We analyzed the management and outcomes of pregnancies of patients with chronic myeloid leukemia at a single center over fifteen years. Among the 203 CML female patients, there were ten pregnancies in seven women, all of them not planned. In three cases, the chronic myeloid leukemia diagnosis was made during pregnancy. Five patients received tyrosine kinase inhibitors in the first weeks of pregnancy and the drug was interrupted until delivery. One patient lost complete cytogenetic response, and two patients lost the hematological response. A patient with a stable major molecular response had two successful pregnancies without loss of response. There were four premature births. There were no maternal adverse events, fetal malformation or death. All patients received Interferon-alpha during gestation, and two received hydroxyurea for a short period. Leukapheresis was performed in two patients for hyperleukocytosis control. One patient with sickle cell disease died from disease progression six months after delivery.
ConclusionsThe tyrosine kinase inhibitors ministration should be interrupted during pregnancy. Patients should be advised to achieve a stable and deep molecular response if they plan to conceive, to avoid the risk of disease progression.
Chronic myeloid leukemia (CML) is a chronic myeloproliferative neoplasm characterized by a reciprocal translocation between the long arms of chromosomes 9:22 t(9:22)(q34,q11), which results in the BCR-ABL fusion gene that encodes a protein with tyrosine kinase activity. Today, the standard of care for this condition is targeted therapy with tyrosine kinase inhibitors (TKIs).1 CML may occur in women in their fertile age, meaning that pregnancy may occur at diagnosis or during the CML treatment.2,3 Rarely, the diagnosis of CML may occur during pregnancy. The management of this situation is challenging, due to the potential adverse effects of TKIs in the mother and the fetus,4 such as increased risk of placental failure, low weight of the newborn (NB), increased prematurity rate, perinatal morbidity and mortality.5 The TKIs are potentially teratogenic.3,4,6 Therefore, they are not recommended during pregnancy. Little is known about their potential toxicity to human embryos,7 but there are reports of cases that have been successfully treated with TKIs.7,8
ObjectivesThis study aimed to analyze all cases of pregnancy in patients with CML at a single center.
MethodsFrom January 2000 to June 2016, we analyzed all cases of pregnancy in patients with CML. At our center, patients are advised to use adequate contraception methods during treatment with TKIs. Data were collected from medical records and prenatal care: age, disease phase at diagnosis and at start of pregnancy, Sokal and Hasford score, treatments for CML before, during and after pregnancy, adverse events, responses at the onset of pregnancy, evolution of disease during pregnancy, type of delivery and complications during or after birth. The Local Research Ethics Committee approved the project, and all patients signed informed consent. Definitions for the classification of the deliveries were: early term: 37 0/7 weeks through 38 6/7 weeks of gestation, full term: 39 0/7 weeks through 40 6/7 weeks, late term: 41 0/7 weeks through 41 6/7 weeks; post-term: 42 0/7 weeks of gestation and beyond.9
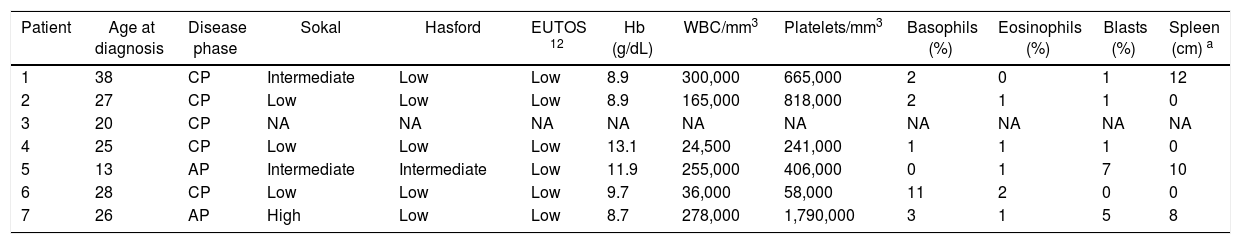
ResultsBetween January 2000 and August 2016, we treated 497 patients (including 203 females) with CML at our center. There were ten pregnancies in 7 women. Pregnant patients had a median age of 29 years (13–38 years) at diagnosis, five were in the chronic phase (CP) and two in the accelerated phase (AP). Clinical and laboratory data at diagnosis are described in Table 1. Data from diagnosis was not available for one patient (patient 2), who had started treatment at another hospital. In 3 patients (1, 2 and 7), CML was diagnosed during pregnancy. All patients were Ph-positive, without any additional abnormality and presented the p210 BCR-ABL transcript. All pregnancies were not planned and TKIs were interrupted after diagnosis of the pregnancy. Five patients received TKIs between the 6th and 21st week of pregnancy.
Clinical and laboratory characteristics of patients with CML at diagnosis (n=7).
| Patient | Age at diagnosis | Disease phase | Sokal | Hasford | EUTOS 12 | Hb (g/dL) | WBC/mm3 | Platelets/mm3 | Basophils (%) | Eosinophils (%) | Blasts (%) | Spleen (cm) a |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 38 | CP | Intermediate | Low | Low | 8.9 | 300,000 | 665,000 | 2 | 0 | 1 | 12 |
| 2 | 27 | CP | Low | Low | Low | 8.9 | 165,000 | 818,000 | 2 | 1 | 1 | 0 |
| 3 | 20 | CP | NA | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| 4 | 25 | CP | Low | Low | Low | 13.1 | 24,500 | 241,000 | 1 | 1 | 1 | 0 |
| 5 | 13 | AP | Intermediate | Intermediate | Low | 11.9 | 255,000 | 406,000 | 0 | 1 | 7 | 10 |
| 6 | 28 | CP | Low | Low | Low | 9.7 | 36,000 | 58,000 | 11 | 2 | 0 | 0 |
| 7 | 26 | AP | High | Low | Low | 8.7 | 278,000 | 1,790,000 | 3 | 1 | 5 | 8 |
CP: chronic phase; AP: accelerated phase. NA: not available. WBC: white blood cells.
The CML treatments during and after pregnancy are described in Table 2.
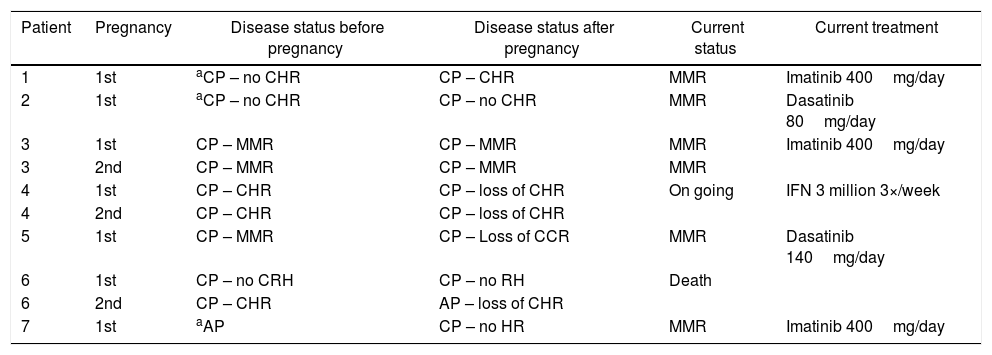
Response to CML treatment before and after pregnancy and the current status of CML patients.
| Patient | Pregnancy | Disease status before pregnancy | Disease status after pregnancy | Current status | Current treatment |
|---|---|---|---|---|---|
| 1 | 1st | aCP – no CHR | CP – CHR | MMR | Imatinib 400mg/day |
| 2 | 1st | aCP – no CHR | CP – no CHR | MMR | Dasatinib 80mg/day |
| 3 | 1st | CP – MMR | CP – MMR | MMR | Imatinib 400mg/day |
| 3 | 2nd | CP – MMR | CP – MMR | MMR | |
| 4 | 1st | CP – CHR | CP – loss of CHR | On going | IFN 3 million 3×/week |
| 4 | 2nd | CP – CHR | CP – loss of CHR | ||
| 5 | 1st | CP – MMR | CP – Loss of CCR | MMR | Dasatinib 140mg/day |
| 6 | 1st | CP – no CRH | CP – no RH | Death | |
| 6 | 2nd | CP – CHR | AP – loss of CHR | ||
| 7 | 1st | aAP | CP – no HR | MMR | Imatinib 400mg/day |
After pregnancy was detected, TKIs were interrupted and all patients received interferon (IFN). Three patients reported severe headache, myalgia and fatigue. One also reported nausea and vomiting grade 2.
Leukapheresis was performed in patients 1 and 7 due to hyperleukocytosis. Patients 1 and 6 received Hydroxyurea. Patient 1 used Hydrea in the 23rd week of pregnancy, at doses of 2.5g/day for five days and then reduced to 1g/day for seven days, associated with leukapheresis due to hyperleukocytosis. Patient 6 also had the diagnosis of sickle cell disease (SS and heterozygous for α-thalassemia) and had two pregnancies. She used Hydrea in the second pregnancy due to recurring painful crisis, 1–1.5g/day, from the 24th to 34th week of pregnancy. This patient also was submitted to erythrocytapheresis in both pregnancies to lower sickle hemoglobin (HbS) before delivery. Data are summarized in Table 3.
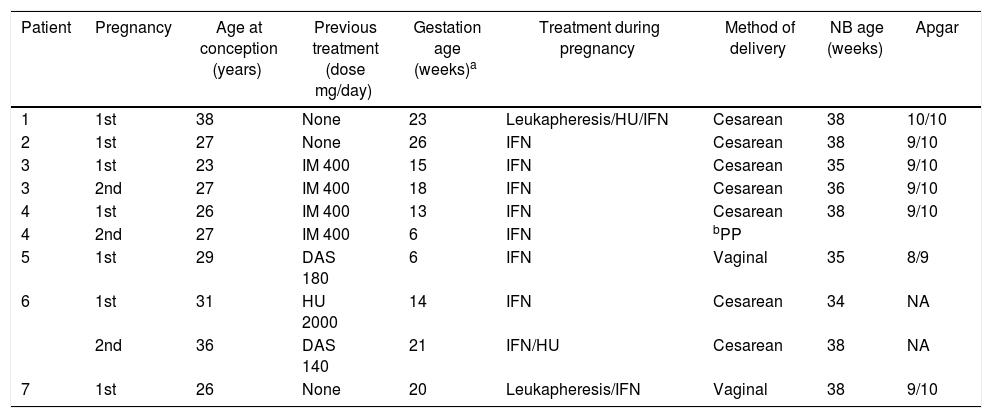
Clinical features and treatment of CML patients during pregnancy, delivery and newborn data.
| Patient | Pregnancy | Age at conception (years) | Previous treatment (dose mg/day) | Gestation age (weeks)a | Treatment during pregnancy | Method of delivery | NB age (weeks) | Apgar |
|---|---|---|---|---|---|---|---|---|
| 1 | 1st | 38 | None | 23 | Leukapheresis/HU/IFN | Cesarean | 38 | 10/10 |
| 2 | 1st | 27 | None | 26 | IFN | Cesarean | 38 | 9/10 |
| 3 | 1st | 23 | IM 400 | 15 | IFN | Cesarean | 35 | 9/10 |
| 3 | 2nd | 27 | IM 400 | 18 | IFN | Cesarean | 36 | 9/10 |
| 4 | 1st | 26 | IM 400 | 13 | IFN | Cesarean | 38 | 9/10 |
| 4 | 2nd | 27 | IM 400 | 6 | IFN | bPP | ||
| 5 | 1st | 29 | DAS 180 | 6 | IFN | Vaginal | 35 | 8/9 |
| 6 | 1st | 31 | HU 2000 | 14 | IFN | Cesarean | 34 | NA |
| 2nd | 36 | DAS 140 | 21 | IFN/HU | Cesarean | 38 | NA | |
| 7 | 1st | 26 | None | 20 | Leukapheresis/IFN | Vaginal | 38 | 9/10 |
Interferon: IFN; IM: imatinib; DAS: dasatinib.
All newborns were classified as early term (34–38 weeks); 7 were born from cesarean sections and 2 from vaginal deliveries. There were no maternal complications during pregnancy and childbirth and no cases of malformation or fetal death. Characteristics of the NB are listed in Table 3.
CML responses before and after deliveryPatients 1 and 7 started imatinib after delivery and achieved major molecular response (MMR). Patient 2 started imatinib treatment after delivery and later switched therapy to nilotinib due to imatinib resistance (loss of complete cytogenetic response (CCR)). She achieved a deep molecular response and participated in a discontinuation trial, but lost MMR six months after nilotinib discontinuation. After nilotinib reintroduction, she had cardiac toxicity and is currently in treatment with dasatinib, with MMR. Patient 3 had achieved MMR before pregnancy, with imatinib 400mg/day and kept MMR after imatinib interruption. Imatinib was reintroduced after each delivery. Patient 4 had a short period of imatinib treatment in the first pregnancy and lost hematologic response (HR) after discontinuation of the drug. Imatinib was introduced after delivery and the patient achieved complete hematologic response (CHR), but treatment was interrupted again due to a second pregnancy, with loss of CHR and treated with IFN. Patient 5 was receiving dasatinib 140mg/day, which was interrupted during pregnancy. She lost MMR and CCR after dasatinib interruption, but subsequently recovered both responses with the reintroduction after delivery. Patient 6 also had sickle cell disease and had two pregnancies. CML was detected during the first pregnancy, while she was using hydroxyurea (HU) for sickle cell disease treatment. In the second pregnancy, she was using dasatinib in a clinical trial (CA-180-035). After dasatinib discontinuation, she lost the hematologic response and was treated with HU from week 24 until delivery. This patient had disease progression to accelerated phase and died seven months after delivery, even with the dasatinib reintroduction.
DiscussionThe presence of pregnancy during the CML is a rare condition5 since the disease is more common between 40 and 60 years,2 and female patients are advised not to conceive while on TKI treatment. Reports are showing the increase in prevalence during the reproductive phase.10 In our series, we observed ten pregnancies in seven patients in fifteen years, all of them not planned, resulting in exposure to TKIs in the first weeks. Besides this short exposure to TKIs in the first trimester, there were no congenital malformations. The TKI was discontinued according to literature recommendation, to avoid the risk of teratogenic effects, as reported in animals.3,4,6,8 Some reports have shown favorable outcomes in patients who were treated with TKIs during pregnancy.13–16 Additionally, we did not observe any abnormality in the newborns exposed to HU during pregnancy.
Leukapheresis was used in two patients with newly diagnosed CML and was well tolerated. This therapy is described as safe in this population, but should be performed in individual cases of hyperleukocytosis.17
All patients used Interferon-alpha as standard therapy during pregnancy, doses from 3 million U/day three times a week to 9 million/day, which was well tolerated, although two patients had headache reports. However, the adverse effects were not significant for drug discontinuation. Five out of nine deliveries were preterm, but no malformation was reported, which corroborates the literature reports on the safety of the drug in pregnancy.14,15 Adverse-event profiles were similar among those described in the literature.13
We observed in our series that the TKI interruption in patients with stable MMR did not affect the patient's outcome. A recent review reported that TKI discontinuation among patients who have achieved very deep and prolonged molecular remission is safe, and that TKIs may be reintroduced after delivery in case of a loss of MMR.17
On the other hand, early discontinuation of imatinib, without achieving a stable response, increases the risk of loss of hematological response and progression, as described in patients 4 and 6. Patient 6 was resistant to imatinib and was receiving dasatinib as second-line therapy, with CHR as the best response, in 7 years of TKI treatment. She progressed to the accelerated phase and died seven months after the last delivery. In this particular case, the presence of the sickle cell disease corroborated a worse prognosis and a poor outcome. Pregnancy itself did not prove to be an aggravating factor of the disease, as suggested in the literature.6
ConclusionHydroxyurea, interferon and leukapheresis are safe options for patients with hyperleukocytosis. The use of TKIs is not recommended in pregnancy because of the risk of teratogenicity and should be started as soon as possible after delivery. In some cases, TKIs can be used during pregnancy, depending on the risk of the disease. Patients who wish to conceive should be advised to interrupt TKIs, with the recommendation that they achieve a stable and deep molecular response or at least a stable MMR before discontinuation. Those patients have less risk of MMR loss or disease progression after TKI discontinuation.
Conflicts of interestKatia B Pagnano: consultancy: Novartis. The other authors have no conflict to declare.