The estimated involvement of the central nervous system in patients with multiple myeloma is rare at about 1%. The infiltration can be identified at the time multiple myeloma is diagnosed or during its progression. However, it is more common in refractory disease or during relapse.
MethodsThis retrospective cohort study reviewed data from medical records of patients followed up at the Gammopathy Outpatient Clinic of Santa Casa de Misericórdia de São Paulo from January 2008 to December 2016.
ResultsTwenty patients were included, with a median follow-up of 33.5 months after central nervous system infiltration. The prevalence was 7%. The median age at diagnosis of multiple myeloma was 56.1 years, with 70% of participants being female. Sixteen patients had central nervous system infiltration at diagnosis of multiple myeloma. Seventeen patients had exclusive osteodural lesions and three had infiltrations of the leptomeninge, of which one had exclusive involvement and two had associated osteodural lesions. The median overall survival was 40.3 months after central nervous system involvement. The median overall survival in the group with central nervous system infiltration at relapse was 7.4 months. The patients with leptomeningeal involvement had a median overall survival of 5.8 months.
ConclusionCentral nervous system infiltration is a rare condition, but it should be considered as a possibility in patients with multiple myeloma and neurological symptoms. The best treatment regimen for this condition remains unknown and, in most cases, the prognosis is unfavorable.
Multiple myeloma (MM) is characterized by the clonal proliferation of plasma cells, which produce monoclonal immunoglobulins found in blood and/or urine.1 Generally, these plasma cells are confined to the bone marrow and vascular compartment; however, dissemination may occur through the bone cortex or hematogenically to other organs, resulting in extramedullary disease.2
Extramedullary involvement in MM is rare, occurring in only 3–5% of patients and usually involving the skin, nasopharynx, larynx, upper respiratory tract and central nervous system (CNS).3 CNS involvement, estimated at 1% of patients, is uncommon.4
The definition of CNS involvement in MM is controversial and differs depending on the group of investigators. Such involvement may occur as single or multiple intraparenchymatous lesions and/or leptomeningeal involvement.5 It is defined by the presence of plasma cells in the cerebrospinal fluid (CSF) and/or leptomeninge, dura mater or intraparenchymatous involvement as assessed by imaging tests and confirmed by CSF analysis, magnetic resonance imaging (MRI), computed tomography (CT) scans and/or tissue biopsy.6,7
The CNS involvement by MM confers a reserved prognosis, with an overall survival (OS) of around 1.5–2 months when there is leptomeningeal infiltration.4–6 For those cases with osteodural involvement, the median survival ranges from one year to 25 months.3,8
The best treatment for MM with CNS involvement is not well defined yet. The therapies used are intrathecal chemotherapy, radiotherapy, and systemic therapy in addition to autologous hematopoietic progenitor cell transplantation (AHPCT).6 These therapies can be used alone or as a combination.
In the present study, 20 patients with MM and CNS infiltration were evaluated with the objective of contributing to a better understanding of the biology, clinical behavior and their treatment, as well as to assess the OS. This is the largest Brazilian single-center study of patients with MM and CNS involvement.
ObjectivesTo evaluate the profile of patients diagnosed with MM and CNS involvement including the OS, and to correlate infiltration with predictive factors and prognostic impact.
MethodsThis is a retrospective cohort study of patients with MM and CNS involvement followed up at the Gammopathy Outpatient Clinic of Santa Casa de Misericórdia de São Paulo from January 2008 to December 2016.
Initially, patients with CNS involvement during that period were identified in the database of the outpatient clinic and a review was carried out of their medical records in the institution's Medical and Statistical Archive Service.
Data regarding the clinical and demographic characteristics of the patients, their diagnosis of MM and CNS infiltration were collected, as well as data on treatment and response.
Inclusion and exclusion criteriaPatients diagnosed with MM with CNS involvement at the time of diagnosis or during progression were included in this study. CNS involvement was defined as having a dura mater lesion with contiguity to the CNS, isolated intraparenchymatous lesions, lesions starting from nasopharyngeal plasmacytoma and those with leptomeningeal involvement as detected by the presence of clonal plasma cells in the CSF and/or by imaging.
Patients were excluded when data in the medical records were incomplete making it impossible to confirm the diagnosis of CNS involvement.
Statistical analysisContinuous variables were summarized by means of variation (minimum and maximum values), mean, standard deviation (SD), median and interquartile range. Categorical variables are described by means of absolute and relative frequencies. The Kolmogorov–Smirnov test was used to evaluate the distribution pattern of the numerical variables in the sample. Survival analyses were performed using the Kaplan–Meier technique and comparison across groups by the log-rank test. All analyses were performed using MedCalc software (Mariakerke, Belgium, V. 11.3.3.0). Two-tailed significance levels of 5% were used as indicative of statistical difference across groups.
ResultsFrom January 2008 to December 2016, 285 patients with MM were followed up at the Gammopathy Outpatient Clinic at Santa Casa de Misericórdia de São Paulo. Of these cases, 21 were identified with CNS involvement, but one was excluded due to incomplete data in their medical record. Accordingly, 20 patients were eligible for the present study. These patients were diagnosed with MM between February 2002 and September 2015.
The median follow up of the patients since the diagnosis of MM was 38.5 months, while the median after CNS infiltration was 33.5 months.
The prevalence of patients with CNS involvement in the period studied was 7%.
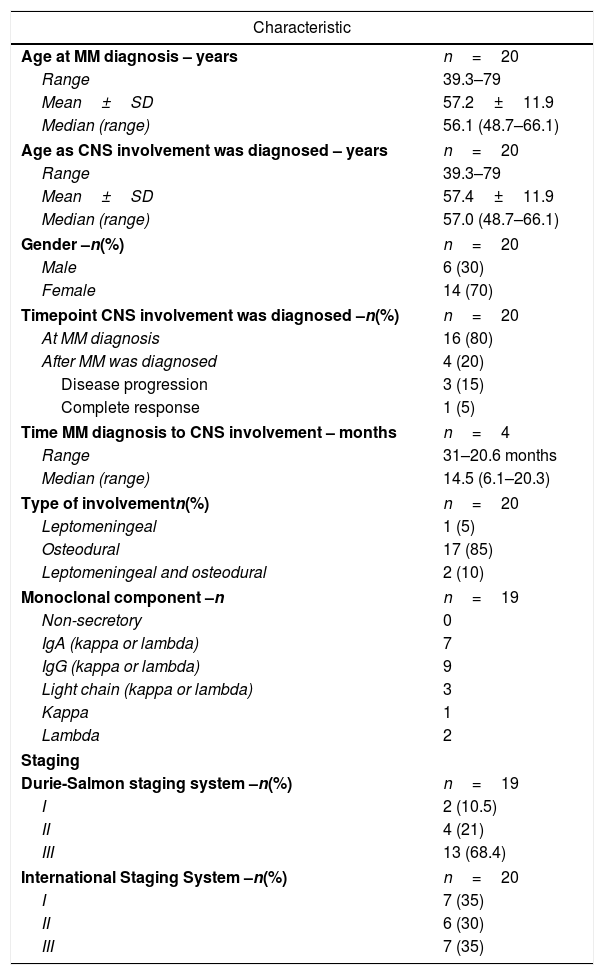
Six (30%) patients were male and 14 (70%) were female. The median age at MM diagnosis was 56.1 years (range: 49.0–66.1 years) and at CNS involvement it was 57.0 years (range: 49.0–66.0 years).
The most frequent immunoglobulin heavy chain subtype was IgG (kappa and lambda – 9 cases) followed by IgA (kappa and lambda – 7 cases). Three patients had light chain disease (one kappa and two lambda).
At diagnosis of MM, two patients (10.5%) had Durie-Salmon stage IA, four (21%) had stage IIA and 13 (68.4%) had stage IIIA/B.
According to the International Staging System (ISS), seven patients (35.0%) had stage I, six (30.0%) had stage II and seven (35.0%) had stage III.
CNS involvement was identified in sixteen patients (80%) at MM diagnosis and in four (20%) during disease progression, of which one patient had a complete response (CR). The median interval between MM diagnosis and the diagnosis of CNS infiltration was 14.5 months (range: 6.1–20.3 – Table 1).
Demographic characteristics of the study population.
| Characteristic | |
|---|---|
| Age at MM diagnosis – years | n=20 |
| Range | 39.3–79 |
| Mean±SD | 57.2±11.9 |
| Median (range) | 56.1 (48.7–66.1) |
| Age as CNS involvement was diagnosed – years | n=20 |
| Range | 39.3–79 |
| Mean±SD | 57.4±11.9 |
| Median (range) | 57.0 (48.7–66.1) |
| Gender –n(%) | n=20 |
| Male | 6 (30) |
| Female | 14 (70) |
| Timepoint CNS involvement was diagnosed –n(%) | n=20 |
| At MM diagnosis | 16 (80) |
| After MM was diagnosed | 4 (20) |
| Disease progression | 3 (15) |
| Complete response | 1 (5) |
| Time MM diagnosis to CNS involvement – months | n=4 |
| Range | 31–20.6 months |
| Median (range) | 14.5 (6.1–20.3) |
| Type of involvementn(%) | n=20 |
| Leptomeningeal | 1 (5) |
| Osteodural | 17 (85) |
| Leptomeningeal and osteodural | 2 (10) |
| Monoclonal component –n | n=19 |
| Non-secretory | 0 |
| IgA (kappa or lambda) | 7 |
| IgG (kappa or lambda) | 9 |
| Light chain (kappa or lambda) | 3 |
| Kappa | 1 |
| Lambda | 2 |
| Staging | |
| Durie-Salmon staging system –n(%) | n=19 |
| I | 2 (10.5) |
| II | 4 (21) |
| III | 13 (68.4) |
| International Staging System –n(%) | n=20 |
| I | 7 (35) |
| II | 6 (30) |
| III | 7 (35) |
Lactate dehydrogenase (LDH) values were elevated in 36.8% of patients at MM diagnosis and elevated in 38.9% at the diagnosis of CNS infiltration.
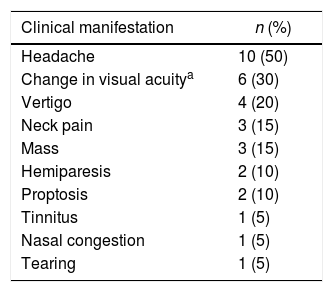
The neurological signs and symptoms related to CNS involvement were headache (50.0%), visual hypoacuity (30.0%), vertigo (20.0%), neck pain (15.0%), brain mass (15.0%), proptosis (10%), nasal congestion, tearing, and tinnitus. Some patients presented more than one neurological symptom (Table 2).
Neurological manifestations when central nervous system involvement was diagnosed.
| Clinical manifestation | n (%) |
|---|---|
| Headache | 10 (50) |
| Change in visual acuitya | 6 (30) |
| Vertigo | 4 (20) |
| Neck pain | 3 (15) |
| Mass | 3 (15) |
| Hemiparesis | 2 (10) |
| Proptosis | 2 (10) |
| Tinnitus | 1 (5) |
| Nasal congestion | 1 (5) |
| Tearing | 1 (5) |
Eighteen patients (90%) underwent a CT scan of the skull, while 16 (80%) had their skull scanned by MRI. All patients had been submitted to at least one of the tests.
As identified both by MRI and CT scans, the most affected CNS site was the sphenoid, followed by the clivus and the parietal region.
Nine patients underwent CSF examination with oncotic cytology but only two patients were positive (1% and 9% plasma cells). CSF immunophenotyping was not performed therefore clonality could not be confirmed. In addition, in one case, although the CSF was negative, brain MRI showed signs of leptomeningeal infiltration. Thus, there were three patients with leptomeningeal involvement, one exclusive and two patients with concomitant osteodural involvement; exclusive osteodural infiltration was observed in the other 17 patients, none of whom had intraparenchymatous involvement alone.
One patient with CNS involvement at MM diagnosis did not undergo systemic treatment (16), whereas another patient was lost to follow-up. Therefore, 14 patients underwent some type of systemic treatment including high doses of dexamethasone (two patients), based on alkylating agents (six patients), proteasome inhibitor (three patients), or immunomodulator (three patients). Of these patients, eight underwent AHPCT.
With regard to a CNS-specific treatment received by the patients with infiltration at the time the disease was diagnosed, three did not perform any type and one had no information about the treatment received. Twelve patients underwent radiotherapy, one patient was given intrathecal chemotherapy, and another patient was submitted to both.
Responses to treatment for patients with CNS involvement at diagnosis of MM were complete response (two patients), partial response (PR – six patients), stable disease (five patients) and three patients had no information on the response.
Regarding the patients diagnosed with CNS infiltration during disease progression, two received one treatment regimen based on an immunomodulator, whereas the other two received two treatment regimens; the first regimen was based on an immunomodulator in one patient and on alkylating agents in the second, whereas the second treatment was polychemotherapy.
The systemic treatment following the diagnosis of CNS involvement was based on a proteasome inhibitor and an immunomodulator in one patient, only an immunomodulator in one and polychemotherapy in two patients. Two of these patients underwent AHPCT before CNS infiltration had been diagnosed, one was diagnosed after transplant and one was not transplanted.
The CNS-specific treatment for patients with CNS infiltration during disease progression was radiotherapy (one patient), intrathecal chemotherapy (two patients), and one patient did not undergo any treatment.
The treatment response for CNS involvement during progression was disease progression in two patients, while no information was available for the other two patients.
Up to the last follow-up, eight patients had died, seven of whom due to disease progression and one due to sepsis originating in the abdomen and progression.
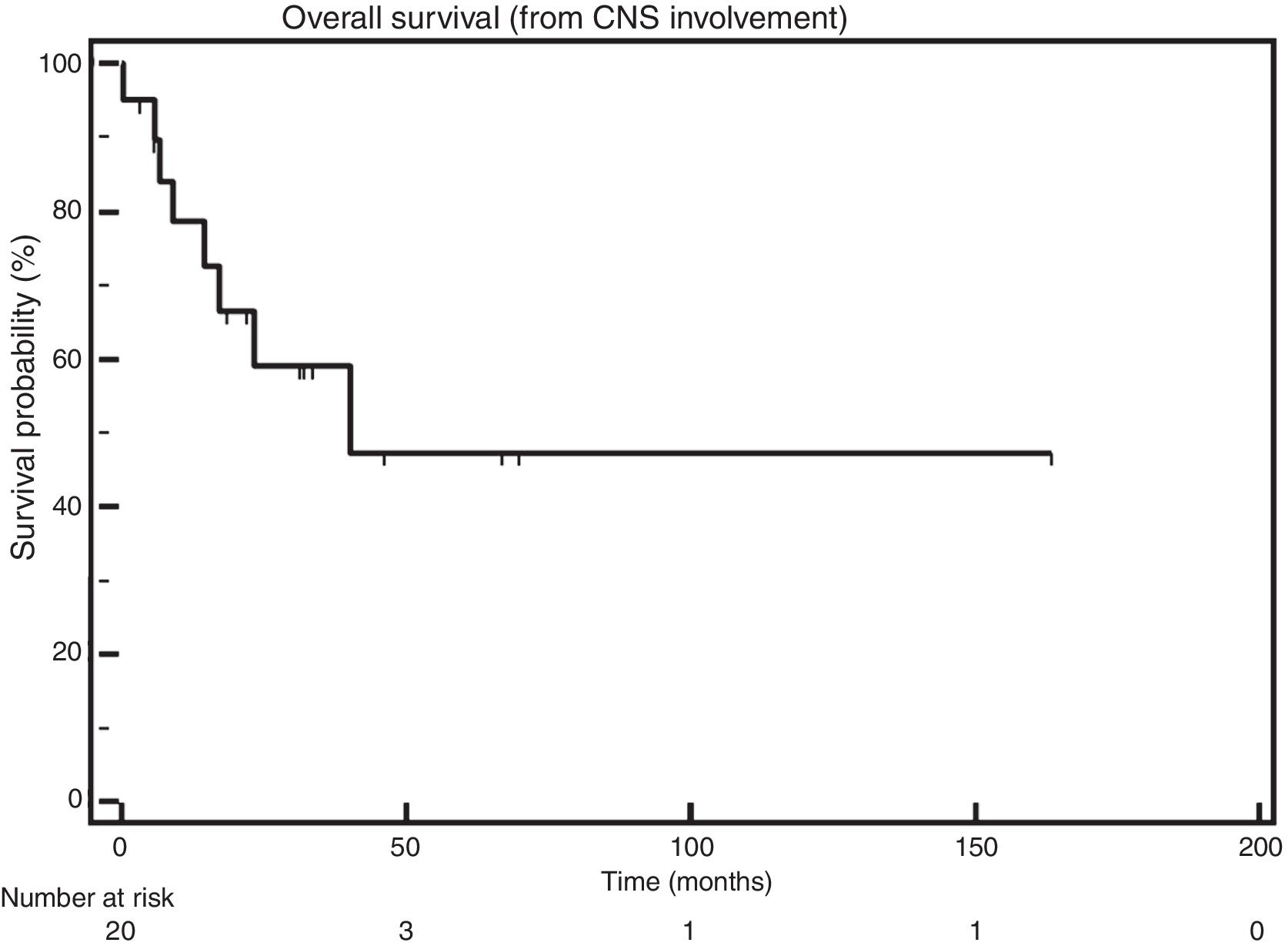
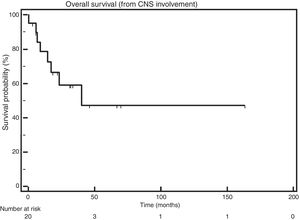
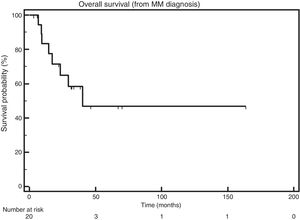
The median OS after CNS infiltration was 40.3 months (Figure 1).
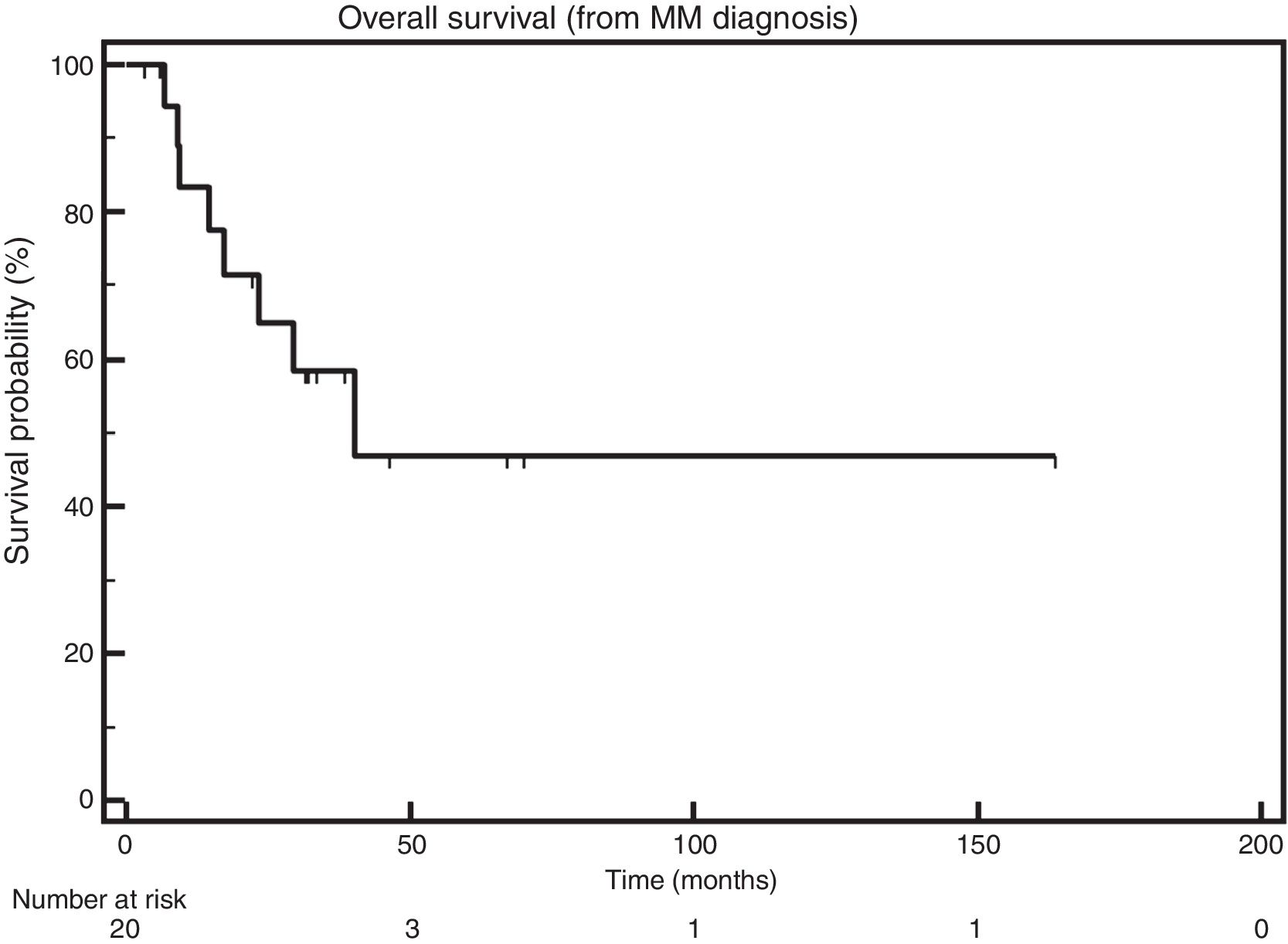
The median OS after MM diagnosis was also 40.3 months (Figure 2).
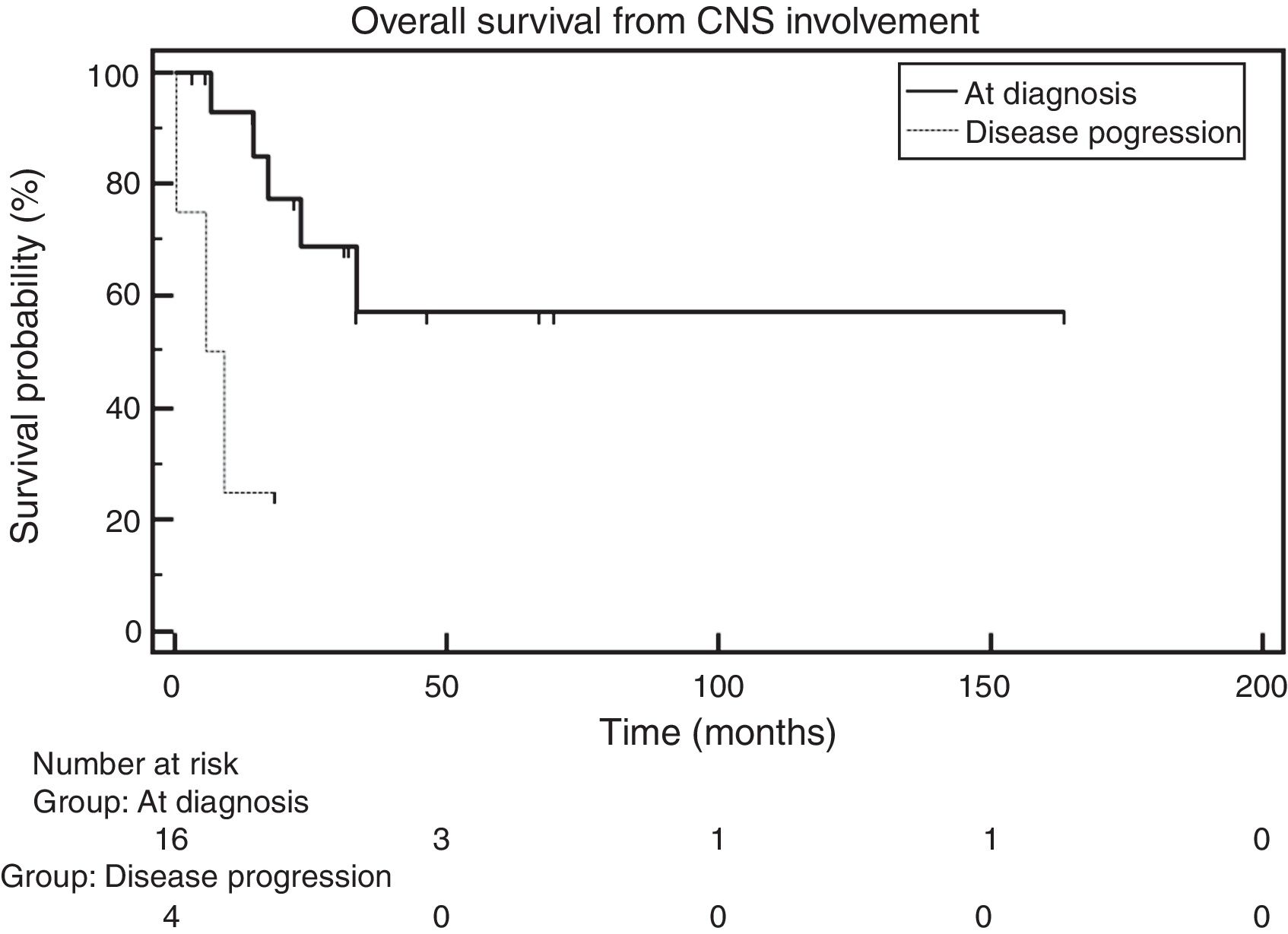
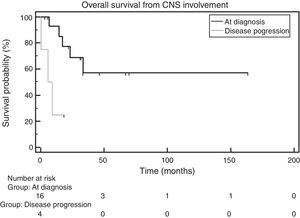
When OS was analyzed between the group of patients with CNS involvement at diagnosis and the one with CNS involvement during relapse, the median OS was 7.4 months in the former, whereas it had not been reached in the latter. The comparison across groups using the log-rank test showed a statistically significant difference [hazard ratio (HR): 5.51; 95% confidence interval (95% CI): 0.537–56.607; p-value=0.0069 – Figure 3].
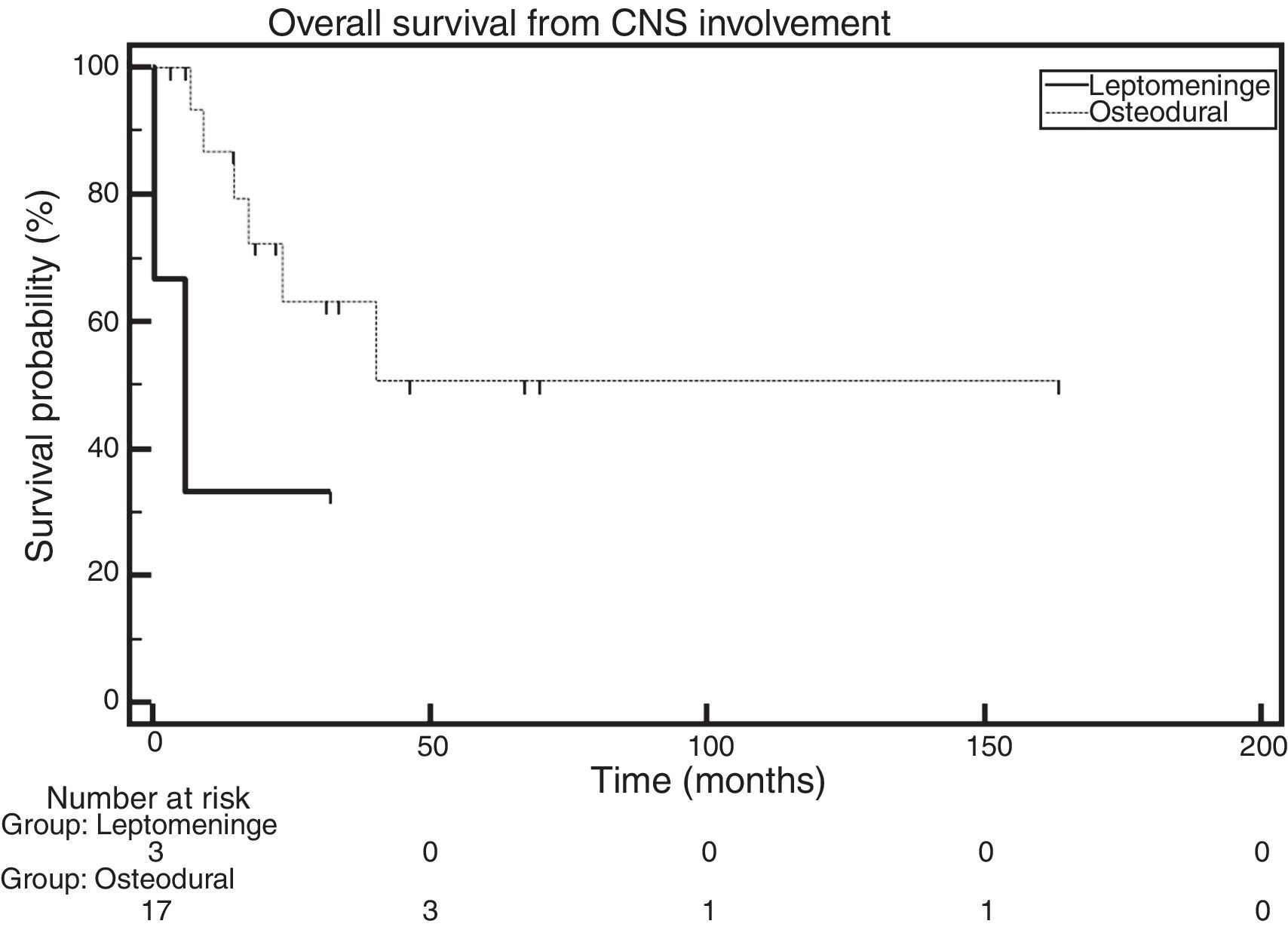
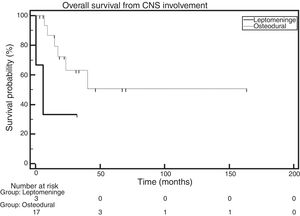
OS was also analyzed while considering the location of the disease (osteodural and leptomeningeal). Figure 4 depicts the analysis of the OS of these groups, defined as the time elapsed between the diagnosis of CNS involvement and death. The median OS in the group of patients with leptomeningeal involvement was 5.8 months, whereas the median OS in the group with osteodural involvement had not been reached. The comparison made with the log-rank test showed no statistically significant difference across groups (HR: 0.27; 95% CI: 0.022–3.35; p-value=0.0823). It is important to note that, in the group with leptomeningeal involvement, there are only three patients (Figure 4).
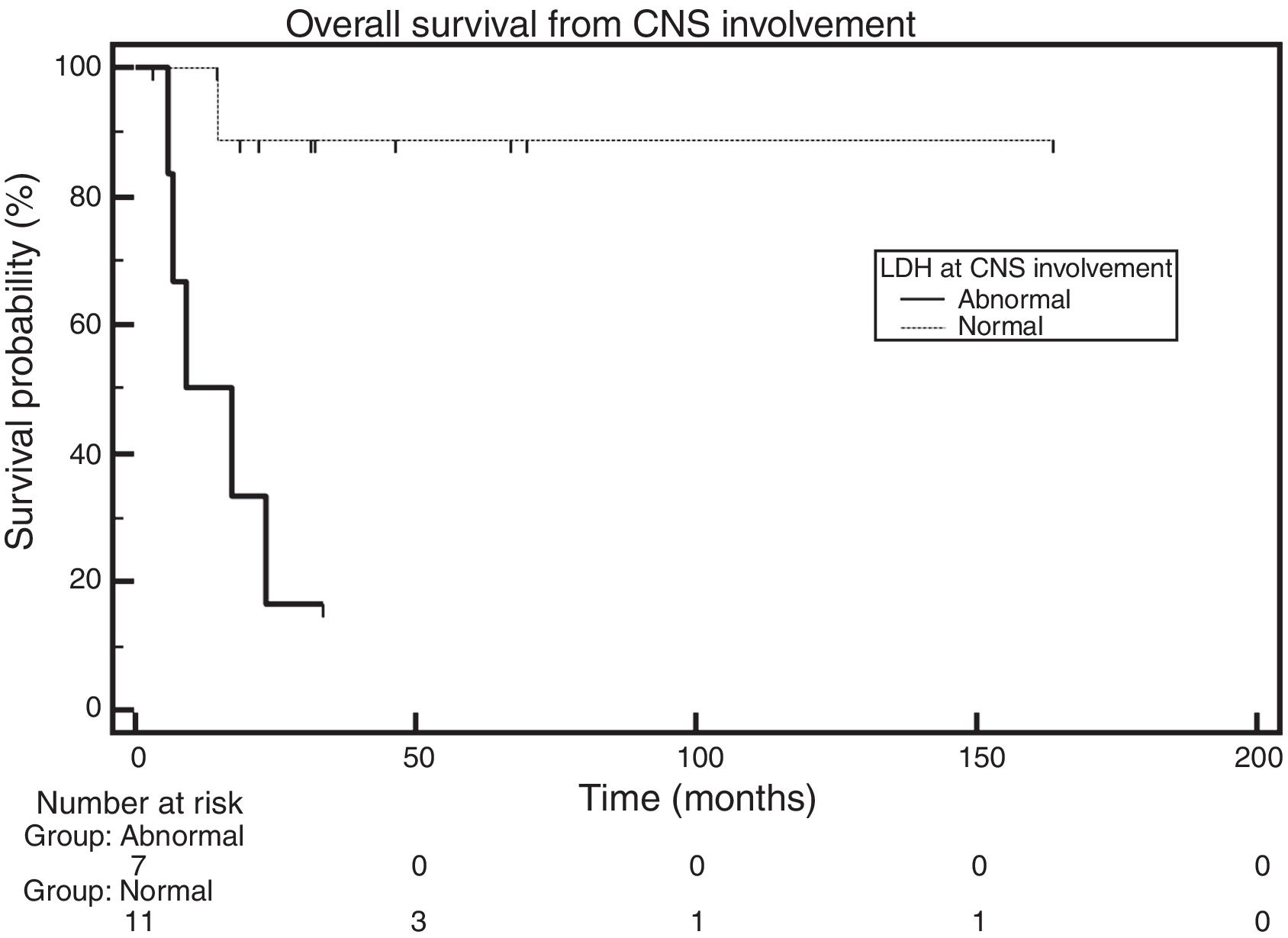
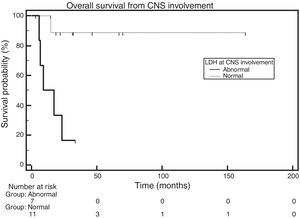
OS was also analyzed according to LDH levels. Of the 20 patients, 11 had normal LDH levels at the diagnosis of CNS infiltration, seven had increased LDH levels, and there was no information available for two patients. The median for the group with increased and normal levels was 13.05 months and had not been reached, respectively. The comparison made with the log-rank test showed a statistically significant difference across groups (HR=0.082; 95% CI: 0.014–0.478; p-value=0.0033 – Figure 5).
DiscussionPatients with MM usually have neurological complications. These include peripheral neuropathy, radiculopathy, spinal cord compression and metabolic encephalopathy. The causes include fracture with vertebral compression, involvement of the base of the skull and other bone regions, metabolic changes (hypercalcemia, uremia and hyperammonemia), hyperviscosity syndrome and amyloidosis, in addition to treatment-related toxicity.9,10
CNS infiltration in MM is rare and poorly described in the literature. The largest retrospective multicenter study has a sample of 172 patients.11 The largest single-institution study was that by a group from Canada with 37 patients.6 Twenty patients with CNS infiltration in MM were identified in this retrospective study. Infiltration was defined as leptomeningeal involvement with positive CSF and/or signs of infiltration on imaging scans (MRI or CT), intraparenchymatous lesion alone or with osteodural extension.
The prevalence of CNS involvement in MM varies in the literature from 1%4 to 1.8%.12 In the current study, the prevalence in the period from January 2008 to December 2016 was 7%.
Regarding the time of CNS involvement, in the literature, most cases occur as the disease relapses.6,7,11,13,14 In a retrospective study of 37 patients, 24% of cases were identified as having infiltration at MM diagnosis, while 76% had infiltration as the disease relapsed.6 Similarly, in a multicenter retrospective study of 172 patients, 78% were diagnosed with CNS infiltration as the disease relapsed.11 The Gruppo Italiano Malattie EMatologiche dell’Adulto (GIMEMA) retrospectively evaluated 50 patients with CNS infiltration (leptomeningeal or osteodural involvement) with 36% having CNS involvement at diagnosis, while 64% had infiltration as the disease relapsed.
In the current study, 80% of the patients had CNS involvement at MM diagnosis. This finding and the higher prevalence of CNS infiltration are possibly explained by the fact that Santa Casa de São Paulo is one of the referral hospitals in the southeastern region of Brazil for the National Health Service (SUS). Thus, it receives patients from all over the country, many of whom take too long to obtain medical care. Due to the difficulty in accessing public healthcare services, patients are admitted when their disease is at a very advanced stage.
Another possibility for disagreement with the literature regarding the time of CNS involvement and prevalence may be the presence of molecular and genetic alterations that would posit these patients at high risk. To date, few studies in the literature have evaluated the biology of patients with MM and CNS involvement. Chang et al.15 found chromosome 17p13.1 deletion in 89% of patients with CNS involvement. Unfortunately, the Brazilian National Health Service does not perform molecular testing. Hence, we did not have access to this type of information in order to confirm this hypothesis. Prospective studies on molecular evaluation in MM and CNS involvement need to be performed.
In the international literature the median age at MM diagnosis is 69 years.16 In Brazil, the median age at diagnosis is 60.5 years.17 In several studies, the median age at MM diagnosis in patients with CNS involvement is lower. For Chen et al.,6 the median age was 53 years. In other studies,4,12,13 the median age at MM diagnosis was 54 years and for Niewenhuizen et al.,3 it was 56.4 years. In the present study, the median age at MM diagnosis was 56.1 years, corroborating the findings reported in the literature and suggesting that young patients with myeloma have a higher chance of CNS involvement.
As seen in other studies,3,6,11–13,18 the neurological symptoms documented in this study were heterogeneous, with headache being the most common (50%) followed by visual hypoacuity (30%). These symptoms are not exclusive to CNS infiltration by MM and can be found in other disease-related situations such as hypercalcemia, uremia and hyperviscosity syndrome.10 Consequently, it is important to investigate possible neurological symptoms as signs of CNS infiltration and hence conduct imaging (CT and/or MRI of the skull) and CSF testing.
The exact etiology of CNS infiltration is unknown. There are several hypotheses about the origin of CNS involvement. Studies found evidence of the contiguous dissemination of lytic lesions to the skull, bones of the skull base and nasopharyngeal mucosa.8,19 Another hypothesis is the hematogenic dissemination of plasma cells in plasma cell leukemia or by the progenitor lymphocytes of MM plasma cells.4,5,8
In this cohort, the CSF of one of the cases of leptomeningeal infiltration during disease progression contained 1% of plasma cells at the diagnosis of CNS involvement; in addition, the peripheral blood of this patient exhibited 1.6% circulating plasma cells at immunophenotyping. This patient had extramedullary plasmacytoma (hepatic, abdominal lymph node and ureteral lesion) at MM diagnosis, thus demonstrating a possible association between circulating plasma cells and plasma cell leukemia with CNS infiltration. In a review study, circulating plasma cells were found in 31% and plasma cell leukemia in 19% of 54 patients.5 In a case–control study, circulating plasma cells were found in 91% of patients with CNS involvement as compared to 63% of the group control. In the same study, the incidence of plasma cell leukemia was 10% versus 5%.20 Hence, a higher prevalence of leptomeningeal infiltration was found in patients with circulating plasma cells and plasma cell leukemia, which may corroborate the hematogenic dissemination hypothesis. Likewise, 17 patients in the current cohort exclusively had osteodural lesions, which also confirms the dissemination hypothesis due to bone contiguity. Two patients exhibited, in addition to osteodural involvement, leptomeningeal infiltration. The etiology of this situation is probably a leptomeningeal infiltration due to the contiguity of the bone lesion to the skull. Of the 35 patients in the study by Abdallah et al.,18 three (8.8%) patients also had leptomeningeal involvement adjacent to the osteodural lesion.
The optimal treatment for CNS infiltration is not defined. This is because it is difficult to define the best evidence-based treatment, as CNS involvement is an exclusion criterion in clinical studies. Additionally, because of the small number of cases, it is also difficult to conduct prospective controlled studies. Systemic chemotherapy, associated with intrathecal chemotherapy and/or radiotherapy, has been the main treatment in this situation. The latter has been reported in the past as the best treatment for CNS involvement,8 but it is currently indicated in association with other treatments.20 Intrathecal chemotherapy may cause plasma cells to disappear from the CSF.6 The systemic treatment aims to control the systemic and CNS disease by crossing the blood-brain barrier.6
In this study, the median OS after the diagnosis of CNS involvement was 40.3 months. In the studies published so far, the median OS varies from 1.5 months5 to 6.7 months,11 thus being lower than the OS found here.
However, when the OS of patients was evaluated according to the location of the infiltration, a median OS of 5.8 months was found for the group with leptomeningeal involvement, whereas the median OS was not reached for the group with osteodural involvement. These data agree with those obtained by the GIMEMA, which showed a difference in the median OS of patients with leptomeningeal and osteodural involvement of six and 25 months, respectively.8
In analyzing the OS of patients with CNS involvement at MM diagnosis or as the disease progressed, this study found that the median OS in the first group was not reached, whereas, in the second group, it was 7.4 months. When the OS of both groups (at diagnosis and during progression) were compared, a statistically significant difference was found between the groups (p-value=0.0069). One explanation for this is that patients with CNS infiltration at MM diagnosis have a greater OS than those who have it as the disease relapsed, since the disease is usually more aggressive and responds less to treatment during relapse. In their study, Chen et al.6 identified this difference with OS of 9.9 months versus 4.1 months (p-value=0.01) between patients with CNS involvement at MM diagnosis and during relapse.
Fassas et al.4 identified that there is possibly an association between increased LDH and CNS involvement in patients with MM. They observed that one-third of patients had high LDH at the time CNS infiltration was diagnosed. In another study, 68.6% of patients had increased LDH values as CNS involvement was diagnosed.18 In the current study, only 38.9% of patients had high LDH values when CNS involvement was diagnosed.
Furthermore, in this cohort, the median OS of patients who had high LDH values when CNS involvement was diagnosed (seven patients) was 13.05 months and was not reached in the normal LDH group. A comparison between the groups using the log-rank test showed a statistically significance difference (HR=0.082; 95% CI: 0.014–0.478; p-value=0.0033). This finding is in disagreement with the finding in the review study with 109 patients,3 which found no correlation between LDH and survival. In the multicenter study of 172 patients, LDH was statistically significant in the univariate analysis; however, a multivariate analysis was not performed due to insufficient information regarding the sample.11 Consequently, it was impossible to compare with the current study. Even though the studies published so far did not compare high LDH values found at the time of the diagnosis of CNS infiltration with survival, this study identifies LDH as a possible prognostic factor in patients with CNS involvement.
ConclusionCNS infiltration is a rare condition that may occur at MM diagnosis or during its relapse and it may even occur after a CR has been achieved. The best treatment regimen for this condition remains unknown and the prognosis is unfavorable in most cases.
Most of the patients in this study, unlike in published studies, had infiltration at MM diagnosis with a median OS above that found in the international literature.
Further studies are required to obtain additional information on the clinical and biological behavior and the best treatment for these patients.
Conflicts of interestThe authors declare no conflicts of interest.