdominal abnormalities. Petechiae were present in her consultation, mainly in the oral cavity. Platelet counts were consistently below 40.0×109/μL and absolute neutrophil counts varied from 1.5×109/L to 2.0×109/L, with relative lymphocytosis (58% to 65%). A discrete normocytic normochromic anemia could be observed in most of the blood tests, with hemoglobin varying from 10.5g/dL to 12g/dL. In addition, the patient presented increased lactate dehydrogenase (LDH) levels, with values ranging from 902U/L to 1020U/L (RR: 313–618U/L). Hepatic enzymes and calcium levels were unremarkable. A high dose of prednisone (60mg per day) was prescribed to increase platelet counts but was stopped as it was not effective.
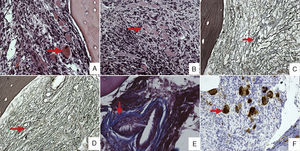
The patient was tested and proved to be negative for: (i) the V617F mutation of the Janus kinase 2 (JAK2) gene, (ii) W515L and W515K mutations of the MPL gene and (iii) the BCR-ABL fusion gene. Furthermore, sequencing of the coding region of exon 12 of JAK2 and exon 9 of the calreticulin (CALR) gene was performed by capillary electrophoresis but no mutations were found. She had a normal karyotype by G banding analysis. The antinuclear antibody test was positive for anti-cytoplasm in 1:360–1:640 dilution (RR: <1:80) and β2-microglobulin was slightly increased (3.47μg/mL; RR: <2.4μg/mL). Bone marrow aspiration and biopsy were repeated in 2013 (Figure 1). The biopsy sample was stained using hematoxylin and eosin, periodic acid-Schiff (PAS), giemsa, silver impregnation (Gordon-Sweet), Masson's trichrome, and Perls. Immunostaining was performed for CD2, CD3, CD4, CD5, CD8, CD20, CD30, CD34, CD56 and FOXP3. The lymphocytes reacted with variable intensity for CD2, CD3, CD4, CD5 and CD8 but were negative for CD56 and FOXP3. The reaction with CD4 was more intense than with CD8. CD34 positive blasts were not identified. The reaction with CD61 highlighted the intense proliferation of atypical/small and medium sized megakaryocytes, with hypolobulated nuclei and rare naked nuclei (Figure 1A, B and F). The remaining parenchyma showed intense subversion of architecture due to severe grade 3 fibrosis (reticulin and collagen) and due to the neovascular formation and osteosclerosis (Figure 1C–E). There was scanty representation of the myeloid and erythrocytic series. At the present, the patient is taking 2mg clonazepam, 10mg Baclofen, and 5mg prednisone daily.
Bone marrow biopsy. (A and B) Bone marrow biopsy stained using hematoxylin and eosin showing clusters of abnormal megakaryocytes (arrow). (C and D) Bone marrow biopsy stained using Gordon Sweet stain showing a marked increase in coarse reticulin fibers (arrow). (E) Perivascular collagen fibrosis stained using Masson's trichrome (arrow). (F) Immunostaining for CD61 with megakaryocyte hyperplasia (arrow).
HTLV-1 infection has long been described in association with hematological and auto-immune disorders.1,2 In this case, ATL was ruled out due to the absence of typical ATL ‘flower cells’ characterized by intermediate to large pleomorphic cells, sometimes with hyperlobulated nuclei CD4+/CD8− and the absence of bone marrow infiltration. Of the myeloproliferative neoplasms (MPN), chronic myeloid leukemia and polycythemia vera were also ruled out due to the absence of the BCR-ABL fusion gene and the JAK2 V617F mutation, respectively, and lack of remaining clinical and laboratory criteria used to diagnose both conditions, including the absence of the W515L and W515K mutations of the MPL gene.
The presence of atypical megakaryocytes, bone marrow fibrosis, increased LDH, and pancytopenia suggested the diagnosis of myelofibrosis. Myelofibrosis, characterized by chronic and persistent pancytopenia associated with bone marrow fibrosis and proliferation of megakaryocytes, is one of the most common types of MPN. It can be primary or develop as an end-stage bone marrow failure, secondary to other MPN. Myelofibrosis can also be present as a reaction of the bone marrow against other neoplasms or inflammatory processes, and can emerge as an autoimmune phenomenon.6,7 Classical findings of primary myelofibrosis are hepatosplenomegaly, teardrop erythrocytes and leukoerythroblastosis in the peripheral blood, which were not present in this patient. Moreover, the benign, non-progressive course of the disease disfavored the diagnosis of primary myelofibrosis as the median survival of patients with this condition is about 69 months.8 The clinical picture and laboratory tests suggest the diagnosis of autoimmune myelofibrosis (AIMF).
AIMF is an under-recognized cause of marrow fibrosis, and has been defined using the following criteria: (1) Grade 3 or 4 reticulin fibrosis of the bone marrow; (2) lack of clustered or atypical megakaryocytes; (3) lack of myeloid or erythroid dysplasia, eosinophilia, or basophilia; (4) lymphocyte infiltration of the bone marrow; (5) lack of osteosclerosis; (6) absent or mild splenomegaly; (7) presence of autoantibodies; and (8) absence of a disorder known to cause myelofibrosis.6 A recent study reported that mild atypias including occasional clustered, left-shifted, small, and hypolobated megakaryocytes, can be observed in AIMF.7
AIMF is classified as primary or secondary, the latter named when associated with autoimmune disorders. Diseases such as systemic lupus erythematous (SLE), scleroderma and Sjögren's syndrome are the main disorders described in association with secondary AIMF.6,7 AIMF has also been described in association with HIV infection9 but not with HTLV infection. The differentiation between AIMF and the MPN-related myelofibrosis is imperative once these disorders have different management and prognoses. AIMF tends to respond well to steroids and/or other immunosuppressive agents and has a better prognosis.
An additional unusual aspect of this case is the rare overlap of HTLV-1-related diseases: HAM/TSP and a hematological disorder. ATL and HAM/TSP seem to differ in their route of transmission (ATL being mainly via breast-feeding, and HAM/TSP via blood transfusion), pathogenesis and immunological response. After infection, both cellular and humoral immune responses are formed against HTLV. The humoral reaction contributes as delayed protection by producing antibodies against viral proteins, including Tax. However, recently, Tax-antibodies have been associated with the development of HAM/TSP, suggesting an autoimmune component to the disease. This antibody could also be associated with AIMF. Furthermore, HAM/TSP apparently relates to patients with high proviral loads, which is supposedly determined by host factors such as polymorphisms in the major histocompatibility complex class I (MHC-I) molecules and its influence on antigen presentation to CD8+ T-cells.
In conclusion, this is the case of a young woman with HTLV-1 infection and HAM/TSP, who evolved with pancytopenia and bone marrow fibrosis. To our knowledge, except for a short report by Engels,10 who does not provide detailed information, this is the first report of AIMF associated with HTLV-1 infection. This case report strengthens the described association between HTLV-1 and autoimmune/hematological disorders.
Conflicts of interestThe authors declare no conflicts of interest.
The authors wish to thank Laboratorio Fleury, São Paulo, Brazil, in particular Dr. Maria Carolina Tostes Pintão, for performing some of the molecular tests for the diagnosis of myeloproliferative neoplasms.