Lymphopenia is a laboratory marker of poor prognosis and severity of disease in the SARS-CoV-2 infection. This study aims to describe the immune profile of a Brazilian population.
MethodsA total of 121 consecutive patients with severe acute respiratory syndrome (SARS) were analyzed between April and June 2020. Routine peripheral blood counts and multiparametric flow cytometry were performed on admission to assess lymphocytes and subsets (CD3, CD4, CD8). Demographic, clinical and laboratory data were collected from hospital sources.
ResultsThe total of 116 patients included 63 (54.3%) males; 76 (62.8%) COVID-19 patients were divided, based on clinical characteristics and mechanical ventilation (MV) use, into moderate (n = 41; no MV) and severe (n = 35; MV) groups. The control group (n = 40) was comprised of patients with SARS of different etiologies. All patients had lymphopenia, with overall lymphocyte counts and their subsets considerably lower in severe patients, when compared to the moderate and controls. Patients with a high neutrophil-to-lymphocyte ratio (> 15.2) and T-cell lymphopenia (CD3 < 593 cells/µL, CD4 < 326 cells/µL, CD8 < 121 cells/µL) had a higher risk of being intubated and progressing to death. A total of 39 patients (95.1%) in the moderate group and 54.3% (n = 19) in the severe group were discharged; 28 patients died.
ConclusionLaboratory assessment of the neutrophil/lymphocyte ratio and T-cell subsets may be predictive of mortality and may be useful for stratifying COVID-19 patients.
The disease caused by the SARS-CoV-2 (Severe Acute Respiratory Syndrome Coronavirus 2) can range from asymptomatic to mild, moderate or even severe symptoms. In fact, patients who present with the severe acute respiratory syndrome (SARS) frequently also present with high morbidity and mortality.1
In patients with the Coronavirus disease (COVID-19), the laboratorial routine was intensified due to the important changes observed in the cell counts, biochemical markers and coagulation function.2 Some biomarkers have been widely used with other clinical information to monitor the evolution of patients.3,4 In hospitalized patients, lymphopenia is considered a factor to define the disease severity and can be considered a marker for a poor prognosis.5,6
In fact, lymphopenia is one of the laboratory markers of the SARS-CoV-2 infection and has been considered a poor prognostic factor regarding the severity of the COVID-19 disease.7,8
Is widely known that parameters reported in the complete blood count (CBC) are associated with the clinical course of the COVID-19 patients.9 In addition to the accentuated reduction in the lymphocyte count, these patients have a relative neutrophilia,10 making the relationship between the neutrophil and lymphocyte counts
(neutrophil-to-lymphocyte ratio, NLR) a benchmark for comparison.11,12
Exploring the CBC more deeply, the evaluation of lymphocytes and their subpopulations (CD4+ and CD8+ T-cells, B-cells and NK cells) could provide information on disease severity and convalescence, when considered in conjunction with other clinical information.7,13–15 However, reports on lymphocyte subpopulations in a Brazilian population treated at a tertiary and highly complex hospital, paired with the clinical outcome, are still sparse in the literature.
Although there is a growing amount of knowledge on the immunological characteristics of COVID-19, we investigated the immune profile and blood counts upon the hospital admission of 121 individuals with SARS, comparing the T and B lymphocytes of patients with severe and moderate COVID-19 and patients with SARS from other causes, to clarify the immune response during the SARS-CoV-2 infection.
MethodsPatients and study designThis retrospective, observational, analytical and cross-sectional study recruited 121 consecutive patients with SARS suspected of COVID-19 admitted to the Hospital de Clínicas da Universidade Federal do Paraná (CHC-UFPR, Curitiba, Brazil) between April and June 2020. Five patients with human immunodeficiency virus and/or oncologic diseases were excluded.
Patients with confirmed SARS-CoV-2 infection by positive molecular (RT-PCR) or serological test (IgM) were included in the disease group (n = 76). The other SARS patients (negative for COVID-19) comprised the control group (n = 40).
Demographic, clinical and laboratory data available from hospital sources were collected. The presence of comorbidities, length of hospitalization and the need for the intensive care unit (ICU) and mechanical ventilation (MV) were assessed.
The CBC, ferritin, C-reactive protein (CRP), D-dimer, troponin and multiparametric flow cytometry (MFC) in peripheral blood were evaluated from day 1 through no later than day 3 of the hospitalization. This study was approved by the Human Research Ethics Committee of the CHC-UFPR (#31226720.7.0000.0096).
Subcategories of patientsThe ¨World Health Organization (WHO) Clinical Progression Scale¨ was used to classify the patients in this study.16 Based on this classification, the confirmed SARS-CoV-2 group was divided according to the use of supplemental oxygen (O2) therapy (MV or non-invasive ventilation, NIV) into moderate and severe groups. The moderate group received only supplemental O2 via nasal catheter and the severe group were those who required either invasive or NIV during hospitalization. Asymptomatic and mild patients were not included in this study because they did not require assistance from health services.
Multiparameter flow cytometryThe multiparametric flow cytometry (MFC) was performed in all patients at the beginning of their care (day one to day three). Absolute leukocyte counts were performed using the Sysmex XN-3000 counter at the time of the MFC analysis. The MFC was conducted using whole blood lysed with the BD FACS Lysing Solution (1:10). The Multitest® (BD Biosciences, San Jose, USA) was used to search for subsets of T-cells. This reagent is composed of the cell surface markers CD3 FITC (clone SK3/Leu3a), CD4 APC (clone 2D1), CD8 PE (clone SK7/Leu-4) and CD45 PerCP (clone SK1). In addition, the CD19 PE-Cy7 (clone SJ25C1, BD Biosciences) were used to identify B-cells. A total of 50,000 cell/events per tube were acquired using the FACSCanto II® cytometer (BD Biosciences) and FACS Diva software (BD Biosciences). The performance, compensation and daily MFI control were performed according to the manufacturer's instructions and the analysis was performed on the Infinicyt™ 2.0 software (Cytognos, Salamanca, Spain). The analysis protocol included the removal of threshold debris and lymphocytes were initially identified based on low frontal (FSC) and side scatter (SSC) and strong CD45 staining, followed by the discrimination of helper and cytotoxic T-cells using CD3, CD4 and CD8, as widely known. The B-cells were identified by the CD19 positive staining.
Statistical analysisDescriptive statistics were calculated for continuous variables and categorical variables were summarized using counts and percentage distribution. The Shapiro-Wilk test was used to verify the normality of the data. Comparisons were made using the Kruskal-Wallis test, followed by the post-hoc analysis when necessary. The cutoff value, sensitivity, specificity, accuracy, predictive values (positive predictive value - PPV and negative predictive value - NPV) of the CBC and lymphocyte subset count were defined using the Receiver Operating Characteristic (ROC) curve, with the discharge from the hospital as the outcome in general parameters and death as the outcome in the NLR. The groups studied in the risk analysis were compared using the Chi-squared test and Cramér's V association test. The results were considered significant when p ≤ 0.05.
ResultsPatients characteristicsBetween April and June 2020, 116 consecutive SARS patients were recruited: 76 (62.8%) with confirmed SARS-CoV-2 infection (by molecular test - RT-PCR positive – 64 patients or serological test with IgM positive – 12 patients). All of them had imaging tests and clinical conditions compatible with COVID-19. The remaining 40 (37.2%) patients with other respiratory diseases (not COVID-19) were classified as the control group (Table 1).
Demographic and clinical data.
The data are described as median and IQR or number and percentage. p-value based on Chi-squared test comparing COVID-19 vs. control group. ns= not significant.
There were 47 males (61.8%) in the disease group and 16 (40,0%) in the control group, p=0.025. Based on clinical characteristics and use of MV as suggested by the WHO, the confirmed SARS-CoV-19 group was divided into moderate (n = 41; 35.3%) and severe (n = 35; 30.2%). Patients in the moderate group were younger than those in the severe group (53 vs. 64 years) and control group (53 vs. 63 years). The severe COVID-19 group had a median length of hospitalization of 26 [14;39] days vs. 6 [4;11] days in the moderate group and 8 [3.5;13] days in the control group. The need for the ICU was greater in the COVID-19 severe group (100%) than in the moderate group (26.8%) and control group (47.5%). The median time of the MV support use was 12 [8;19] days in the severe COVID-19 group.
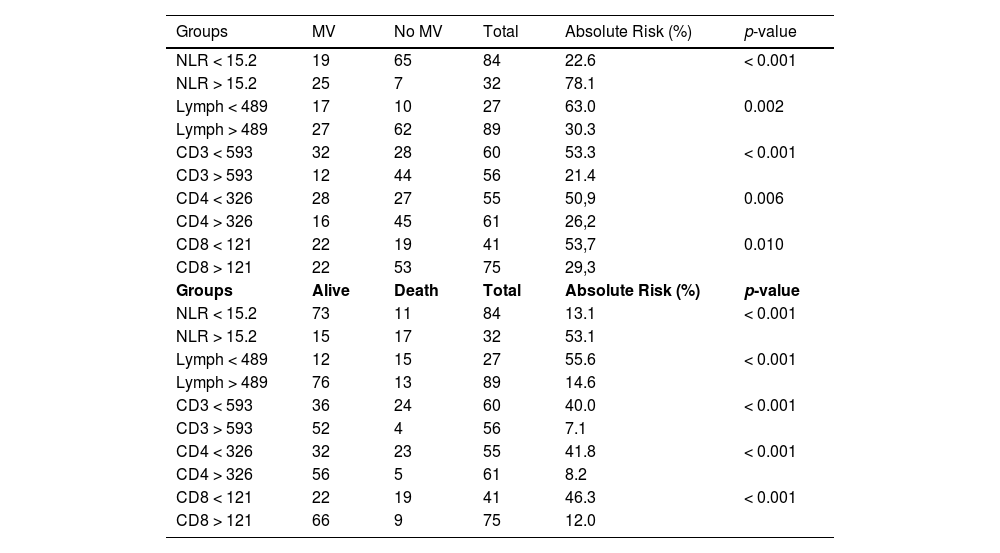
Laboratory resultsAll patients had lymphopenia, although the lymphocyte count was lower in the severe (596 cells/µL), than in the moderate patients (1,052 cells/µL), p < 0.001. It was noted that lymphocyte counts in the COVID-19 groups were not statistically different from that in the control group (836 cells/µL). Severe patients had leukocytosis (11,920 cells/µL) with neutrophilia (10,635 cells/µL), unlike moderate COVID-19 patients (WBC: 7,120 cells/µL, neutrophils: 5,148 cells/µL), p < 0.001.
The T-cell subset counts were different among diseased patients: severe patients had lower CD3+ (374 cells/µL), CD4+ (184 cells/µL) and CD8+ (108 cells/µL) cell counts, when compared to the moderate group (CD3+: 740 cells/µL, CD4+: 389 cells/µL and CD8+: 215 cells/µL), p < 0.001. The total CD3+ cell count in the control group (566 cells/µL) showed a significant difference, when compared to the severe group (p = 0.009). Other counts (CD4+: 356 cells/µL; CD8+: 172 cells/µL) showed no difference with the remaining groups.
Severe patients had lower NK-cell counts (64 cells/µL), when compared to moderate patients (160 cells/µL), p = 0.001 and the control group (90 cells/µL), p = 0.018. The B-cell count was not different between groups (Table 2).
Laboratorial results.
| Characteristics | Control (I) | Moderate (II) | Severe (III) | p-value (IxIIxIII) | IxII | IxIII | IIxIII |
|---|---|---|---|---|---|---|---|
| Globular volume (%) | 35.9 [32.2;41.8] | 41.6 [37.8;44.2] | 38.1 [31.5;42.7] | 0.006 | 0.008 | 1.000 | 0.045 |
| Hemoglobin (g/dL) | 12.0 [10.2;13.4] | 13.9 [12.5;14.4] | 12.3 [10.4;13.8] | 0.001 | 0.001 | 1.000 | 0.015 |
| Platelets (103 units/µL) | 208.5 [161;316] | 269.0 [191;351] | 241.0 [181;360] | 0.142 | - | - | - |
| Leukocytes (cells/µL) | 9565 [6185;13315] | 7120 [5700;9140] | 11920 [8870;16110] | <0.001 | 0.126 | 0.088 | <0.001 |
| Neutrophils (cells/µL) | 7433 [4281;11711] | 5148 [4282;8035] | 10635 [7628;14926] | <0.001 | 0.154 | 0.021 | <0.001 |
| Lymphocytes (cell/µL) | 836 [498;1,370] | 1052 [594;1,413] | 596 [315;977] | 0.001 | 0.526 | 0.040 | <0.001 |
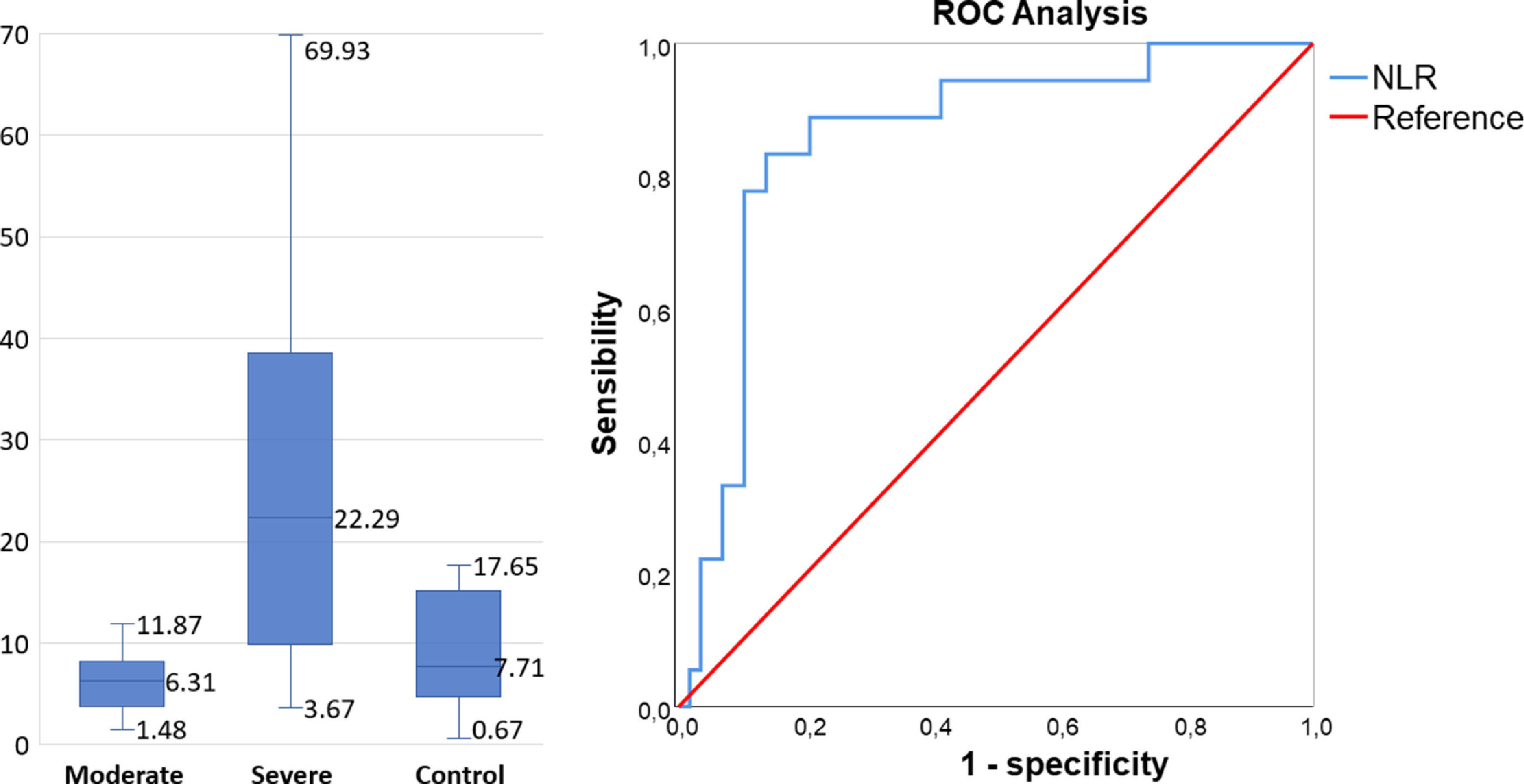
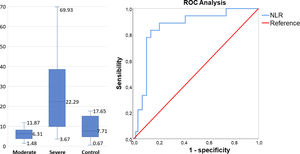
| NLR | 7.71 [4.72;15.07] | 6.3 [3.82;8.16] | 22.29 [9.90;38.60] | <0.001 | 0.082 | 0.003 | <0.001 |
| CD3+ (cells/µL) | 566 [259;962] | 740 [409;1,036] | 374 [146;633] | <0.001 | 0.726 | 0.009 | <0.001 |
| CD4+ (cells/µL) | 356 [132;613] | 389 [244;679] | 184 [84;353] | 0.001 | 0.941 | 0.018 | 0.001 |
| CD8+ (cells/µL) | 172 [93;308] | 215 [142;354] | 108 [60;219] | 0.001 | 0.587 | 0.061 | 0.001 |
| CD19+ (cells/µL) | 89 [33;162] | 125 [81;194] | 115 [65;159] | 0.080 | - | - | - |
| CD56+ (cells/µL) | 90 [34;154] | 160 [101;209] | 64 [31;106] | 0.001 | 0.018 | 0.852 | 0.001 |
| D-dimer a (ng/mL FEU) | 2.01 [0.82;3.45] | 0.58 [0.47;1.65] | 2.21 [1.12;3.65] | 0.004 | 0.044 | 1.000 | 0.004 |
| C-reactive proteinb (mg/L) | 5.95 [2.45;12.37] | 5.24 [1.58;9.39] | 12.66 [8.16;16.00*] | 0.005 | 1.000 | 0.029 | 0.005 |
| Ferritinc (µg/L) | 159.82 [106;320] | 601 [398;2437] | 1052 [427;1675*] | <0.001 | <0.001 | <0.001 | 1.000 |
| Troponind (ng/mL) | 10.0 [10.0;26.6] | 10.0 [10.0;10,1] | 12.2 [10.0;62.8] | 0.093 | - | - | - |
Data are described as median and IQR; p-value based on Kruskal-Wallis with post-hoc analysis.
Moderate patients had a lower D-dimer dosage (0.58 ng/mL), when compared to patients in the severe (2.21 ng/mL, p = 0.004) and control (2.01 ng/mL, p = 0.044) groups. The troponin and CRP dosages were not statistically different between groups. The ferritin dosage was higher in COVID-19 severe (1,052 µg/L) and moderate patients (601 µg/L) than in those in the control group (159.82 µg/L), p < 0.001.
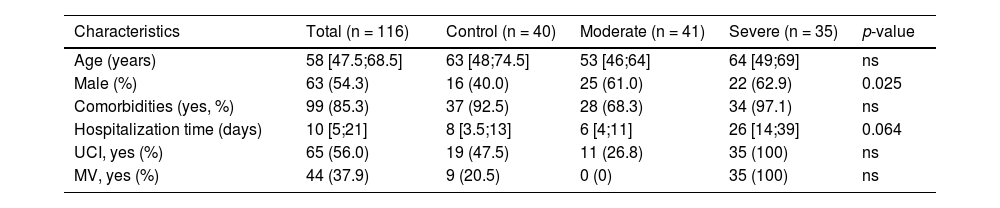
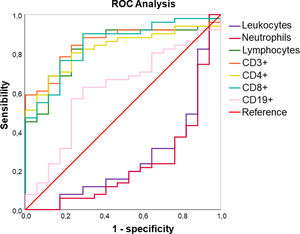
Receiver operating characteristic (ROC) curveA ROC curve was constructed to compare the NLR (Figure 1), leucocyte count and lymphocyte subsets (Figure 2). The hospital discharge was considered as the endpoint for cell counts and death, for the NLR. The results are described in the Supplementary data.
The NLR was higher in COVID-19 severe patients (22.29 vs. 6.3 in moderate and 7.71 in control groups), p < 0.001 (Table 2). Based on these results, the cutoff of 15.2 was related to a higher risk of death (AUC: 0.856, 95% CI: 0.757-0.956, p < 0.001). The NLR analysis showed high sensitivity (83.3%), specificity (86.2%), accuracy (85.5%) and NPV (94.3%), indicating that the higher the ratio elevation was, the worse the clinical outcome.
The overall lymphocyte count (AUC: 0. 836, 95% CI: 0.738-0.935, p < 0.001) and all T-cell subsets (CD3+ AUC: 0.861, 95% CI: 0.777 - 0.945, p < 0.001; CD4+ AUC: 0.833, 95% CI: 0.742 - 0.924, p < 0.001; CD8+ AUC: 0.833, 95% CI: 0.742-0.924, p < 0.001) were highly correlated with the patient recovery.
Total lymphocyte counts below 489 cells/µL showed high sensitivity (91.4%) in detecting patients with COVID-19 and all T-cell subset counts showed high specificity in identifying severe patients (CD3+: 100.0%; CD4+: 94.4%; CD8+: 83.3%). The CD4+ cells above 326 cells/µL and CD8+ above 121 cells/µL had a high PPV (CD4+: 97.3%; CD8+: 93.7%) for improvement and discharge from hospitalization. The CD8+ counts above cutoff had high sensitivity (77.6%), specificity (83.3%) and accuracy (79.9%) for recovery.
OutcomesConsidering all patients, the mortality rate was 24.1%. Most patients in the moderate group recovered from the disease and were discharged from the hospital (n = 39, 95.1%), while only 19 patients (54.3%) in the severe group recovered completely and were discharged from the hospital. A total of 16 patients in the severe group (45.7%) and 2 patients in the moderate group (4.9%) died (p < 0.001). In the control group, 30 patients (75%) recovered from the disease, while 10 patients (25%) died.
Risk assessmentTo determine the absolute risk of the MV use and death, the analysis groups consisted of patients with the NLR, total lymphocyte count, CD3+, CD4+ and CD8+ greater or lower than the established cutoff (Table 3).
Risk assessment of use of VM and death.
NLR= neutrophil-to-lymphocyte relation. MV= mechanical ventilation; p-value based on Chi-squared test.
The NLR showed a strong association with the MV use (Cramér's V = 0.511), followed by the CD3+ count, which showed a moderate association (Cramér's V = 0.329). The overall lymphocyte count and CD4+ and CD8+ counts above the cutoff showed a weaker association with the MV use. Global lymphocyte counts and their subsets above the cutoff were a risk factor associated with death, presenting a moderate association in Cramér's V test.
DiscussionThe COVID-19 pandemic has disrupted the daily lives of millions of people and caused more than 5 million deaths worldwide.17 The illness resulting from this viral infection presents a range of possible symptoms and has a clinical course that is difficult to predict. Therefore, it becomes increasingly necessary to implement early indicators of severity and support clinical management. For example, rapidly identification of cases with the potential for clinical worsening could help hospitals prioritize care and allocate critical (and often limited) resources, such as ICU beds and ventilators. Similarly, identifying low-risk cases could help reduce hospital admissions and decrease patient anxiety.18
In Brazil, several factors may have contributed to the historical record of cases and deaths by COVID-19 during the pandemic. The lack of investment in health care resulted in the overcrowding, sometimes beyond the limit, of ICUs throughout the country, bringing the public health system to the edge of collapse.19
In the present study, patients with moderate COVID-19 were younger and had a shorter hospital stay than patients with severe COVID-19 and the control group. These, in turn, had a longer ICU requirement, indicating that, although they had not been confirmed as having the SARS-CoV-2 infection, the acute respiratory syndrome was of considerable clinical relevance.
This study was conducted in a public, highly complex hospital with a higher associated risk in care due to the severity of the diseases seen and high demand for medical-hospital support in care.20 One indicator that can be used to estimate the severity of patients treated is the rate of mechanical ventilation demand. In this study, 44% of patients used the MV. Other Brazilian centers report a rate of approximately 50%, while European and North American countries report rates ranging from 12 to 26%.21
The NLR in patients with COVID-19 has been found to be a valuable laboratory marker in the follow-up of severe and moderate patients and a significant discrepancy between neutrophils and lymphocytes has been used as a laboratory marker of severity.22 In an early cohort of 210 patients treated in Wuhan,22 the mean NLR of the severe group was higher than that of the mild group (6.6 vs. 3.3, p < 0.001). The current study showed a lower NLR ratio in moderate patients (6.3) and an extremely high value in the severe group (22.3). These results suggests that patients with an increased NLR should be admitted to an isolation ward with respiratory monitoring and supportive care, even when they did not have acute clinical manifestations. Furthermore, patients with an NLR lower than the established cutoff value (15.2) are less likely to progress to death, with a high negative predictive value.
It is likely that T-cell counts and their subset (CD4+ and CD8+) below cutoff values were directly related to the need for invasive ventilatory support and death as a clinical outcome, suggesting that assessment of these parameters may be predictive of a worse prognosis in the COVID-19 disease.
The host immunity of COVID-19 patients with varying degrees of disease severity has been evaluated since the first report and the number of absolute total lymphocytes and T-cell subsets (CD4+ and CD8+) appears to be lower in extremely severe and severe cases, compared to moderate cases.3,13,14 In addition, several studies have suggested that low T-cell counts and subsets in patients with severe COVID-19 are a poor prognostic factor.23–25 Similarly, the present study showed that severe patients have a unique profile, with dramatically lower counts of CD3, CD4 and CD8, compared to patients with moderate COVID-19, indicating that the T-cell lymphopenia is highly related to the COVID-19 infection.
The ability of a laboratory test to predict clinical outcome or status (hospital discharge vs. decease) was assessed by the area under the curve (AUC) after performing a receiver operating characteristic (ROC) curve. Thereafter, cutoff values for a variable were employed to determine the positive and negative predictive values and other parameters.
In our study, patients with severe lymphopenia, low T-cell subsets and high NLR were more likely to use mechanical ventilation or die, compared to the opposite group. In this context, lymphocyte counts below 489 cells/µl and the T-cell lymphopenia below 593 cells/µL, as well as the CD4 below 326 cells/µL had worse outcomes than patients with higher values. Therefore, T-lymphocyte counts as well as CD4 T-cell counts at the beginning of the hospitalization can provide valuable information that can help in the rational distribution of supplies to patients who are inclined to develop a disease with greater complications. Incidentally, the NLR beyond the cutoff, along with a lymphocyte count above the defined limits, were shown to be strong risk factors associated with death, presenting a variable association with the outcome in the Cramér's V test.
The number of vaccinated individuals is growing daily and all currently available vaccines promote an important immune response by providing a means to control the infection.26,27 Vaccination is a highly effective preventive strategy against COVID-19. The induction of the immune response by vaccination can lead to two major outcomes: the secretion of antibodies by antibody-producing cells and the production of memory B and T cells, responsible for long-term immune response against pathogens.28 After the complete immunization cycle (including the booster shot), a significant increase in the total lymphocyte count is expected due to the stimulation in the development of memory B and T cells.29,30 As the main results of this study refer to lymphocyte (total and subsets) counts and vaccination determines the activation of T and B lymphocytes, which could last for months, the NLR may not be a useful laboratory marker in vaccinated patients with COVID-19. Therefore, the risk stratification proposed in this study should be used with precaution when considering fully vaccinated patients. In these situations, assessment of other biochemical and respiratory parameters, in addition to the clinical performance of the patient is recommended.
ConclusionIn conclusion, the laboratory assessment of the neutrophil/lymphocyte ratio (NLR) and T-cell subsets in hospital admission was predictive of mortality and may be useful for stratifying COVID-19 patients, helping in managing hospital resources. While there was no vaccination program implemented, this strategy was useful for attending and directing hospital resources to patients with a higher risk of a worse evolution. Currently, this strategy can still be employed for unvaccinated patients, which correspond to the highest number of hospitalizations.
The authors are grateful to Maria Tadeu Lemes Rocha and Edna Aparecida Martins for research assistance and to Hugo Morales, Sonia Raboni and Hipólito Carraro Junior, ULAC, PRIMAH/COREMU and CHC/UFPR for support.