The clinical utility of more recent hematological parameters, such as the analyzer-derived platelet (PLT) indexes, has been explored in recent years.1 The mean platelet volume (MPV) and platelet distribution width (PDW) have been reported to be correlated with the PLT function and may be more sensitive indexes than the PLT count as a marker of clinical interest in various disorders, such as cardiovascular disease and stroke.2 The platelet-large cell ratio (P-LCR), when analyzed along with the other PLT parameters, helps in the differential diagnosis of the thrombocytosis-related disease.3 Meanwhile, recent studies have explored the role of the plateletcrit (PCT) as independent risk factors of spontaneous echo contrast in patients with mitral stenosis.4 Regarding immature platelet fraction (IPF), is important to highlight that this is a low-cost, reproducible and accurate technology in the assessment of reticulated platelets.5 The IPF has been shown to be able to screen and differentiate thrombocytopenia from different causes.6 Many of these indexes are not being used in the clinical and laboratory routine due to the absence of reference values for each population evaluated. Since the use of reference intervals described in other populations may not reflect the same characteristics of the population evaluated, an adequate interpretation of the laboratory results depends on the evaluation of local reference values. It is already well established that significant differences in these values can occur according to different geographic regions.7
In this context, the objective of this study is to determine the reference values of new hematological platelet indexes (MPV, PCT, PDW, P-LCR and IPF) available in the most recent automation in order to facilitate its use in the laboratorial and clinical routine.
A total of 197 participants (81 females), aged between 18 and 65 years, who volunteered at the Hemocentro Regional de Santa Maria (blood center) for blood donation from February to March 2017 was included. Of the participants, 175 (88.8%) were self-reported to be Caucasians. All participants included were routinely screened for hepatitis B and C, HIV (human immunodeficiency virus), Chagas disease, syphilis and HTLV (Human T lymphotropic virus). As long as all of the serology was not reagent, the participant was considered suitable for blood donation. The Institutional Ethics Committee from the Federal University of Santa Maria approved this study (CAAE 12303113.0.0000.5346) and a written informed consent was obtained from all participants prior to the inclusion in the study.
Whole blood in K3-EDTA tubes was used to measure the PLT, MPV, PCT, PDW, P-LCR and IPF, using a Sysmex XE-5000 (Sysmex®, Japan). In this equipment, the MPV count method employs hydrodynamic focus and direct current technology; the PCT, PDW, P-LCR and IPF were determined by fluorescence and scattered light, using fluorescent RNA/DNA markers.
For the determination of the reference value of non-parametric continuous variables, the interquartile range of 2.5–97.5 was used, as recommended by the IFCC.8 The groups were compared using the Mann–Whitney test. Statistical significance was assumed with p<0.05. The data were analyzed using the GraphPad Prism®, version 4.00 for Windows (GraphPad Software, CA).
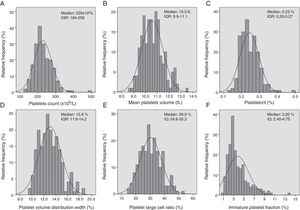
The median age (min, max) of the total group was 33 (23–44) years. The median age of males and females was 34 (23–46) and 32 (24–42) years, respectively (p=0.766). The data for the PLT, MPV, PCT, PDW, P-LCR and IPF in healthy individuals are shown as median and 2.5% and 97.5% percentiles, and additionally stratified by age ranges and sex in Table 1. While the distribution of the complete reference data for these parameters is presented in a scatter plot (Figure 1), no statistical differences were observed between males and females for the MPV, PDW, P-LCR and IPF (p>0.05). However, the females presented a higher PLT (p<0.001) and PCT (p<0.001), when compared with the males. The participants were stratified into three groups according to age as follows: group 1: 18–30 years (n=86); group 2: 31–45 years (n=66); and group 3: 46–65 years (n=45). The PCT was higher in group 1, compared to group 3 (p=0.016), while no differences were observed in the PLT, MPV, PDW, P-LCR and IPF (Table 1).
Reference interval for platelet count (PLT) and platelet indexes according to the age and sex in 197 healthy individuals from Santa Maria, Brazil.
| All | Age | p value | Sex | p value | ||||
|---|---|---|---|---|---|---|---|---|
| n=197 | 18–30 yn=86 | 31–45 yn=66 | 46–65 yn=45 | Malen=116 | Femalen=81 | |||
| PLT (×103/μL) | 220 (149–323) | 231 (159–321) | 220 (143–418) | 207 (141–311) | 0.054 | 211 (140–288) | 242 (161–385) | <0.001 |
| MPV (fL) | 10.5 (9.1–12.5) | 10.6 (9.2–12.5) | 10.4 (8.9–12.8) | 10.5 (10.5–12.6) | 0.547 | 10.5 (9.0–12.2) | 10.5 (9.4–12.7) | 0.575 |
| PCT (%) | 0.23 (0.17–0.35) | 0.25 (0.18–0.35)a | 0.23 (0.16–0.43)a,b | 0.22 (0.05–0.33)b | 0.016 | 0.22 (0.14–0.30) | 0.26 (0.18–0.40) | <0.001 |
| PDW (%) | 12.8 (9.9–17.7) | 12.9 (10.6–17.6) | 12.9 (9.7–18.0) | 13.0 (9.9–17.5) | 0.545 | 12.9 (9.9–17.1) | 12.7 (10.3–17.8) | 0.880 |
| P-LCR (%) | 29.9 (17.3–46.4) | 30.3 (18.8–46.6) | 29.1 (15.8–48.2) | 29.3 (15.3–45.4) | 0.418 | 29.9 (16.5–44.4) | 29.3 (19.3–46.9) | 0.644 |
| IPF (%) | 3.20 (1.30–9.12) | 3.30 (1.40–8.76) | 2.90 (1.14–9.76) | 3.40 (1.20–13.1) | 0.586 | 3.30 (1.20–8.89) | 2.90 (1.40–9.57) | 0.180 |
Data were expressed as a median (2.5–975). MPV: mean platelet volume; PCT: plateletcrit; PDW: platelet distribution width; P-LCR: platelet large cell ratio; IPF: immature platelet fraction. Differences between the groups were tested using Mann–Whitney or Kruskal–Wallis test and when significant the Dunn's multiple comparison test was performed. Labeled means in a row without a common superscript letter differ, p<0.05.
Bold value mean significant value of p.
The reference values for the MPV found in this study were similar to the values found in 580 Brazilian participants in the ELSA-Brazil study7 and 1670 healthy Danish individuals9 [10.5fL (8.9–12.2), using the Sysmex XE 5000, and 10.5fL (8.9–12.2), using the Sysmex XE 2100 D, respectively]. Regarding the PDW, the values obtained in our study were very similar to those previously reported in 231 Brazilian subjects10 [13.3% (10.0–17.9)], using the Pentra 120 analyzer (Horiba Medical, France) and in 816 healthy South Korean pregnant women at 20 weeks of gestation11 [PDW 11.6% (8.9–15.5)]. In regard to the PCT, our results are comparable to those previously described in 565 healthy pregnant Chinese women, 0.17% (0.12–0.27) using a Coulter 780 5Diff analyzer (Beckman Coulter, USA)12; and in 945 healthy blood donors in India, whose reported interval was 0.15–0.62%, using a Sysmex XE 2100 D (Sysmex, Japan).13 In a study with 1882 Italian patients undergoing coronary angiography, De Lucca et al. (2010) obtained an average P-LCR of 28.8% for the studied groups, using the Sysmex XE 2100 (Sysmex, Japan).14 These results are comparable with those found in our study. As for the IPF, it has been receiving particular attention because it has demonstrated the capacity to differentiate the types of thrombocytopenia.6 In a study performed with 945 blood donors for the IPF reference range determination, a range of 0.3–8.7% was observed.13 Such results are similar to the range obtained by us.
The evaluated indexes were not affected by the age and most of the indexes did not present differences as for the sex, with the exception of the PLT and PCT, which were higher in men. The reference interval obtained in our study resembled those previously described in the literature, in a general manner. Thus, such values reported in this study contributed to the clinical application of these indexes.
Conflicts of interestThe authors declare no conflicts of interest.
We thank Dr. Helena Zerlotti Wolf Grotto for the scientific support and the medical advice on the Sysmex. We thank Dr. Elehu Moura de Oliveira and the Lac/HUSM for the technical support. We also thank all those who participated in this study.