Infection is a major complication in patients with chronic lymphocytic leukemia (CLL). Newly diagnosed patients are at high risk of developing infection caused by encapsulated bacteria, such as Streptococcus pneumoniae and Haemophylus influenzae.
Method and ResultsHowever, once treatment is initiated, the spectrum of pathogens causing infection broadens, depending on the treatment regimens. With disease progression, cumulative immunosuppression occurs as a consequence of multiple treatment lines and the risk of infection further increases. On the other hand, the use of targeted therapies in the treatment of CLL have brought new risks of infection, with an increased incidence of invasive fungal diseases, particularly aspergillosis, in patients receiving Bruton kinase inhibitors.
ConclusionIn this article, we review the epidemiology of infection in patients with CLL, taking into account the treatment regimen, and briefly discuss the management of infection.
Infection is a major complication in patients with hematologic malignancies.1 The increased risk of infection is a consequence of severe immunosuppression caused by the disease itself and its treatment. The effects of cancer chemotherapy on the immune system include bone marrow suppression resulting in neutropenia, mucosal barrier disruption and suppressive effects on T-cells and B-cells.1 On the other hand, the treatment of hematologic malignancies has changed substantially over the past 15 years, with the introduction of novel therapies that, unlike conventional chemotherapy, have specific targets in neoplastic cells, potentially reducing the deleterious effects of the treatment on host immune defenses. However, infections may occur, either by a predicted effect of these agents on the immune system, or by an unanticipated immunosuppression. Therefore, the introduction of these novel therapies represents new challenges for the proper management of patients with hematologic malignancies.
Chronic lymphocytic leukemia (CLL) is the most common leukemia in adults in developed countries, with an age-adjusted incidence of 4 to 5 cases per 100,000 of the population.2 Infection is a major complication in patients with CLL and a frequent cause of death.3 Changes in the treatment of CLL over the past decade, including the use of targeted therapies,4 have expanded the list of pathogens causing infection. The knowledge of the potential effects of these new drugs on the immune system and the risk of infection associated with these agents is crucial in order to implement appropriate strategies for the prevention and treatment of infection. In this paper, we will review the infectious complications in patients with CLL, with a special emphasis on infections with the use of the new agents.
Immunodeficiency in newly diagnosed CLL patientsNewly diagnosed patients with CLL have inherent immune defects that predispose them to infection, including hypogammaglobulinemia, defects in T-cells, suppressor natural killer cells, dendritic cells, neutrophils and complement system.5 Hypogammaglobulinemia is the main immune defect associated with an increased risk of severe infection, with a 5-fold increase in the risk if IgG serum levels are below 600 mg/dL.6 However, a clear-cut correlation between serum immunoglobulin levels and infection is not always present and patients with normal immunoglobulin serum levels may have recurrent infections.7 Serum levels of immunoglobulins rarely return to normal values with treatment, although one study reported an increase in IgA serum levels in patients successfully treated with ibrutinib.8
In addition to hypogammaglobulinemia, other parameters that are obtained in newly diagnosed patients to assess the prognosis of CLL may help to define the risk of infection, as patients with unmutated immunoglobulin heavy chain variable region (IGHV) or with mutations of the TP53 gene may have a shorter time to the first infection.9
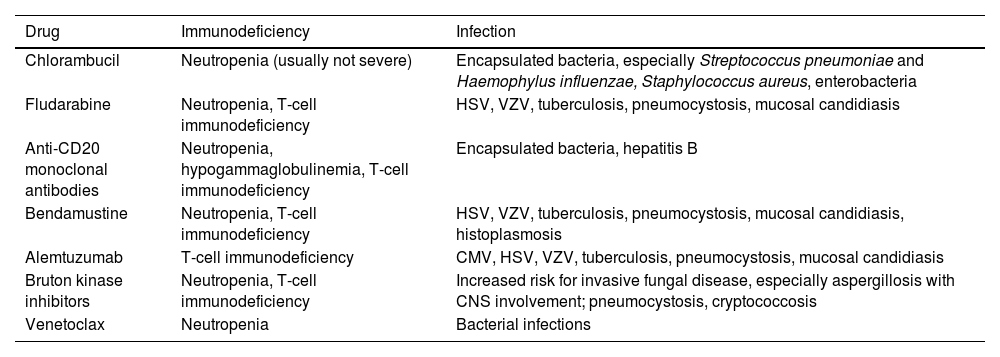
Infections in newly diagnosed patients and in patients receiving chlorambucilAs shown in Table 1, bacteria are responsible for the majority of infections in newly diagnosed CLL patients and in those treated with alkylating agents. Of the encapsulated bacteria (Streptococcus pneumoniae, Haemophylus influenzae and Neisseria meningitidis), Staphylococcus aureus and Enterobacteriaceae are the most frequent agents.5 Common sites of infection are the skin and respiratory and urinary tracts. Sinus and pulmonary infections appear to be more frequent in patients with decreased IgA serum levels.10 Infections caused by other pathogens, such as the Herpes Simplex virus (HSV), occur only in the setting of relapsed/refractory (R/R) disease. On the other hand, Pneumocystis jirovecii pneumonia is uncommon in patients treated with chlorambucil.5
Drugs and regimens used in the treatment of chronic lymphocytic leukemia and the risk of infection.
HSV = herpes simplex virus; VZV = varicella-zoster virus; CMV = cytomegalovirus; CNS = central nervous system.
Infections are more frequent in relapsed / refractory cases, compared to those of first-line therapy.
The introduction of fludarabine into the therapeutic armamentarium of CLL brought new challenges for the management of infectious complications. Fludarabine causes a marked reduction in T-cell subpopulations, especially CD4+ cells, which decrease sharply after three cycles of chemotherapy, persisting for over 2 years after the discontinuation of the drug.11 As a result, infections caused by opportunistic pathogens associated with severe T-cell immunodeficiency have emerged, including Listeria, mycobacteria, P. jirovecii, Candida, Aspergillus and herpes viruses (cytomegalovirus [CMV], HSV and varicella-zoster virus [VZV]).12 The risk of infection correlates with CD4 counts13 and increases with the concomitant use of corticosteroids, advanced stage disease, previous treatment for CLL, renal failure, neutropenia and absence of response to fludarabine.7,11,12
Compared to alkylating agents, the use of fludarabine as first-line therapy is associated with a higher incidence of infection.14 By contrast, in a randomized trial, the addition of cyclophosphamide to fludarabine did not increase the risk of infection, compared to fludarabine alone.15
Infection in patients treated with rituximab-containing regimensRituximab was the first anti-CD20 monoclonal antibody on the market and has been used in the treatment of B-cell malignancies and, more recently, in autoimmune diseases. It promotes the lysis of B lymphocytes through the antibody-dependent cytotoxicity, antibody-dependent cellular phagocytosis and complement-dependent cytotoxicity.16 Patients receiving rituximab are at increased risk for infection, as shown in two meta-analyses of randomized trials of rituximab given as maintenance therapy, versus no maintenance in patients with lymphoma.17,18 In a randomized study comparing fludarabine plus cyclophosphamide with fludarabine, cyclophosphamide and rituximab (FCR), neutropenia was more frequent in patients receiving FCR (34% vs. 21%, p < 0.001), but the rates of infection were similar (25% vs. 21%, p = 0.18).19 Because both progression-free-survival and overall survival were significantly improved in FCR recipients, this regimen became the standard of care as the primary therapy for younger and fit CLL patients.20
Hypogammaglobulinemia is frequent in patients receiving rituximab. A study evaluated serum levels of immunoglobulins before and after rituximab use in 179 patients with non-Hodgkin lymphoma.21 Hypogammaglobulinemia was observed in 69 patients (38.5%). In the majority of the cases (77%), serum levels of IgG dropped to values between 400 and 600 mg/dL, but only 14% of the 69 patients had “symptomatic” hypogammaglobulinemia, defined as ≥ 2 episodes of infection in a 6-month period. The large majority of patients with symptomatic hypogammaglobulinemia presented with low serum levels of > 1 isotype (IgG, IgM or IgA). Sinopulmonary infections were the most common presentation and the median time between rituximab use and infection was 1 year. Intravenous immunoglobulin was given only to patients with recurrent infections.
Another complication of the use of rituximab is late-onset neutropenia, defined as grades III and IV neutropenia (according to the National Cancer Institute Common Toxicity Criteria). This complication has been reported in 3 to 27% of the patients, develops between 1 and 6 months after the end of therapy and lasts for 1 to 11 weeks. The bone marrow examination shows myeloid maturation arrest. The mechanism of late-onset neutropenia is not clear, but may involve imbalances in T-cell lymphocyte subpopulations, B-cell depletion, and a decreased level of cytokines. The risk of late-onset neutropenia is higher in patients receiving multiple courses of rituximab (as in maintenance therapy) and in those with previous exposure to chemotherapy or in advanced disease stages, as well as in patients who had received hematopoietic cell transplantation (HCT).22
An inhibitory effect of anti-CD20 monoclonal antibodies on the function of regulatory B-cells results in the T-cell immunodeficiency,23 with an increased risk of infections, such as enterovirus encephalitis, babesiosis, infection caused by Parvovirus B19, CMV reactivation and P. jirovecii pneumonia.24 A meta-analysis of 11 studies showed that the use of rituximab in the treatment of lymphoma was associated with an increased risk for pneumonia due to P. jirovecii (3.65 risk ratio), but with a low incidence overall (2.97%).25
Another infection associated with the use of rituximab is progressive multifocal leukoencephalopathy (PML). In a series of 57 cases published in 2009, PML developed after a median of 16 months from the first dose of rituximab. The clinical presentation included confusion/disorientation (54%), focal signs (33%), poor motor coordination (25%), speech changes (21%) and visual changes (18%). The case-fatality rate was 90%.26
Hepatitis B virus (HBV) reactivation may occur in patients receiving chemotherapy and the risk increases with the use of rituximab. Because HBV reactivation may occasionally evolve to liver failure, screening before starting chemotherapy is mandatory.27 Patients positive for HBsAg should receive antiviral prophylaxis with lamivudine, entecavir or tenofovir.27 Patients who test negative for the HBsAg, but have either the anti-HBc or anti-HBs, should undergo serial HBV DNA measurements and, if the HBV DNA is detected in the serum, preemptive therapy should be started. Patients on anti-HBV prophylaxis or preemptive therapy should be monitored with monthly HBV DNA serum levels. Discontinuation of anti-HBV drugs is safe one year after the end of chemotherapy, provided that no HNV DNA is detected during this period. Once the prophylaxis / preemptive therapy is discontinued, it is recommended that monthly HBV DNA measurements be continued for an additional six-month period.27
New anti-CD20 agents, obinutuzumab and ofatumumabTwo new anti-CD20 monoclonal antibodies have been approved in the treatment of CLL: ofatumumab 28 and obinutuzumab.29 Their action on CD20 B cells is somehow different, compared to rituximab, but the spectrum of infection with these agents is expected to be similar to those reported with rituximab. A randomized trial testing chlorambucil with or without obinutuzumab as first-line therapy in CLL patients with comorbidities showed higher rates of neutropenia (35% vs. 16%), but not infection (11% vs. 14%), in patients receiving obinutuzumab.29 In another study on patients with follicular lymphoma, the addition of obinutuzumab to bendamustine increased the rates of upper respiratory tract infections and neutropenia, compared to patients receiving bendamustine alone.30 Similar to obinutuzumab, studies with ofatumumab on untreated CLL patients showed higher rates of neutropenia (26% vs. 14% in chlorambucil recipients), but not infection (26% vs, 14%).31 Cases of fatal reactivation of HBV32 and PML33 have been reported. In summary, the spectrum of infectious complications associated with the use of obinutuzumab and ofatumumab appears to be similar to that which has been reported with rituximab.
AlemtuzumabAlemtuzumab is a monoclonal antibody directed against CD52, approved in 2001 for the treatment of patients with CLL previously treated with alkylating agents. This drug induces profound depletion of both B and T-cells (especially CD4+), which predisposes the patient to infections in a pattern similar to that of patients with advanced HIV infection. Opportunistic infections include herpes simplex, herpes zoster, mucosal candidiasis, cryptococcosis, pneumocystosis and mycobacteriosis, among others.34–36 In addition, CMV reactivation is frequent, requiring monitoring with CMV DNA detection in the blood, plus antiviral preemptive therapy.37
Bruton kinase inhibitorsIbrutinib was the first Bruton tyrosine kinase (BTK) inhibitor approved for the treatment of CLL, Waldenström's macroglobulinemia, mantle cell lymphoma and graft versus host disease. A randomized trial comparing ibrutinib with chlorambucil in untreated CLL patients reported similar rates of neutropenia (16% vs. 23%) and pneumonia (4% vs. 2%).38 In a phase 3 study comparing ibrutinib with ofatumumab as a secondary line in R/R CLL patients, the overall rates of infection were 70% in ibrutinib recipients versus 54% in patients receiving ofatumumab. Moreover, neutropenia, pneumonia and sinusitis were more common in the ibrutinib arm, but the rates of grades III to IV infection were similar (24% vs. 22%).39 In a five-year follow-up of patients with CLL receiving ibrutinib (R/R or as first-line therapy), the rates of infection were higher in the R/R setting: neutropenia (21% vs. 3%), febrile neutropenia (5% vs. 0%), pneumonia (27% vs. 10%), cellulitis (9% vs. 0%), sepsis (8% vs. 3%) and sinusitis (5% vs. 0%).40
Ibrutinib and invasive fungal diseaseVarughese et al. evaluated the frequency and characteristics of serious infection (requiring hospitalization) among 378 patients with lymphoproliferative diseases (44% CLL) receiving ibrutinib at daily doses of 420 to 560 mg.41 The median treatment duration was 413 days, 84% received ibrutinib as monotherapy and 81% as rescue therapy after a median of 2 lines of treatment. Forty-three patients (11.3%) developed serious infection, with 6 deaths. The median time from ibrutinib initiation and infection was 4 months; 84% of the episodes occurred in the first year of treatment. There were 23 cases of bacterial infections (53.5%), with Staphylococcus aureus being the most frequent pathogen; 4 patients (9.3%) had a viral infection, and; 16 (37.2%) had invasive fungal disease (IFD). Of note, invasive aspergillosis was diagnosed in eight of the 16 episodes of IFD, 2 of which had central nervous system (CNS) involvement. The factors associated with serious infection were neutropenia and the patient having had ≥ 3 lines of therapy. The risk factors for IFD were receipt of corticosteroids and ≥ 3 treatment lines before treatment with ibrutinib.41
Just after the first report of the association between ibrutinib and IFD, Ghez et al. reviewed 33 cases of IFD in patients receiving ibrutinib at 16 centers in France. Thirty patients had CLL and only one was receiving ibrutinib as first-line therapy. Invasive aspergillosis was diagnosed in 27 of the 33 patients (82%), 11 of which (40.7%) had CNS involvement. Cryptococcosis was diagnosed in four patients and pneumocystosis and mucormycosis in one patient each. The median time from the initiation of ibrutinib treatment and the development of IFD was 3 months and 85% developed IDF within 6 months from the initiation. All of the patients had some sort of immunosuppression associated with prior therapies, including corticosteroid use and neutropenia.42 In another study, among 18 patients receiving high doses of ibrutinib for the treatment of CNS lymphoma (5 as first-line therapy), seven (39%) developed invasive aspergillosis, with two cases with CNS involvement (29%).43
Various questions emerge from these observations of the association between ibrutinib and invasive aspergillosis and the CNS involvement. Ibrutinib has effects on the immune system outside the Bruton kinase that may predispose to IFD, including the impairment of the function of alveolar macrophages, diminished Th1 → Th17 response, poor recognition of fungal antigens by Dectin-1 and reduction in the recruitment of neutrophils to the site of infection.44 However, it seems that the Bruton kinase itself is important to prevent the development of invasive aspergillosis, as shown in an animal model that studied the outcome of invasive aspergillosis in the Bruton kinase gene knockout (Btk -/-) and wild-type (Btk +/+) mice. In the Btk -/- mice, lung damage was more pronounced and the fungal load and mortality rate were higher.43
More recently, second-generation Bruton kinase inhibitors were approved for the treatment of CLL and other lymphoproliferative diseases. In a randomized trial, acalabrutinib was compared with ibrutinib in the treatment of R/R CLL patients. The overall rate of grade III-IV infection was 30.8% in acalabrutinib recipients and 30% in patients receiving ibrutinib. Ten of 268 patients receiving acalabrutinib (3.8%) developed IFD (5 cases each of invasive aspergillosis and pneumocystis pneumonia) compared with 5 of 265 (1.9%) receiving ibrutinib (including 2 cases of invasive aspergillosis).45 In another study in naïve CLL patients, acalabrutinib was compared with acalabrutinib plus obinutuzumab, or chlorambucil plus obinutuzumab. Infection rates were 20.8% in patients receiving acalabrutinib plus obinutuzumab, 14% in recipients of acalabrutinib alone and 8.3% in patients receiving chlorambucil plus obinutuzumab. Two cases of invasive aspergillosis were diagnosed in patients receiving acalabrutinib alone.46 A pooled safety analysis of zanubrutinib monotherapy in the treatment of lymphoid malignancies reported 27% of grade ≥ 3 infections and 3% of opportunistic infections (23 patients), including 7 cases of cryptococcosis and 5 of invasive aspergillosis.47
IdelalisibIdelalisib is a phosfatidilinositol-3-kinase delta inhibitor approved for treatment of R/R CLL and R/R patients with follicular lymphoma. A study comparing rituximab with idelalisib or placebo in R/R CLL reported few episodes of infection and no difference concerning neutropenia, pneumonia or febrile neutropenia in the two arms.48 In a phase 3 randomized trial, R/R CLL patients were randomly assigned to receive bendamustine plus rituximab with idelalisib or placebo. The rate of infection was higher in patients receiving idelalisib, including opportunistic infections, such as P. jirovecii and CMV.49
VenetoclaxVenetoclax is a Bcl-2 inhibitor. Except for neutropenia, the use of venetoclax is not associated with significant immune dysfunction. In a randomized trial that compared venetoclax plus rituximab with bendamustine plus rituximab, both arms had the same frequency of febrile neutropenia and pneumonia, but patients receiving venetoclax had more neutropenia.50
Covid-19 in patients with CLLThe pandemic of infection by SARS-CoV-2 in 2020 brought new challenges for the management of infection in hematologic patients, including CLL. The first publications showed higher death rates in patients with hematologic malignancies compared with the general population, as shown in a study that reported a mortality 2x higher overall and 3.7x higher in individuals over 70 years old.51 A population-based registry study evaluated 697 patients with hematologic malignancies and Covid-19. Of the 479 patients with lymphoproliferative diseases, the highest mortality was observed in patients with CLL (36%). Active treatment with monoclonal antibodies was significantly associated with higher mortality by multivariate analysis.52
The higher death rate in patients with CLL and Covid-19 may be explained by the fact that these patients are usually severely immunosuppressed because of past and current therapies (cumulative immunosuppression), including fludarabine, bendamustine, Bruton kinase inhibitors and anti-CD20 monoclonal antibodies. In addition, CLL is more likely to occur in elderly patients, with frequent comorbidities, such as diabetes, cardiac diseases and others. Indeed, the presence of comorbidities seems to be the main driver for the poor outcome of Covid-19 in CLL patients observed in the first waves of the pandemic, as seen in a study evaluating 594 CLL patients with Covid-19. Factors associated with death were older age and cardiac failure, while no treatment in the previous 12 months was protective.53
The introduction of Covid-19 vaccines using different platforms resulted in a marked reduction in the frequency of severe disease, reflected in a reduction in hospitalizations and deaths. However, response to vaccines in immunosuppressive patients has been poor. A meta-analysis of 64 studies involving 8546 adults with hematologic malignancies showed a serologic response of 44% only among patients with CLL and 16% in patients with prior treatment with anti-CD20 monoclonal antibodies.54 In another study, a risk score was built to predict response to the SARS-CoV-2 vaccine in patients with CLL with the following variables: no anti-CD20 treatment in the preceding year (30 points) and treatment naïve, normal IgG, IgM, IgA or hemoglobin (10 points each). The score predicted a response of < 5% in patients with < 20 points, 36% in patients with 20 to 70 points and 75% in those with over 70 points.55
The consequence of poor response to the vaccine is the occurrence of breakthrough infections. In a study, among 287 patients with CLL who had been fully vaccinated (mostly 2 doses of RNA-based vaccine), the rate of breakthrough Covid-19 infection was 15.2%. In the entire population of patients with hematologic malignancies (5956), the overall hospitalization risk was 37.8%, with a 5.7% death rate.56 In another study of 113 patients with hematologic malignancies who had received at least one dose of the vaccine (78% with 2 doses) and developed Covid-19, 66% required hospitalization, 14% needed intensive care and 12% died.57
At the end of the year 2021, a new SARS-CoV-2 variant (Omicron) became prevalent worldwide. The variant showed higher transmission rates, but was a less severe disease in the general population. In a study in CLL patients, the rates of hospitalization and death declined sharply after the emergence and predominance of the Omicron variant.58
Management of infection in patients with CLLThe first step in the management of infection in patients with CLL is to estimate the net state of immunosuppression, taking into account the status of the disease, prior and current therapies, co-morbidities and history of infections that may recur, such as tuberculosis, herpes simplex and herpes zoster. Newly diagnosed patients should have immunoglobulin serum levels verified to estimate the severity of hypogammaglobulinemia. The optimal time for vaccinating patients with CLL is before treatment, including patients in whom a “watch and wait” strategy is applied. Therefore, before starting treatment, patients should receive the recombinant zoster vaccine (2 doses), the conjugate pneumococcal vaccine followed by the polysaccharide pneumococcal vaccine, meningococcal vaccine, Haemophylus influenzae vaccine, yearly seasonal influenza vaccine and, more recently, the SARS-Cov-2 vaccine.
The use of prophylactic antimicrobial agents in patients receiving treatment should be individualized, taking into account the regimen and the treatment line. In the first-line therapy, the only situation in which acyclovir and sulfamethoxazole-trimethoprim are recommended is when fludarabine is part of the treatment. For other first-line therapeutic regimens, the decision should be individualized. By contrast, in the majority of patients receiving treatment for R/ R disease, more severe T-cell immunodeficiency is present because of the cumulative immunosuppression and anti-herpes and anti-pneumocystis prophylaxis are usually indicated.59
As commented, patients receiving Bruton kinase inhibitors are at an increased risk of developing invasive fungal disease, particularly aspergillosis. However, the incidence (about 2%) is not high enough to justify primary prophylaxis. Instead, serum galactomannan and chest computed tomography should be part of the workup for infection in patients receiving Bruton kinase inhibitors, especially if given in second or later treatment lines.
The use of prophylactic immunoglobulin in CLL patients with hypogammaglobulinemia is controversial. A meta-analysis of randomized clinical trials conducted in patients with hematologic malignancies with hypogammaglobulinemia showed that immunoglobulin replacement reduced the risk of clinically documented infections, but did not prevent microbiologically documented infections or infectious-related deaths.60 Our approach to the management of hematologic patients with hypogammaglobulinemia is to give prophylactic antibiotics (amoxicillin is a good option) to patients who develop a first episode of infection. If a new episode of infection occurs in patients receiving a prophylactic antibiotic, we start a program of immunoglobulin replacement.61