Peripheral blood stem cell concentrations are traditionally adjusted to 20–40×106leukocytes/mL prior to freezing. This low cell concentration at cryopreservation implies larger volumes with more dimethyl sulfoxide being used, and higher cost and toxicity at the time of transplant. Higher cell concentrations have been reported but this is not widely accepted. Moreover, the influence of cell concentration on engraftment has not been well documented. Therefore, this study retrospectively analyzed the influence of peripheral blood stem cell concentration at freezing on engraftment after autologous hematopoietic stem cell transplantation.
MethodLeukapheresis products were plasma-depleted and cryopreserved with 5% dimethyl sulfoxide, 6% hydroxyethylamide solution and 4% albumin in a −80°C freezer. Individual patient data from hospital records were reviewed.
ResultsFifty consecutive patients with oncological diseases underwent 88 leukaphereses. Median age was six years (range: 1–32 years) and median weight was 19kg (range: 8–94kg). Median leukocyte concentration was 109×106/mL at collection and 359×106 (range: 58–676×106) at freezing with 78% viability (range: 53–95%); leukocyte recovery after thawing was 95% (range: 70–100%). In multivariate analysis, cell concentration (p-value=0.001) had a negative impact on engraftment. Patients infused with bags frozen with <200×106leukocytes/mL engrafted after a median of nine days (range: 8–12 days), 200–400×106leukocytes/mL after 11 days (range: 9–20 days); 400–600×106leukocytes/mL after 12 days (range: 8–19 days) and with cell concentrations >600×106leukocytes/mL, engraftment was after 14 days (range: 13–22 days).
ConclusionIn patients with adequate CD34 cell collections, total leukocyte concentrations of 282×106/mL, freezing with 5% dimethyl sulfoxide and 6% hydroxyethylamide solution without a controlled-rate freezer, and storing cells at −80°C yielded excellent engraftment. Further increases in cell concentration may delay engraftment, without affecting safety.
Cryopreservation of hematopoietic stem cells (HSC) has been used for many years to maintain their viability and proliferative potential for autologous hematopoietic stem cell transplants (HSCT). Bone marrow (BM) or peripheral blood stem cell (PBSC) concentration is usually adjusted before freezing in a cell suspension with 20–40×106leukocytes/mL.1
Dimethyl sulfoxide (DMSO) is an intra-cellular cryoprotectant that can maintain cell viability after thawing, but it is associated to many side effects.2 Four methods have been used to decrease the toxicity of the DMSO infusion: divide the cell infusion over consecutive days, to wash out the DMSO prior to transplant, to decrease the DMSO concentration at freezing,3 and finally, to concentrate the cells in order to have a smaller volume to be frozen and consequently, less DMSO to be infused.4–8 The Fred Hutchinson Cancer Research Center published their experience freezing cells at higher concentrations (median 370×106leukocytes/mL) over 20 years ago, with good viability and engraftment.1 Other centers have used two cryoprotectants, combining intracellular and extracellular activity [5% DMSO and 6% hydroxyethylamide solution (HES)], freezing cells without a controlled-rate freezer, and storing bags at −80°C until the time of transplant.6 For more than a decade, many children were transplanted at the Instituto de Oncologia Pediátrica-Grupo de Apoio ao Adolescente e a Criança com Câncer (GRAACC) of the Universidade Federal de São Paulo (Unifesp) using the latter method combined with high cell concentrations without a single graft failure, but the impact of using different cell concentrations on engraftment has not been adequately addressed.
ObjectiveThe objective of this paper is to retrospectively evaluate the impact of cell concentration at freezing on neutrophil engraftment.
MethodThis is a retrospective study of 50 consecutive patients with neoplastic diseases and complete laboratory data submitted to HSCT with autologous PBSC at the GRAACC. All parents or guardians signed the institutional consent form for leukapheresis and for the transplant. This study was approved by the Institutional Ethics Research Committee (CEP 047/08).
The leukapheresis products were completely plasma-depleted after centrifugation at 3000rpm (2616×g) for 15minutes at room temperature. The leukocytes remaining in the bag were not adjusted for any specific predetermined concentration. Patients with a very high CD34 cell count in the peripheral blood, who were expected to undergo only one leukapheresis procedure, had at least 30mL remaining in the bag, so PBSC were frozen in at least two canisters for safety reasons. The cells were cryopreserved in a solution of DMSO (Edwards Lifesciences Research Medical®, Irvine, CAN) and HES as previously described,6 with a final concentration of 5% DMSO, 6% HES and 4% human albumin. The bags were placed into metal canisters, transferred to an −80°C freezer and, up to 2002, stored in this freezer until used. After 2002, the bags were transferred to a nitrogen tank. As these products had already shown adequate viability, cell recovery and engraftment, a controlled-rate freezer was not installed in 2002 because it would increase costs.
Quality control before freezing included automated and differential leukocyte count (ACT Diff Beckman, Coulter Fullerton, CAN or micros 60 – CT Horiba® ABX, Montpellier, FRA), cell viability testing using 0.4% Trypan Blue dye exclusion (Gibco BRL, NY, USA) in a cell counter chamber (BOECO®, Neubauer, DEU), and CD34 cell counting, all performed before centrifugation. Sterility tests for aerobic bacteria and fungi used 3mL of the final supernatant (pediatric bottles – Bactec® – Becton, Dickinson and Company, County Clare, IRL), which was cultured for seven and 30 days, respectively. CD34 positive cells were counted in a Facs Calibur® flow cytometer (Becton Dickinson, San Jose, CA, USA) using anti-CD45-FITC and anti-CD34-PE (BD, Biosciences, San Jose, CA, EUA) monoclonal antibodies, according to the ISHAGE protocol.9 On the day of the transplant, cells were thawed in the laboratory in a 37°C water bath (Precision Scientific – Winchester, VA, USA) in a sterile plastic wrap (Sterifarma Produtos Cirúrgicos Ltda, São Paulo, Brazil).
Two thawing methods were used according to the attending physician's request, removing the DMSO or not. All patients who would receive a DMSO dose greater than 1g/kg or who were less than 20–25kg in weight, and those who had renal, respiratory or cardiac insufficiency, had the DMSO removed from the bag prior to the infusion using the previously validated New York Blood Center procedure to remove DMSO from cord blood products.10 The cells were rapidly infused though the central line without filters or infusion pumps. Patients were monitored (heart rate, respiratory rate, oxygen saturation) from the beginning of the infusions until two hours after the end of the last bag. All side effects were registered on a specific laboratory form, which was returned to the laboratory at the end of the infusion. Neutrophil engraftment was defined as the first of three consecutive days with a neutrophil count equal to or greater than 0.5×109/L.
The statistical analysis was designed to evaluate patient and product characteristics that could influence the number of days from infusion to engraftment. All patients had more than one bag frozen, and some underwent multiple apheresis procedures. The mean leukocyte count and cell concentration in the bags that were infused at the time of transplant were used in all calculations. The variables that were included in the analyses were underlying diagnosis, disease status (first remission vs. more advanced disease), CD34 cells (number of CD34/kg and concentration, i.e., number of CD34 cells/mL in the infused bags), leukocytes/mL (in the apheresis and in the product that was frozen and infused), percentage of leukocytes recovered after thawing (leukocyte count in the bag after thawing divided by the pre-freezing leukocyte count×100), volume reduction (original volume of the apheresis divided by the final volume of the buffy coat), total volume collected per patient (mL), number of leukaphereses needed to achieve the target of 5×106 CD34 cells/kg (one vs. more than one), freezer (mechanical vs. nitrogen), and DMSO removal or not. Using the 50 patients with complete information, the model was adjusted using all variables to select the significant ones, thereafter excluding the least significant variables, one by one. The variables CD34/kg, nucleated cells/mL (collected and infused) and volume reduction (%) were maintained in this step due to their importance to answer the study question. At each selection step, the variable with the highest p-value was eliminated, using a testing hypothesis in which the coefficient equaled zero. In the second step, a model including only significant variables was used to determine which influenced engraftment.
ResultsFifty patients with oncological diseases submitted to chemotherapy followed by autologous PBSC collection and transplantation were included in this study. Median age was six years (range: 1–32 years), with 24 being male. Median weight at the time of apheresis and at the time of HSCT was 19kg (range: 8–94kg). Underlying diagnoses were neuroblastoma (20), germ cell tumor (8), Ewing Sarcoma (5), lymphoma (5 Hodgkin's and 4 non-Hodgkin lymphoma), and other solid tumors (8). The most commonly used conditioning regimens were carboplatin, etoposide, melphalan, or busulfan and melphalan. Granulocyte-colony stimulating factor (G-CSF) was routinely used in only the first 21 transplants.
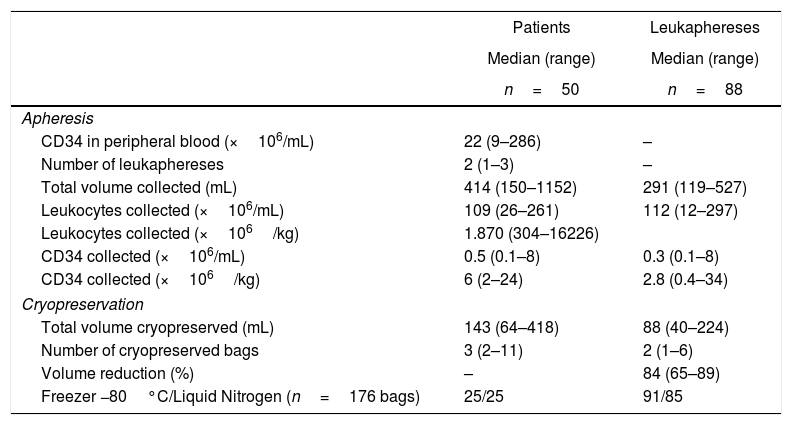
No patient had graft failure or died due to infection. The median time to neutrophil engraftment of 12 days was not influenced by prophylactic G-CSF administration (median engraftment 11 vs. 12 days). After a median follow-up of nine years (range: 5–15 years), overall survival was 51%. The median number of CD34 in the peripheral blood on the day of the first leukapheresis was 22×106cells/mL (range: 9–286×106cells/mL) (Table 1). The 50 patients underwent 88 leukaphereses, a median of two procedures per patient (range: 2–4).
Cell concentration at cryopreservation.
| Patients | Leukaphereses | |
|---|---|---|
| Median (range) | Median (range) | |
| n=50 | n=88 | |
| Apheresis | ||
| CD34 in peripheral blood (×106/mL) | 22 (9–286) | – |
| Number of leukaphereses | 2 (1–3) | – |
| Total volume collected (mL) | 414 (150–1152) | 291 (119–527) |
| Leukocytes collected (×106/mL) | 109 (26–261) | 112 (12–297) |
| Leukocytes collected (×106/kg) | 1.870 (304–16226) | |
| CD34 collected (×106/mL) | 0.5 (0.1–8) | 0.3 (0.1–8) |
| CD34 collected (×106/kg) | 6 (2–24) | 2.8 (0.4–34) |
| Cryopreservation | ||
| Total volume cryopreserved (mL) | 143 (64–418) | 88 (40–224) |
| Number of cryopreserved bags | 3 (2–11) | 2 (1–6) |
| Volume reduction (%) | – | 84 (65–89) |
| Freezer −80°C/Liquid Nitrogen (n=176 bags) | 25/25 | 91/85 |
The median number of leukocytes was 109×106/mL (range: 26–261×106/mL), with 0.5×106CD34/mL per collection bag, (range: 0.1–8×106/mL). The median viability at leukapheresis was 100%. Sterility test of all procedures was negative. To prepare the product for cryopreservation, the median volume was reduced by 84% (range: 65–89%). The median cell concentration at freezing, after adding the cryopreservation media and DMSO, was 359×106leukocytes/mL (range: 58–676×106/mL). The leukapheresis product was divided in a median of two bags (range: 1–6).
From each leukapheresis, the median cryopreserved volume was 88mL (range: 40–224mL). For the patients, the median cryopreserved volume was 143mL (range: 64–418mL) frozen in three bags (range: 2–11). The total number of bags for the 50 patients was 176 containing 10,814mL (buffy coat and cryopreservation media). The cells from 25 patients (91 bags) were stored in an −80°C freezer and from 25 subsequent patients (85 bags), in liquid nitrogen. The bags were stored for a median of 1.4 months (0.2–17 months).
A total of 165 bags were infused. The median number of cells infused was 1683×106leukocytes/kg (range: 209–8248×106/kg) and 6×106 CD34 cells/kg (range: 2–24×106/kg). The median viability was 78% (range: 53–95%) after thawing; leukocyte recovery was 95% (range: 70–100%). Patients who had infusions with no further manipulation had a total of 7×106CD34/kg (range: 3–15×106/kg) with 94% cell recovery (range: 73–100%) and a viability of 78% (53–95%), and neutrophil engraftment was observed after a median of 11 days (range: 8–16). DMSO was removed prior to infusion for 31 patients, who received 6×106CD34/kg (range: 2–25×106/kg) and 100% cell recovery (range: 70–100%), with a viability of 78% (range: 53–95%), and engraftment was documented after a median of 12 days (range: 8–22) (Table 2).
Characteristics of products with or without DMSO removal.
| Infusion without DMSO removal | Infusion with DMSO removal | |
|---|---|---|
| Median (min–max) | Median (min–max) | |
| n=19 | n=31 | |
| Age (years) | 13 (1–32) | 5 (1–25) |
| Weight (kg) | 39 (12–94) | 18 (8–91) |
| Leukocytes infused (×106/mL) | 406 (101–676) | 358 (58–666) |
| CD34 infused (×106/kg) | 7 (3–15) | 6 (2–24) |
| Total volume infused (mL) | 84 (40–184) | 160 (66–418) |
| Viability after thawing (%) | 78 (53–95) | 78 (53–95) |
| Cell recovery (%) | 94 (73–100) | 100 (70–100) |
| Neutrophil engraftment (days) | 11 (8–16) | 12 (8–22) |
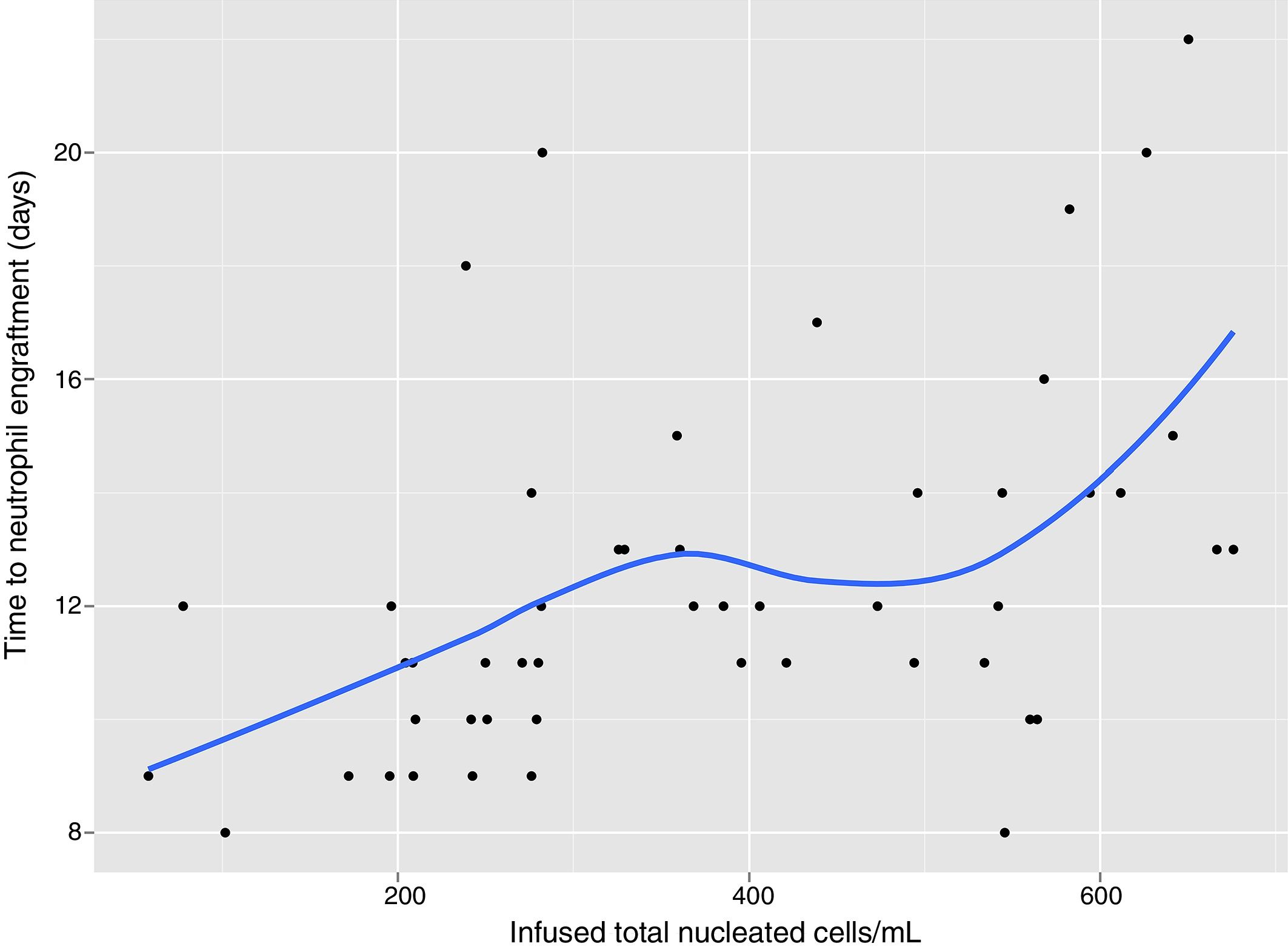
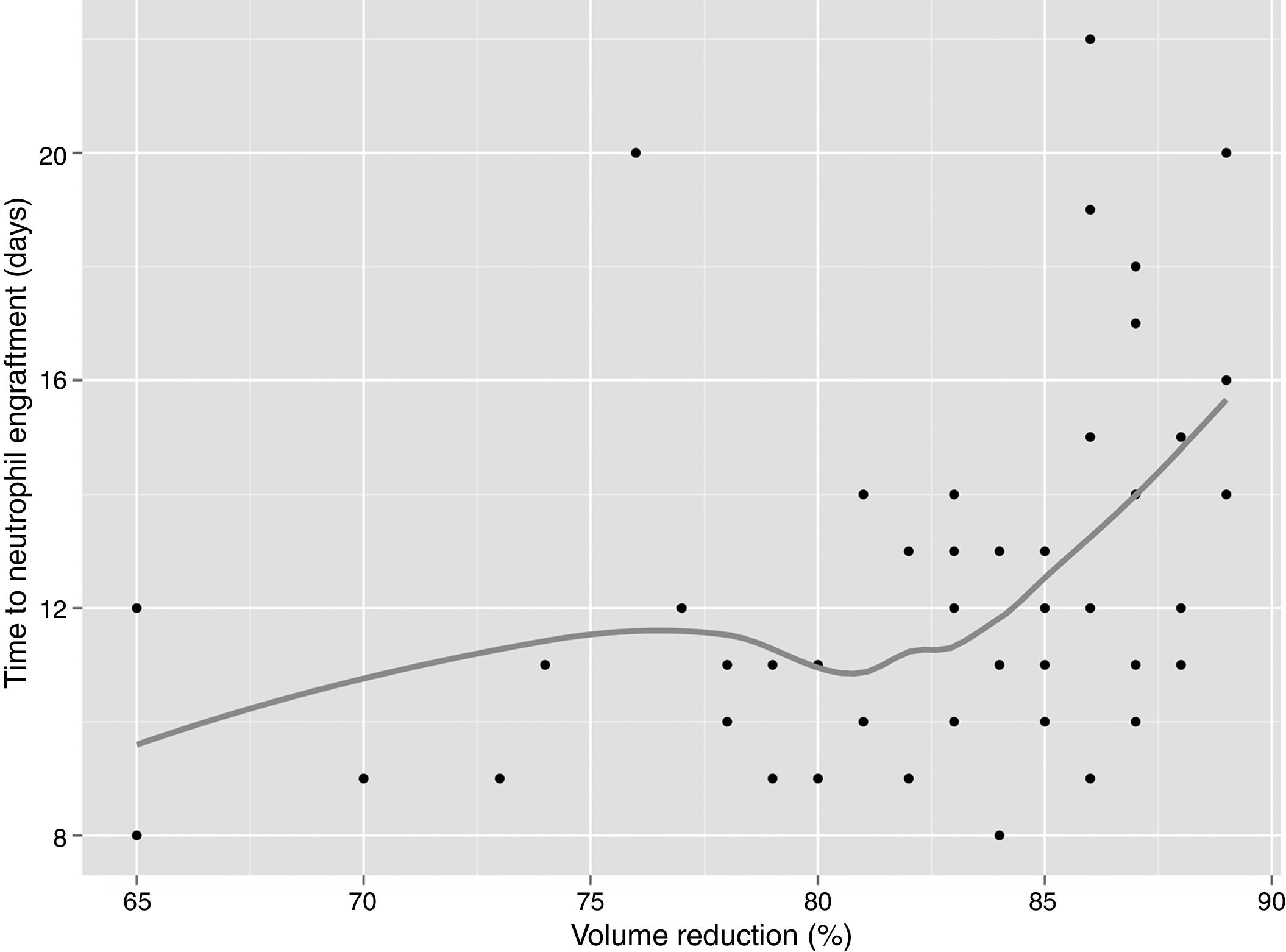
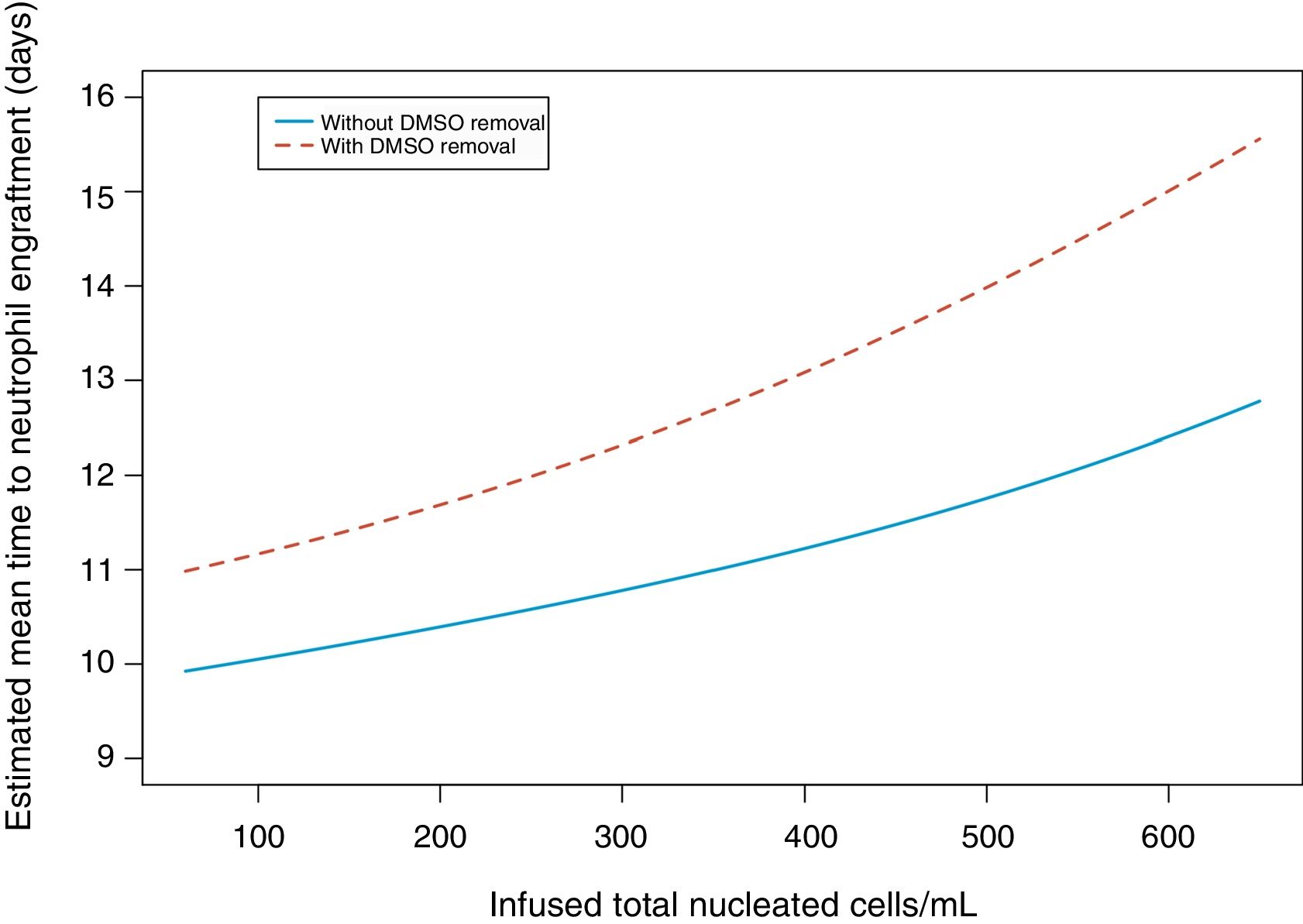
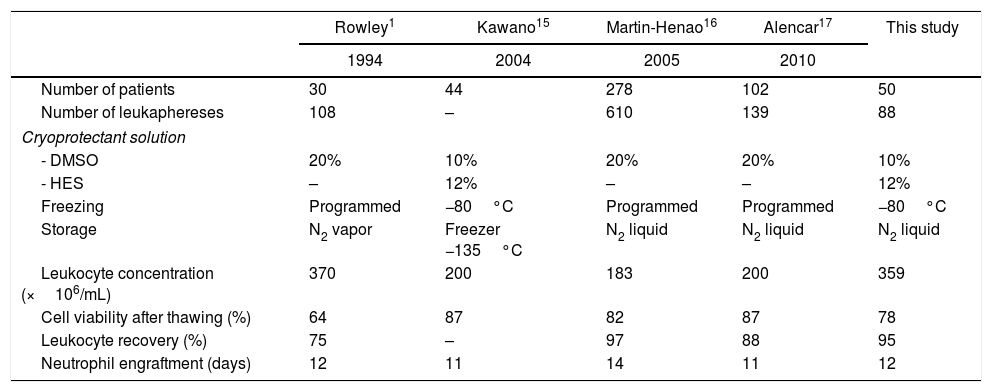
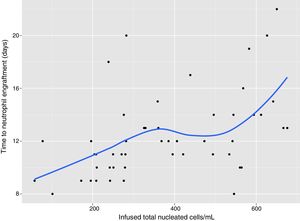
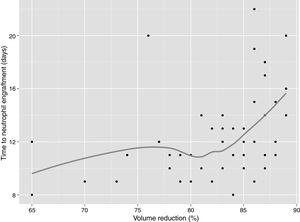
In univariate analysis, engraftment was not influenced (p-value >0.1) by underlying diagnosis, remission status, number of leukaphereses, total volume collected, number of leucocytes/mL at collection, type of freezer, CD34 infused/kg or cell recovery (%) after thawing. The variables with a negative impact on engraftment were number of leukocytes frozen/mL (RR: 0.99; 95% confidence interval: 0.995–0.990; p-value=0.001 – Figure 1), volume reduction (RR: 0.94; 95% confidence interval: 0.90–0.99; p-value=0.03 – Figure 2), and DMSO removal (RR: 0.42; 95% confidence interval: 0.22–0.80; p-value=0.009 – Figure 3).
The higher the cell concentration, the slower the engraftment, although there was not a single graft failure and the longest time to achieve 0.5×109neutrophils/L was 22 days. Patients infused with bags frozen with less than 200×106leukocytes/mL (n=6) engrafted after a median of nine days (range: 8–12 days); between 200 and 400×106 leukocytes/mL (n=23), engraftment was after 11 days (range: 9–20 days); between 400 and 600×106leukocytes/mL (n=15), the engraftment was after a median of 12 days (range: 8–19 days) and with cell concentrations above 600×106leukocytes/mL (n=6), it was after 14 days (range: 13–22 days).
DiscussionIn our experience, no patient had the cells frozen at the standard concentration of 20–40×106leukocytes/mL and all patients had engrafted in a median of 12 days (range: 8–22 days).
Because of working in a Pediatric HSCT Center, we chose to decrease the amount of DMSO infused using the techniques described by both Stiff 6 (5% DMSO, 6% HES) and Rowley1 (high cell concentration), although most HSCT centers still freeze leukapheresis products at 20–40×106leukocytes/mL, as originally described, due to concerns with cell viability and engraftment.1,6,8 The lower cell concentration, however, implies higher cost due to the higher volume cryopreserved, more supplies, more DMSO, need for more space in freezers, and a higher toxicity at the time of HSCT when the cells are infused.
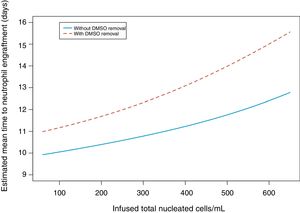
Since the first report by Rowley,1 other authors have published their experience using 5% DMSO and cell concentrations up to 250×106leukocytes/mL without a programmed freezing rate, storing the cells in −80°C freezers.11–13 Some authors have compared viability and engraftment using cell concentrations varying from 183–370×106leukocytes/mL and did not find any significant differences (Table 3). The published viability was 64–87%, cell recovery was 75–97% and neutrophil engraftment was in 11–14 days.8,14–17 Accordingly, the median leukocyte concentration in this study was 359×106/mL (range: 58–676×106/mL), viability 78%, cell recovery 95%, and engraftment in a median of 12 days.
Comparison between papers studying the impact of cell concentration on engraftment.
| Rowley1 | Kawano15 | Martin-Henao16 | Alencar17 | This study | |
|---|---|---|---|---|---|
| 1994 | 2004 | 2005 | 2010 | ||
| Number of patients | 30 | 44 | 278 | 102 | 50 |
| Number of leukaphereses | 108 | – | 610 | 139 | 88 |
| Cryoprotectant solution | |||||
| - DMSO | 20% | 10% | 20% | 20% | 10% |
| - HES | – | 12% | – | – | 12% |
| Freezing | Programmed | −80°C | Programmed | Programmed | −80°C |
| Storage | N2 vapor | Freezer −135°C | N2 liquid | N2 liquid | N2 liquid |
| Leukocyte concentration (×106/mL) | 370 | 200 | 183 | 200 | 359 |
| Cell viability after thawing (%) | 64 | 87 | 82 | 87 | 78 |
| Leukocyte recovery (%) | 75 | – | 97 | 88 | 95 |
| Neutrophil engraftment (days) | 12 | 11 | 14 | 11 | 12 |
In our patients, cell concentration at freezing above 282×106leukocytes/mL delayed neutrophil engraftment, although all patients engrafted and the longest time was 22 days. The median time to engraft increased from 9 days when bags were frozen with less than 200×106leukocytes/mL to 14 days with cell concentrations above 600×106leukocytes/mL. Keeping all other transplant variables constant, the number of leukocytes frozen/mL and volume reduction had statistically significant negative impacts on engraftment (p-value=0.001 and 0.03, respectively), although we can argue a biologically small effect. This may be due to the high cell numbers infused in these patients: only two received a cell dose of less than 2.5×106CD34/kg and half received more than 6×106CD34/kg. Therefore, when the CD34 cell number is adequate, the relevance of other factors such as cell concentration at freezing and volume reduction may emerge. An important limitation of this study is that we could not infer that this would hold true when lower CD34 cell numbers are infused, or when cells are frozen in a controlled-rate freezer and stored only in a nitrogen tank.
DMSO removal also had a significantly negative impact on engraftment (p-value=0.009). Without DMSO, median engraftment was after 11 days (range: 8–16 days) and with removal, 12 days (range: 8–22 days) and there was not any measurable cell loss (Table 2). Small children can easily have DMSO doses higher than 1g/kg in the cell products; this is associated with increased infusion-related toxicity. As our first 8kg patient had a cardiac arrest at the time of transplant, the procedure was not randomized and cells were not washed to remove the DMSO in all small children. High numbers of granulocytes in the apheresis can also cause infusion-related side effects,5,18–24 but this factor could not be analyzed in the current study.
ConclusionIn patients with adequate CD34 cell collections, total leukocyte concentrations of 200×106/mL, freezing with 5% DMSO and 6% HES without a controlled-rate freezer, and cell storage at −80°C yield excellent engraftment. Further increases in cell concentration may delay engraftment, without affecting safety. DMSO removal may delay engraftment by a median of one day, and its safety for autologous HSCT infusions is currently being studied in our institution.
Conflicts of interestThe authors declare no conflicts of interest.