Angioimmunoblastic T-cell lymphoma (AITL) is a malignancy of mature T-cells. It is characterized as a polymorphic lymphonodal lymphoid infiltrate accompanied by prominent proliferation of endothelial venules and follicular dendritic cells. AITL was first described in 1974 by Frizzera et al. as an angioimmunoblastic lymphadenopathy with dysproteinemia.1 A short time later, the name was changed to immunoblastic lymphadenopathy, and then to lymphogranulomatosis X in 1979.2
AITL comprises 15–20% of all peripheral T-cell lymphomas and 1–2% of all non-Hodgkin lymphomas (NHL). Most frequently, it occurs in aged patients, with equal prevalence between males and females. Typically, AITL displays an aggressive behavior, which makes the diagnosis difficult and it must be differentiated from other malignant lymphoproliferative diseases, drug reactions and viral infections. Patients with AITL frequently exhibit B-symptoms (e.g., fever and weight loss) and a generalized enlargement of the lymph nodes. Other common symptoms include hepatomegaly, splenomegaly, polymorphic skin rash and pleural effusion. Advanced stage disease (Ann Arbor III/IV) is observed in 80% of cases. AITL is also associated with autoimmune phenomena. Polyclonal hypergammaglobulinemia occurs in approximately 50% of AITL cases.3
Histological analyses of AITL specimens show effacement of the lymph node architecture, particularly in advanced stages. Malignant cells tend to distribute into interfollicular regions, and are typically positive for T-helper cell markers and T-cell receptors (TCRs) alpha and beta.2,4 Immunoblasts, often positive for Epstein–Barr virus (EBV), are frequently dispersed in paracortical regions. This characteristic can be confused with Reed-Sternberg cells, which can lead to a mistaken diagnosis of Hodgkin's lymphoma (HL). TCR gene rearrangements are found in 70% of cases. On the other hand, immunoglobulin gene rearrangements are found in only 10% of patients with AITL.5
There is no standard treatment for AITL. Consequently, patients may be treated with different drugs, including steroids, immunomodulators or by cytotoxic chemotherapy. However, the most commonly used treatment modality is the cyclophosphamide, vincristine, doxorubicin and prednisone (CHOP) regimen, associated or not with etoposide. This treatment is typically followed by autologous hematopoietic stem cell transplantation. Furthermore, the natural history of AITL is characterized by several relapses, with a five-year overall survival of 30%.6,7
In this case report, we analyzed the main clinical characteristics that make AITL diagnosis difficult. As AITL is a rare disease with a poor prognosis, an early and correct diagnosis is essential to improve survival and quality of life.
Case reportA 56-year-old man with generalized lymphadenomegaly (neck, abdomen, inguinal, supraclavicular and axillar regions) came to the Instituto do Câncer do Estado de São Paulo for treatment. The disease was first noticed three months previously. The initial lymph node biopsy, performed in another hospital, suggested nodular sclerosis HL. It was described as a lymphoid infiltrate in a background of eosinophils, prominent vessels and large cells, suggestive of Reed-Sternberg cells (CD45+, CD3− CD20−, CD30+, CD15+, and EBV negative).
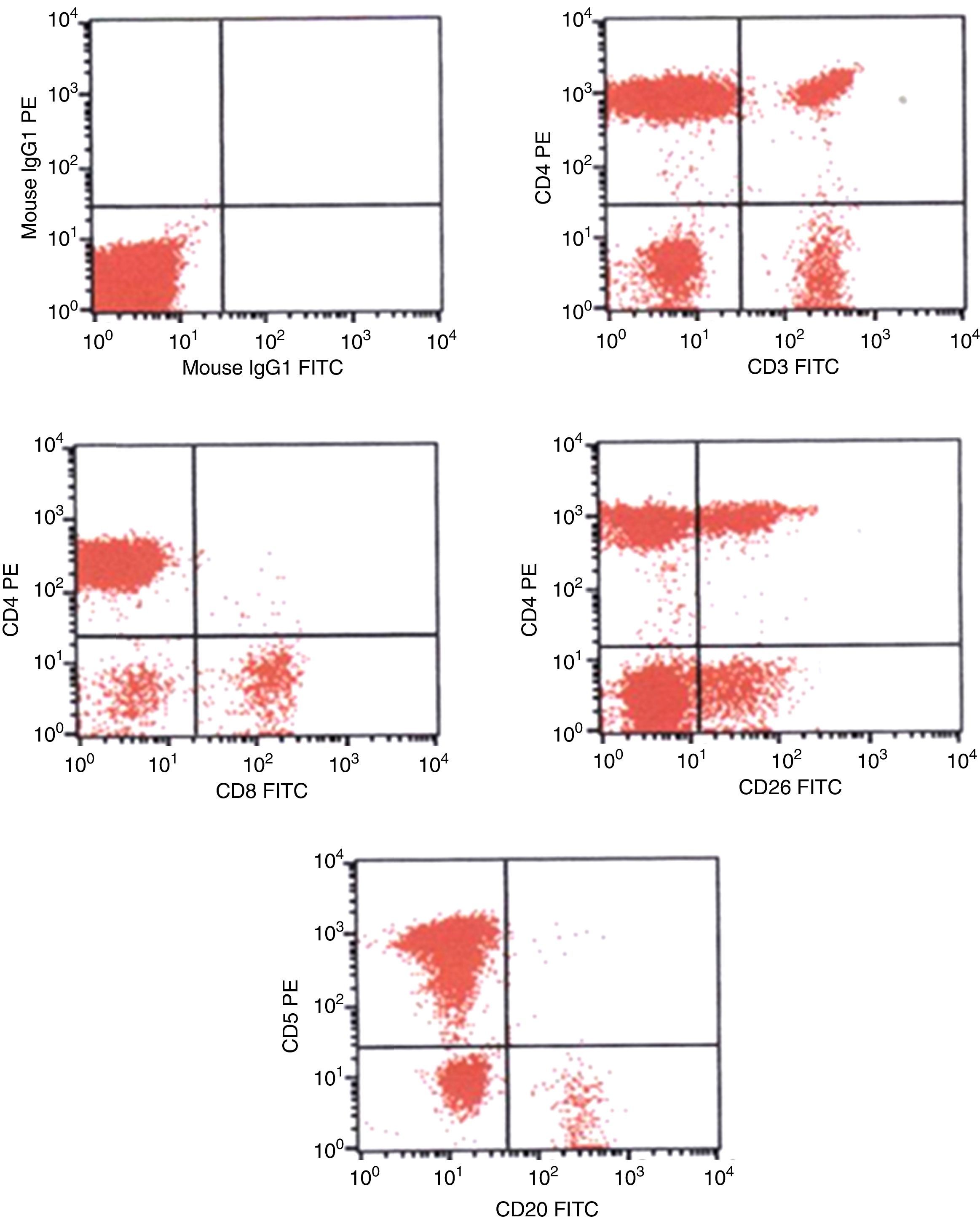
At admission, the complete blood count showed 10.2g/L hemoglobin, 12.4×109/L white blood cell count with 5.4×109/L lymphocytes and 270×109/L platelets. Leukocyte immunophenotyping by flow cytometry showed 11% of mCD3− CD5+, CD4bright, CD8− and CD26partial lymphoid T-cells (Figure 1). Furthermore, a bone marrow biopsy demonstrated absence of lymphoma infiltration.
The patient was submitted to a fluorine-18 fluorodeoxyglucose positron emission tomography scan that showed increased uptake of FDG located in superficial and deep lymph node chains. The exam also indicated a reversal of the metabolic pattern between liver and spleen.
A review of the histological specimens from the previous biopsy at our center revealed atypical lymphoid cells with expansion in the paracortical zone and a moderate number of eosinophils. However, we did not repeat the immunohistochemical analysis in this sample because it was very small. Therefore, another biopsy was indicated, and a core biopsy was chosen because it could be done faster. The histological analysis of the new material was compatible with reactive lymphadenopathy.
In the next few days, the patient's condition worsened. In the meantime, another biopsy was performed, this time a surgical lymph node biopsy. However, the immunochemistry analysis was incomplete when the patient's condition became aggravated. As the revision of the initial biopsy was compatible with HL, and due to the quick deterioration in the patient's performance status, our team opted to start treatment for HL with the doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) regimen.
After two ABVD cycles the patient developed severe pancytopenia. The treatment was interrupted due to toxicity. As it was not the expected clinical response, the diagnosis of HL was once more questioned. At that time, a new bone marrow biopsy was made, and the histology showed that the bone marrow tissue was totally substituted by fibrosis. Simultaneously, the results of a polymerase chain reaction (PCR) evaluation of the monoclonal rearrangement of the TCR-gamma gene became available, and they were positive: these new data suggested a mature T-cell lymphoproliferative disease.
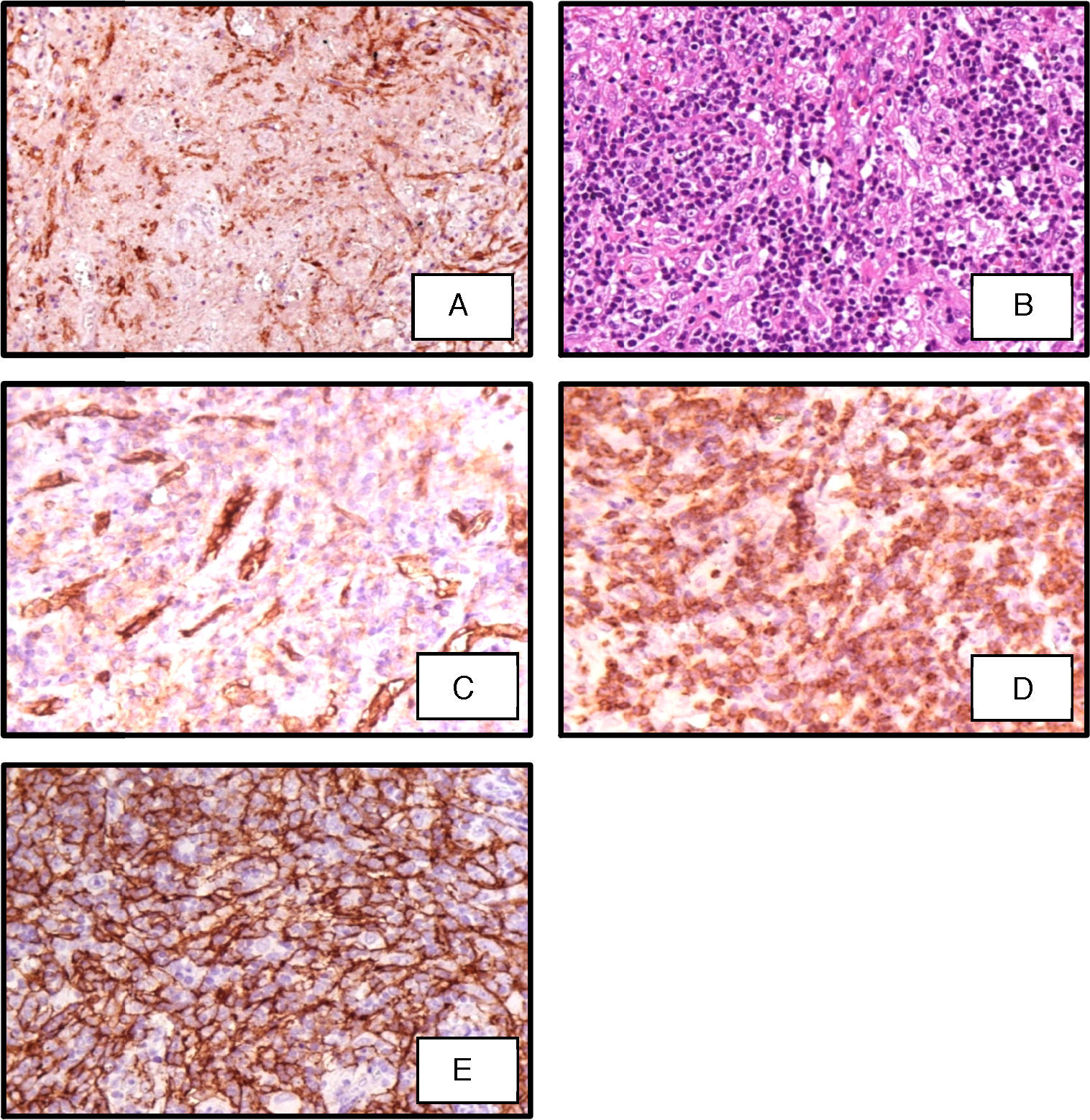
A few days later, the immunohistochemistry of the excisional lymph node biopsy was completed and the diagnosis of AITL was confirmed (Figure 2). In the meantime, the patient's clinical condition deteriorated drastically, so another appropriate chemotherapeutic scheme was not prescribed. Unfortunately, the patient died in a short period due to respiratory complications.
Section of lymph node biopsy showing (A) immunoperoxidase (IP) CD2 showing lymphoid cells CD2+; (B) polymorphic infiltrate including eosinophils and small lymphocytes (hematoxylin–eosin 400×); (C) IP stain showing lymphoid cells CD10+; (D) IP stain for CD21 showing dendritic follicular cells; (E) IP stain for CD31 showing a high quantity of vessels.
AITL is an aggressive malignancy. It is the second most common peripheral T-cell lymphoma in Western countries. Although many patients achieve complete remission, disease relapse is frequent, and the three-year overall survival is less than 50%. The clinical symptoms of AITL are not specific and, consequently, the diagnosis can be complex and sometimes delayed. The clinical features of AITL are more related to immune dysfunction than to tumor growth. For example, AITL symptoms include skin rash, fever, generalized lymphadenopathy and polyclonal hypergammaglobulinemia, which are common features of infections and autoimmune disorders.3
The gold standard for AITL diagnosis is excisional lymph node biopsy. However, to save time and to avoid exposure to invasive procedures, many centers perform a core biopsy to obtain samples for pathological analyses. Unfortunately, the samples obtained by core biopsy can be insufficient to perform a complete immunohistochemical panel and ensure the correct diagnosis.
Previous studies have shown that the accuracy of a lymphoma diagnosis based on core biopsies varies from 68% to 94%.8 Inadequate core biopsies are associated with misdiagnoses or delayed diagnoses. Gupta et al. compared lymph node biopsies acquired with either fine-needle aspiration or surgical excision in 100 patients. They found that the rates of accurate diagnoses based on fine-needle aspirations were 77% in reactive hyperplasia, 75% in NHL, and 85% in metastatic carcinoma. A recent meta-analysis showed that core biopsies provided adequate material for histology in 95% of cases, particularly in salivary gland lesions, but inadequate material in 39 (2.6%) cases of lymphadenopathies of the head and neck. In differentiating between malignant lymphoma and reactive lymph nodes, core biopsies showed a high false-negative rate and a low negative predictive value (85%).9
A full histological and immunohistochemical analysis of the lymph node is essential for the differentiation between AITL and other diseases. For example, it can differentiate between large cells with two or more nuclei, which are frequently observed in AITL tumor microenvironments, and Reed-Sternberg cells.3,4 In the present case study, the small amount of tissue was insufficient to perform an expanded immunohistochemical panel, and consequently, the diagnosis was incorrect.
Singh et al. demonstrated that 17/17 patients with AITL in the leukemic phase harbored a distinct population of sCD3−/CD4+ T-cells in the peripheral blood. Furthermore, this phenotype was highly specific to AITL because it was found in only 1/40 patients with other T-cell lymphomas in the leukemic phase. They showed that this phenotype provided a positive predictive value of 94% for a diagnosis of AITL. Those authors concluded that immunophenotyping peripheral blood with flow cytometry might be a useful method for achieving a differential diagnosis of AITL, even though the aberrant T-cell population occurs at a very low frequency in peripheral blood.10 Therefore, this assay should be part of lymphoma investigations, especially in inconclusive cases, because it can save time and improve the accuracy of diagnosis.
In this case report, we describe a case of AITL that was erroneously treated as HL. We state that this mistaken diagnosis was mainly a consequence of an insufficient biopsy sample, which led to an incomplete histological analysis. We recommend that in refractory cases a complete revision of the initial biopsy should be performed as soon as possible. We also strongly recommend that to obtain an accurate and precise diagnosis for lymphoma, excisional biopsies should be performed instead of core biopsies. Ancillary studies, such as peripheral blood immunophenotyping and PCR detection of TCR rearrangements, are very important tools to establish differential diagnoses, and should be part of the investigation to improve the accuracy or to confirm the diagnosis of AITL.
Conflicts of interestThe authors declare no conflicts of interest.