Sickle cell intrahepatic cholestasis (SCIC) is an uncommon but severe complication in sickle cell disease (SCD) patients homozygous for hemoglobin (Hb) S or with Hb S/β thalassemia.1–3 It is clinically characterized by marked conjugated hyperbilirubinemia, right upper quadrant pain, enlarged liver and moderately elevated hepatic enzymes. In more severe cases, coagulopathy and renal dysfunction may be observed and the condition occasionally progresses to liver failure. SCIC is most commonly described in its acute or recurrent forms but it eventually becomes chronic. The pathogenesis of SCIC involves sickling within the hepatic sinusoids leading to vascular stasis, hypoxia and ballooning of the hepatocytes, resulting in intracanalicular cholesthasis.1,4 Treatment is based on exchange blood transfusions (EBT) and aims to reduce Hb S blood levels and consequently the sickling process. Although several case reports show reversal of SCIC with prompt EBT,1,3,5 we describe a case of a non-responsive patient who perished despite treatment.
Case reportThe patient was a 38-year-old Afro-Brazilian woman with SCD (Hb SS) and a medical history of complications of her disease: acute painful vaso-occlusive crises, bone infarcts, skin ulcer of the lower limb, acute chest syndrome and autosplenectomy. She had no history of alcohol abuse and had a normal body mass index. She complained of jaundice, choluria, dyspnea on light exertion and pain in the right upper abdominal quadrant and in the lower limbs that started four months prior to hospital admission.
The physical examination at hospital admission revealed a patient in distress, presenting a blood pressure of 140×90mm Hg, heart rate of 81 beats per minute, respiratory rate of 18 breaths per minute, temperature of 36.7°C, and oxygen saturation of 96%. Respiratory sounds were normal and a painful enlarged liver was identified.
Laboratory tests showed anemia (Hb: 6.7g/dL; hematocrit: 21%) and cholestasis (total bilirubin: 16.01mg/dL; direct bilirubin: 12.11mg/dL) with a slight elevation of liver enzymes [aspartate aminotransferase (AST): 138U/L; alanine aminotransferase (ALT): 46U/L; alkaline phosphatase (ALP) 445U/L; gamma-glutamyl transferase (GGT) 437mg/dL]. Albumin levels were at the lower normal limit (3.4g/dL) and the international normalized ratio (INR) was at the upper normal limit (1.37). Serology tests for hepatitis B and C, HIV and autoimmune conditions were negative. She had positive immunoglobulin (Ig)G antibodies for cytomegalovirus and Epstein–Barr virus. Alpha-fetoprotein and ceruloplasmin were within the normal ranges and iron metabolism markers were slightly altered (iron: 174μg/dL; transferrin saturation: 68%; total iron binding capacity: 256μg/dL; ferritin: 575ng/dL). In addition, renal function was preserved (creatinine: 0.53mg/dL), blood cultures were negative and chest X-ray revealed no change of the pulmonary parenchyma. The Hb S fraction one month before had been 74%.
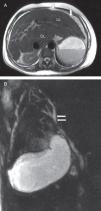
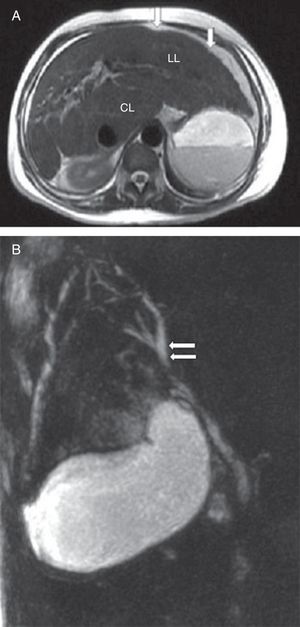
Magnetic resonance imaging (MRI) of the abdomen showed an enlarged liver with slightly irregular contours and heterogeneous parenchyma suggestive of chronic liver disease, associated with small volume ascites (Figure 1A). Gallbladder and choledocus duct were typical at magnetic resonance cholangiopancreatography (MRCP). There was a slight focal dilatation of the biliary tree in the right hepatic lobe, but no biliary obstruction was detected (Figure 1B).
(A) Axial T2 weighted magnetic resonance imaging of the abdomen showing liver enlargement with increased left (LL) and caudate (CL) lobes, associated to slightly irregular contours (arrows). (B) Magnetic resonance cholangiopancreatography showing normal gallbladder and slight focal dilatation of the biliary tree (arrows) in the right hepatic lobe without biliary obstruction.
The patient was given intravenous fluids, analgesia, folate and supplementary oxygen therapy. She received 900mL of packed red blood cells in order to increase the hematocrit to 25%, followed by EBT. Transfusions led to clinical improvement and the Hb S fraction dropped to 14.3–20.4%. Despite resolution of respiratory distress and lower limb pain, she persisted with jaundice, choluria and a slight abdominal pain.
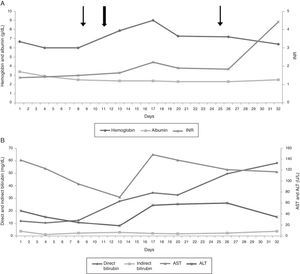
On the 13th day of hospitalization, she presented with acute respiratory distress, associated with fever and hypoxemia. A chest X-ray revealed diffuse lung opacities and the diagnosis of acute chest syndrome was reached. The patient was transferred to the intensive care unit (ICU), placed on mechanical ventilation and meropenem therapy was initiated. She presented mental confusion and an isolated seizure. Her clinical condition and computed tomography (CT) findings suggested posterior reversible encephalopathy syndrome (PRES). Normal levels of ammonia discarded the hypothesis of hepatic encephalopathy. She gradually evolved with increasing levels of direct bilirubin and elevated INR, despite maintenance of Hb S below 25%. Figure 2 shows the evolution of albumin, hemoglobin, INR, bilirubin, AST and ALT during hospitalization. Renal function progressively deteriorated over her last six days of life, reaching a creatinine level of 3.31mg/dL on the day of her death. Blood and airway secretion cultures collected in the ICU were negative. On the 32nd day of hospitalization, she evolved with refractory shock resulting in death.
(A) Evolution of albumin (g/dL), hemoglobin (g/dL) and international normalized ratio (INR) during hospitalization. (B) Evolution of direct and indirect bilirubin levels (mg/dL), aspartate aminotransferase (AST - U/L) and alanine aminotransferase (ALT - U/L) during hospitalization. Narrow arrows refer to red blood cell transfusions and the wide arrow indicates exchange blood transfusion (EBT).
Diagnosis of SCIC is based on clinical and laboratory evidence of non-obstructive cholestasis, moderately elevated hepatic enzymes and an enlarged and painful liver. The patient described in this report showed all the characteristic features of SCIC, including marked conjugated hyperbilirubinemia, right upper quadrant pain, enlarged liver, and a coagulation disorder, but only moderately elevated liver enzymes. According to Ahn et al., SCIC can be subdivided as mild (average direct bilirubin 27.6mg/dL) and severe diseases (average direct bilirubin 76.8mg/dL or presence of change in mental status or coagulation disorder), with the death rates being 4% and 64%, respectively.5 Our patient fit the criteria of severe disease (direct bilirubin 58.17mg/dL and coagulation disorder). Other hepatic diseases, such as viral hepatitis, Wilson's disease and liver cancer were excluded. Due to technical limitations, MRI quantification of hepatic iron concentration was not performed, but hemochromatosis was unlikely. Despite history of complications of her disease, previous multiple transfusions were not reported and iron metabolism markers were just a little changed. The ferritin level, although high (575ng/dL), has low specificity and can be high in both liver and inflammatory diseases. Furthermore, the patient did not report using any hepatotoxic substance. Liver biopsy was not carried out, since this procedure involves high morbidity and mortality, particularly in the presence of an acute hepatic lesion.4,6,7
Regarding treatment, there are no randomized clinical trials with sufficient evidence to prove efficacy of therapeutic measures.8 However, the prognosis of SCIC improved significantly with the advent of EBT. Ahn et al. showed that seven out of nine patients with severe SCIC who received EBT survived, while 12 of 13 patients not undergoing EBT died.5 Hb S fraction levels below 20–30% were established as ideal, but again there is lack of evidence, with only pragmatism in relation to these values.1–5 There is evidence that ursodeoxycholic acid protects against the cytotoxicity and apoptosis induced by hydrophobic bile acids. It is a therapeutic tool in primary biliary cirrhosis, sclerosing cholangitis, intrahepatic cholestasis of pregnancy, cystic fibrosis, progressive familial intrahepatic cholestasis and chronic graft-versus-host disease.9 Some authors claim that it could improve biliary flow in SCIC,4 but its use has not yet been established in this condition.8
In this case, EBT did not alter the course of the disease, so that direct bilirubin levels continued to rise and the coagulation disorder and albumin levels continued to worsen until death. Even with an adequate Hb S percentage (13.4% and 20.4% in different measures), there was deterioration in the clinical course of SCIC. Starting treatment with the disease already in an advanced stage may have contributed to the bad outcome in this case. The lack of response could also be due to the concomitance of another undiagnosed hepatic disease. Although blood and airway secretion cultures were negative, the patient probably developed sepsis, which could have contributed to cholestasis, liver dysfunction and death.7 Additionally, acute chest syndrome clearly influenced clinical deterioration and fatal outcome.
Berry et al. described 38 patients with sickle cell hepatopathy, varying from self-limited cholestasis to acute liver failure and cirrhosis.7 The authors reported 55% mortality among patients with acute presentations of hepatic disease, requiring emergency admission. All four cirrhotic patients admitted with acute hepatic decompensation died. The patient described herein had no clinical signs of cirrhosis or portal hypertension. Laboratory tests revealed albumin and INR to be within the limits of normality, while MRI showed signs of chronic liver disease. Nevertheless, we decided not to perform a biopsy to discard such an condition, as this procedure in her severe acute condition is correlated to high mortality and complications such as bleeding.4,6,7 Costa et al. reported a case of SCIC unresponsive to EBT in which the 48-year-old patient had cirrhosis without major clinical changes as identified by liver biopsy but had a history of hepatitis C virus infection with the viral load being undetectable for years before the cholestasis. The main hypothesis about the unfavorable outcome was related to a less effective response to EBT in older patients and/or the presence of other liver conditions such as cirrhosis.2 Gardner et al. described a 27-year-old patient with SCIC who was successfully submitted to liver transplantation and the authors suggested that this procedure is an alternative treatment in special cases.4
Finally, it is known that the sickling process depends not only on the percentage of Hb S but also on inflammatory events, such as those related to infection, that often trigger Hb S polymerization. This results from the dynamic interaction between the cells and the vascular endothelium with subsequent tissue damage resulting from ischemia and oxidative stress. The subsequent inflammatory status with increased expression of endothelial adhesion molecules and cytokines could trigger sickling in the microvasculature and perpetuation of SCIC.10
In conclusion, it is very important to recognize intrahepatic cholestasis, an uncommon but severe complication of sickle cell disease. Efforts to elucidate the physiopathological mechanisms involved in this process are needed. Although EBTs seem to be the most effective treatment for sickle cell intrahepatic cholestasis, studies are still necessary to define the best therapeutic approach and to identify factors associated to clinical outcomes.
Conflicts of interestThe authors declare no conflicts of interest.