Osteopetrosis (OP) variably referred to as ‘Marble bone disease’ or ‘Albers Schonberg disease’ was first described by a German radiologist in 1904. It is a group of genetically and clinically heterogeneous disorders characterized by increased skeletal density. Clinical severity varies from asymptomatic adults to a life-threatening condition in infants. Autosomal recessive osteopetrosis has an incidence of 1 in 250,000 births while the dominant form has an incidence of 1 in 20,000 births.1 We report on twin infants with osteopetrosis presenting a leukoerythroblastic picture and hepatosplenomegaly mimicking leukemia.
Case reportWe report on two male twin infants born of a consanguineous Muslim marriage. Case 1 was a nine-month male with complaints of recurrent chest infection and fever for four months. He had delayed developmental milestones (inability to hold neck, inability to sit) along with failure to gain weight. No history of bleeding, jaundice and neurological symptoms was present. He had received a one-unit packed red blood cell transfusion at the age of five months. On examination, he had pallor and severe wasting. Abdominal examination revealed moderate hepatosplenomegaly. Case 2 was the twin sibling of Case 1 who had similar complaints but for six months. On examination, he had pallor, bulging anterior fontanel along with moderate hepatosplenomegaly. The antenatal period was uneventful. There was no other similar family history with a healthy elder female sibling.
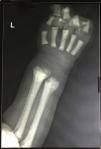
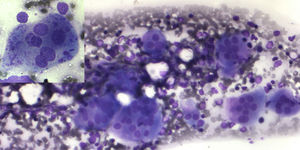
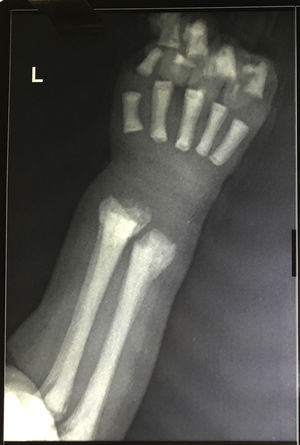
The peripheral blood film examination of Case 1 showed a leukoerythroblastic blood picture along with anemia (Hb: 7g/dL) and thrombocytopenia (80×109/L). Case 2 had similar findings with severe anemia (Hb: 6.2g/dL) and thrombocytopenia (38×109/L). In view of peripheral blood findings, differentials of bone marrow infiltration related to possible hematopoietic malignancy, metastatic disease, non-neoplastic storage disorder or extra medullary hematopoiesis (compensatory to marrow infiltration or stress) was carried out. The bone marrow examination was performed from medial tibial tuberosity with slight difficulty. The smears were cellular. Megakaryocytes were reduced. Mild erythroid hyperplasia (M:E ratio 1:1) was seen, however no abnormal increase in immature cells was found. Histiocytic cells were normal in morphology. The only significant finding was of increased number of osteoclasts in large clusters (Figure 1). Considering abnormal osteolytic activity, an X-ray evaluation was advised. X-ray of the forearm revealed increased bone density with characteristic bone in bone appearance of radius and ulna (Figure 2). On evaluating vitamin D, both had insufficient levels (15.9mg/dL and 21.5mg/dL, respectively). Other parameters of calcium metabolism, parathormone, phosphorus, alkaline phosphatase and calcium were within reference ranges. A head computed tomography scan was unremarkable with no evidence of hydrocephalous or obliteration of optic foramina. No features of renal tubular acidosis were present. Liver function and renal function were within reference ranges.
Both siblings were managed conservatively with antibiotics and transfusion support.
DiscussionOsteopetrosis is derived from the Greek words osteo ‘bone’ and petrosis ‘stone’. It is due to defective osteoclast function or differentiation. The Nosology and Classification of Genetic Skeletal Disorders 20062 categorizes OP into severe neonatal/infant form, intermediate form, and late onset form. Also categorized are variants associated with renal tubular acidosis (OP with RTA), with ectodermal dysplasia and immune defect (OLEDAID), with leukocyte adhesion deficiency syndrome (LAD-III).
In autosomal recessive osteopetrosis (AROP), the most common mutation (60%) is seen in the TCIRG1 gene affecting the proton pump function involved in acidification of resorption lacunae.3 Mutations affecting the chloride channel (15%) and carbonic anhydrase (<5%) are others on the list leading to AROP.
AROP classically manifests within the first year of life (frequently within three months). Visual impairment due to compression of optic nerve is usually the most common presenting complaint.4 The majority of cases present with bone marrow failure with extra medullary hematopoiesis (hepatosplenomegaly). Transfusion dependency prior to three months of age is a sign of severe disease with poor prognosis. Recurrent infections are commonly seen due to the defective generation of superoxide by peripheral blood leucocytes. Deafness due to auditory nerve compression is seen in approximately one-third of patients. Failure to thrive is commonly seen due to bone marrow failure and feeding problems. Other clinical features include skeletal abnormalities (short stature, macrocephaly, frontal bossing leading to hydrocephalous and choanal stenosis). Fractures due to paradoxical bone weakening are seen. Abnormalities of dentition and caries are common. Children with AROP are at risk to develop hypocalcaemia and tetanic seizures and secondary hyperparathyroidism. RTA with cerebral calcification is a feature of OP with RTA usually with a defect of the carbonic anhydrase enzyme.3
Our cases presented with failure to thrive, recurrent infections and bone marrow failure. Features of cranial nerve involvement, acidosis, and metabolic abnormality were not seen at the time of presentation.
Variable spectrum of osteoclast appearances has been reported in AROP.5 There are variable increases in number, size and nucleation of osteoclasts. Electron microscopy reveals defects in ruffled border-clear zone complex. Our case had increased number of osteoclasts with presence of clusters in bone marrow aspirate smears.
Bone marrow aspirate is usually difficult to obtain; we managed to prepare smears with difficulty. The trephine biopsy shows interweaving bone trabeculae with plates of hyaline cartilage with minimal medullary cavity and markedly reduced hematopoietic precursors.
The radiological examination is diagnostic showing increased bone density with diffuse or focal sclerosis of varying severity. Our cases revealed the characteristic bone in bone appearance.
Infants with severe OP frequently die during childhood with the most common cause of death being bone marrow failure and infections. A child of two years without transfusion dependency indicates favorable prognosis. An urgent bone marrow transplant is the only curative treatment so far. Patients receiving grafts from human leukocyte antigen (HLA)-identical siblings have a five-year disease free survival of 73–79%. For cases with unrelated or mismatched donors, the survival after transplant is 13–45%.6 Other alternative therapies such as calcitriol and interferon-γ have been tried with minimal success. Targeted gene therapy with genetically modified retroviral vector for the TCIRG1 gene mutation has been explored in mice models, with a survival of approximately 50%.7
The disease has an autosomal recessive inheritance pattern, implying a risk of phenotypic manifestation in one of four offspring. The identification of molecular defects in parents is useful for antenatal screening in subsequent pregnancies.
Although AROP is a rare disorder, it mimics a hematological malignancy. A careful evaluation of all components of bone marrow may provide clues to the diagnosis. A radiological survey must be considered in infants with unexplained anemia and hepatosplenomegaly.
Conflicts of interestThe authors declare no conflicts of interest.