This study investigated the influence of two conditioning regimens on the chimerical status of 104 patients with acquired severe aplastic anemia.
MethodsPatients were monitored for at least 18 months after related bone marrow transplantation and reaching partial or complete hematologic recovery. Group I patients (n=55) received 200mg/kg cyclophosphamide alone and Group II (n=49) received 120mg/kg cyclophosphamide associated with 12mg/kg busulfan. Patients were classified in three chimerism levels according to the percentage of donor cells in the peripheral blood.
ResultsChimerism ≤50% occurred in 36.4% of Group I and none of Group II; chimerism 51–90% was found in 20.0% of Group I and 10.2% of Group II; and chimerism >90% was found in 43.6% of Group I versus 89.8% of Group II. A significant association (p-value<0.001) was found between conditioning type and chimerism levels. A higher number of infused cells was associated with higher levels of chimerism only in Group I (p-value=0.013). Multivariate analysis showed that chimerism >90% is associated with the cyclophosphamide plus busulfan conditioning (p-value<0.001) and higher number of infused cells (p-value=0.009), suggesting that these factors are predictive of graft outcome. Regarding hematological recovery, higher chimerism levels were associated with higher neutrophil (p-value=0.003) and platelet counts (p-value<0.001) in Group I only. These results show that myeloablative conditioning favors full donor chimerism and non-myeloablative conditioning predisposes to mixed chimerism or autologous recovery of hematopoiesis.
ConclusionThese data show that autologous recovery depends on the intensity of immunosuppression and that the immunosuppressive function of cyclophosphamide alone can induce this type of hematopoietic recovery.
Hematopoietic stem cell transplantation (HSCT) is the treatment of choice for young patients with severe aplastic anemia (SAA).1 An intensity of conditioning regimen that achieves immunosuppressive effects to avoid graft rejection is a crucial factor in the allogeneic bone marrow transplantation (BMT) setting for SAA patients. The standard conditioning for patients who have an human leukocyte antigen (HLA)-matched sibling donor comprises high doses of cyclophosphamide (CY – 200mg/kg) with or without anti-thymocyte globulin (ATG).2
Until the early 90s, SAA patients transplanted at Hospital de Clínicas (HC) of the Universidade Federal do Paraná (UFPR) received non-myeloablative conditioning such as 200mg/kg of body weight of CY. Subsequently, it was observed that patients who received more than 15 transfusions prior to transplant had higher rejection rates and lower survival compared to those receiving fewer transfusions (unpublished data). Piccin et al.3 also found a significant difference in the survival rates of patients with SAA or Fanconi Anemia who received more than 20 units of red blood cells and/or platelets. For this reason and because of the irregular supply of ATG, from 1993 all polytransfused SAA patients received CY (120mg/kg) associated with busulfan (BUS – 12mg/kg of body weight). The CY+BUS combination showed a marked reduction in rejection rates.4
Transplant outcome is assessed by hematologic recovery and chimerism analysis that provide information on the hematopoietic reconstitution of patients, which can be autologous, allogeneic or chimerical. The determination of chimerism status has been based on the amplification of markers with a variable number of tandem repeats (VNTRs) or short tandem repeats (STRs) scattered throughout the genome.5–7
This retrospective study analyzed the levels of chimerism in two groups of patients with acquired SAA who received different conditioning regimens before undergoing BMT with HLA-identical sibling donors. The goal was to investigate the association between chimerism levels with different conditioning as well as with other variables in order to identify factors that could predict the outcome of the allogeneic hematopoietic stem cell graft. Among the study variables were pre-transplant characteristics such as patient's age and gender, time between diagnosis and BMT, and transplant characteristics such as donor's age and gender and number of infused cells. The chimerism status of patients with normal or near normal hematological levels was also analyzed. Peripheral blood hematimetric levels, such as the neutrophil and platelet counts, and hemoglobin concentration were evaluated in the period equal to or greater than 18 months after transplantation. Patients with time equal to or greater than 18 months post-transplant were included because they had already reached stable chimerism and because hematological changes rarely occur after this time.
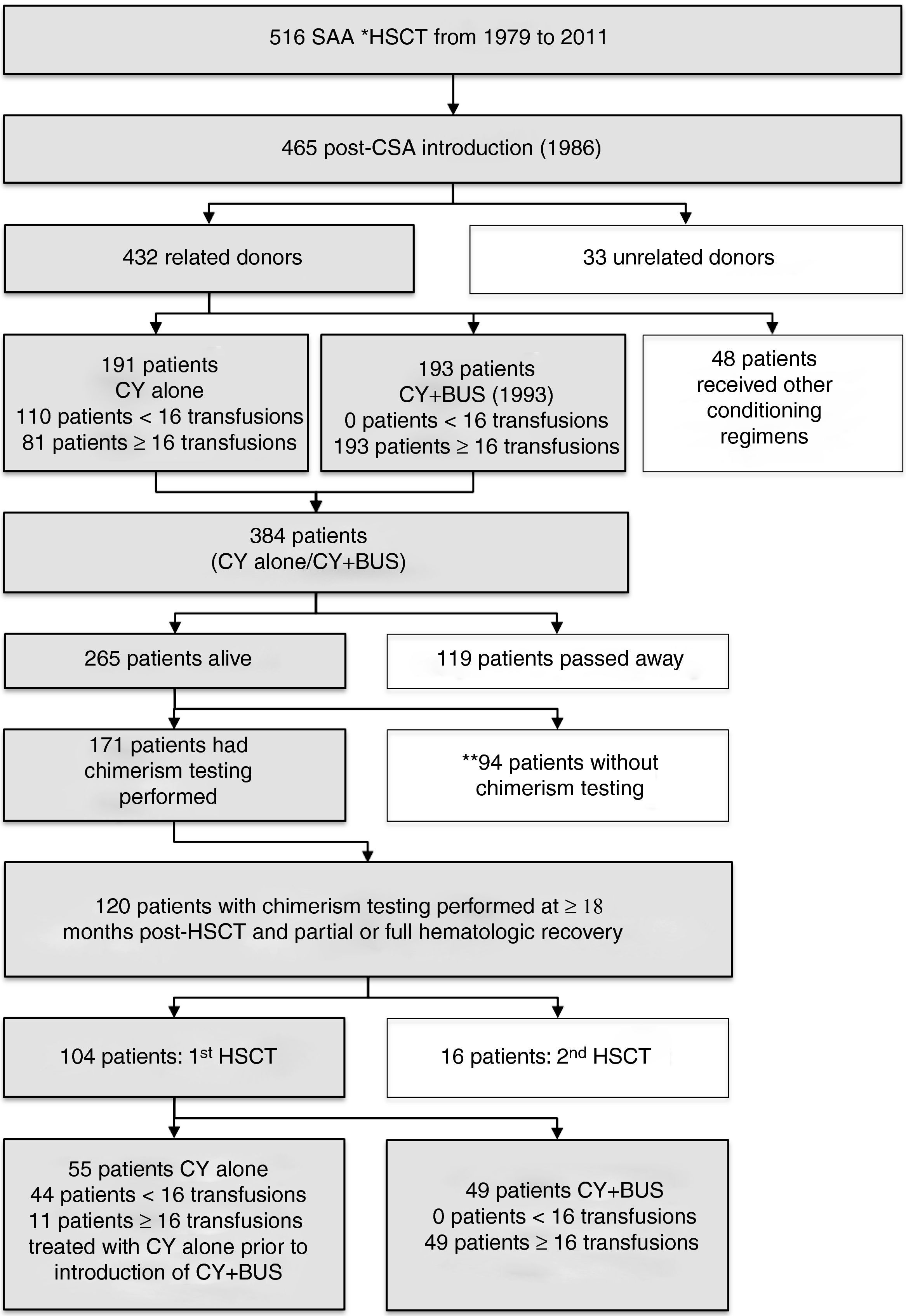
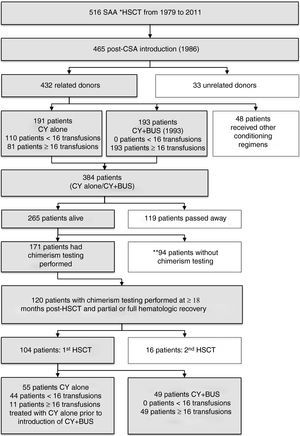
MethodsPatientsThe Bone Marrow Transplantation Service at HC-UFPR started its activities in 1979, and from that time up to October 2011, this center has performed 516 transplants in SAA patients. This study involved 104 SAA patients who underwent the first transplant at HC-UFPR between August 1987 and July 2009. Data on chimerism levels were collected up to April 2011. The inclusion criteria for the patients analyzed in this cohort were a diagnosis of SAA, first BMT with a related donor, alive at the time of this study, chimerism analysis performed at a time ≥18 months after the transplant, partial or complete hematologic recovery, and conditioned with CY alone or with CY+BUS (Figure 1). Pre-transplant characteristics of these patients are shown in Table 1. The characteristics of patients who passed away (not included in this study) are described in Table 2.
Patients who underwent hematopoietic stem cell transplantation at Hospital de Clinicas, Universidade Federal do Parana, Curitiba, Brazil from 1979 to 2011. *HSCT: hematopoietic stem cell transplantation. **Ninety-four patients did not have variable number of tandem repeat (VNTR)/short tandem repeat (STR) chimerism results either because this test was not performed in our center at the time they were transplanted or due to no show to their follow up medical appointment.
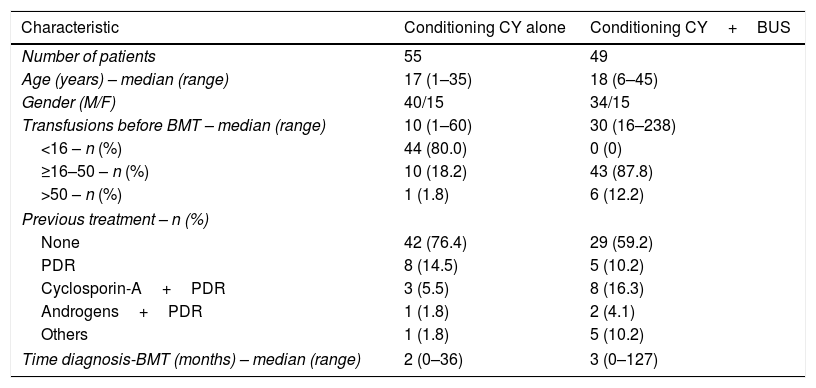
Pre-transplant characteristics.
| Characteristic | Conditioning CY alone | Conditioning CY+BUS |
|---|---|---|
| Number of patients | 55 | 49 |
| Age (years) – median (range) | 17 (1–35) | 18 (6–45) |
| Gender (M/F) | 40/15 | 34/15 |
| Transfusions before BMT – median (range) | 10 (1–60) | 30 (16–238) |
| <16 – n (%) | 44 (80.0) | 0 (0) |
| ≥16–50 – n (%) | 10 (18.2) | 43 (87.8) |
| >50 – n (%) | 1 (1.8) | 6 (12.2) |
| Previous treatment – n (%) | ||
| None | 42 (76.4) | 29 (59.2) |
| PDR | 8 (14.5) | 5 (10.2) |
| Cyclosporin-A+PDR | 3 (5.5) | 8 (16.3) |
| Androgens+PDR | 1 (1.8) | 2 (4.1) |
| Others | 1 (1.8) | 5 (10.2) |
| Time diagnosis-BMT (months) – median (range) | 2 (0–36) | 3 (0–127) |
CY: cyclophosphamide; BUS: busulfan; M: male; F: female; BMT: bone marrow transplant; PDR: prednisone.
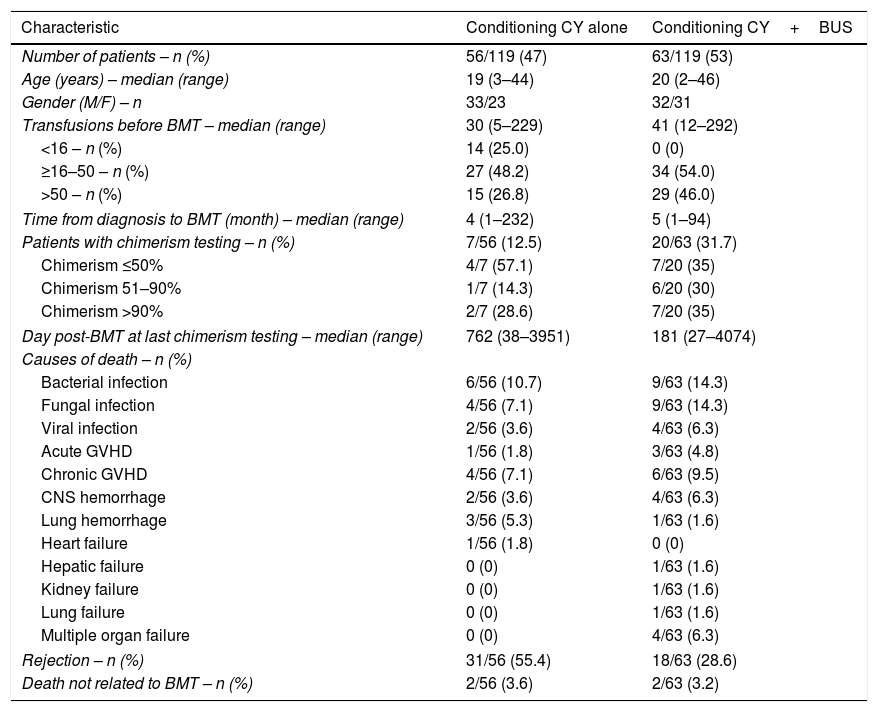
Characteristics of patients not included due to death at the time of data collection for this study.
| Characteristic | Conditioning CY alone | Conditioning CY+BUS |
|---|---|---|
| Number of patients – n (%) | 56/119 (47) | 63/119 (53) |
| Age (years) – median (range) | 19 (3–44) | 20 (2–46) |
| Gender (M/F) – n | 33/23 | 32/31 |
| Transfusions before BMT – median (range) | 30 (5–229) | 41 (12–292) |
| <16 – n (%) | 14 (25.0) | 0 (0) |
| ≥16–50 – n (%) | 27 (48.2) | 34 (54.0) |
| >50 – n (%) | 15 (26.8) | 29 (46.0) |
| Time from diagnosis to BMT (month) – median (range) | 4 (1–232) | 5 (1–94) |
| Patients with chimerism testing – n (%) | 7/56 (12.5) | 20/63 (31.7) |
| Chimerism ≤50% | 4/7 (57.1) | 7/20 (35) |
| Chimerism 51–90% | 1/7 (14.3) | 6/20 (30) |
| Chimerism >90% | 2/7 (28.6) | 7/20 (35) |
| Day post-BMT at last chimerism testing – median (range) | 762 (38–3951) | 181 (27–4074) |
| Causes of death – n (%) | ||
| Bacterial infection | 6/56 (10.7) | 9/63 (14.3) |
| Fungal infection | 4/56 (7.1) | 9/63 (14.3) |
| Viral infection | 2/56 (3.6) | 4/63 (6.3) |
| Acute GVHD | 1/56 (1.8) | 3/63 (4.8) |
| Chronic GVHD | 4/56 (7.1) | 6/63 (9.5) |
| CNS hemorrhage | 2/56 (3.6) | 4/63 (6.3) |
| Lung hemorrhage | 3/56 (5.3) | 1/63 (1.6) |
| Heart failure | 1/56 (1.8) | 0 (0) |
| Hepatic failure | 0 (0) | 1/63 (1.6) |
| Kidney failure | 0 (0) | 1/63 (1.6) |
| Lung failure | 0 (0) | 1/63 (1.6) |
| Multiple organ failure | 0 (0) | 4/63 (6.3) |
| Rejection – n (%) | 31/56 (55.4) | 18/63 (28.6) |
| Death not related to BMT – n (%) | 2/56 (3.6) | 2/63 (3.2) |
CY: cyclophosphamide; BUS: busulfan; M: male; F: female; BMT: bone marrow transplant; GVHD: graft-versus-host disease; CNS: central nervous system.
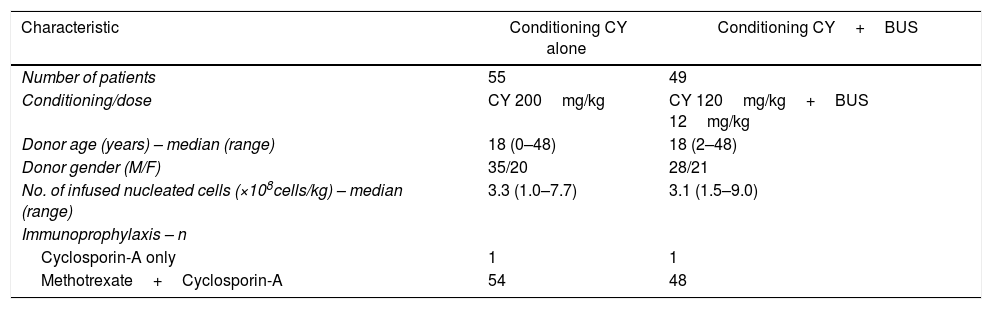
All patients who received bone marrow from HLA-identical siblings were divided into two groups according to the conditioning regimen. The choice of the conditioning protocol was based on the number of previous transfusions with this information being obtained from medical records as well as from reports from the patient or family members. Forty-four patients with less than 16 transfusions were treated with CY alone at 200mg/kg body weight in four doses and those with ≥16 transfusions (n=49), in general, received CY at 120mg/kg body weight divided in two doses associated with BUS at 12mg/kg of body weight in 16 oral doses.4 However, eleven patients who received ≥16 transfusions and were included in the CY alone group (n=44+11) because they were transplanted before the implementation of the CY+BUS protocol. The decision to combine CY+BUS was based on the experience of Lucarelli et al.8 who used this regimen to treat thalassemia patients, but the doses utilized in our center were modified. The transplant characteristics of this cohort are shown in Table 3.
Transplant characteristics.
| Characteristic | Conditioning CY alone | Conditioning CY+BUS |
|---|---|---|
| Number of patients | 55 | 49 |
| Conditioning/dose | CY 200mg/kg | CY 120mg/kg+BUS 12mg/kg |
| Donor age (years) – median (range) | 18 (0–48) | 18 (2–48) |
| Donor gender (M/F) | 35/20 | 28/21 |
| No. of infused nucleated cells (×108cells/kg) – median (range) | 3.3 (1.0–7.7) | 3.1 (1.5–9.0) |
| Immunoprophylaxis – n | ||
| Cyclosporin-A only | 1 | 1 |
| Methotrexate+Cyclosporin-A | 54 | 48 |
CY: cyclophosphamide; BUS: busulfan.
The evaluation of late chimerism status was based on the last test performed for each patient at a time ≥18 months with the date of post-BMT sample collection for testing varying from 18.8 to 187.8 months (median: 55.5 months) according to the clinical follow-up. The neutrophil and platelet counts as well as hemoglobin level were obtained from the last available blood count with the date also varying according to the clinical follow-up of each patient, but it was always ≥18 months after BMT (range: 21.4–285.1; median: 113 months).
Chimerism analysisPost-BMT chimerism analysis between August 1995 and July 2009 was performed by PCR amplification of VNTR/STR loci, and detection of amplified fragments was visualized by vertical discontinuous polyacrylamide gel electrophoresis stained with silver salts (n=84).9 However, from August 2009 onward, chimerism was evaluated by PCR amplification of STR loci using fluorescent labeled primers (Applied Biosystems Inc., USA) with the amplified fragments being identified by capillary electrophoresis in an ABI 3130 Genetic Analyzer (n=20). In order to minimize possible variations between these two methods, besides proposing an alternative approach, the results were stratified in chimerism levels of ≤50%, between 51% and 90% and >90% of donor cells for both conditioning groups.
DNA isolationGenomic DNA was isolated from peripheral blood nucleated cells in EDTA or from buccal cells collected in saline using a modified salting out method.10 Chimerism testing was performed with DNA from peripheral blood or buccal cells to obtain the constitutional pattern of patients from samples considered pre-transplant. Buccal cells were collected after transplant only when the patient's pre-transplant DNA samples were no longer available for chimerism testing. Post-transplant samples of patients and their donors were isolated from peripheral blood.
Polymerase chain reaction amplification of VNTR/STR loci and fragment detection in polyacrylamide gels stained with silver saltsVNTR (D1S80, 33.6) and STR (SE33, HumTH01, HumARA, HumTPO) loci of pre- and post-transplant samples from patients as well as donor samples were amplified by PCR.9 Specific primers for these loci were divided into two multiplex systems named triplex I and II.11 Amplifications were performed in ABI 9600 or 9700 thermal cyclers, using 28 cycles at 94°C for 30s, 62°C for 30s, followed by one cycle at 62°C for 20min and 4°C forever. Amplified samples were loaded into vertical discontinuous polyacrylamide gel and electrophoresed at 105V for 18h at room temperature.12 Fragments were detected by silver staining with the percentage of donor derived cells being inferred by visual observation of intensity of the electrophoretic bands for the informative loci.9
Polymerase chain reaction amplification of single tandem repeat loci using fluorescent-tagged primers and fragments detection by capillary electrophoresisPre- and post-transplant samples from patient as well as from donors were amplified by a multiplex system including 15 STR loci (D8S1179, D21S11, D7S820, CSF1PO, D3S1358, TH01, D13S317, D16S539, D2S1338, D19S433, VWA, TPOX, D18S51, D5S818 and FGA) and the amelogenin gene (Kit AmpFℓSTR Identifiler, ABI, USA). Amplifications were done in an ABI 9700 thermal cycler and fragments separated by capillary electrophoresis in an ABI 3130 Genetic Analyzer according to manufacturer's instructions. Data were analyzed using GeneMapper v. 4.0 software (ABI, USA). Calculation of percentage of donor derived cells in the post-transplant sample was based on the presence of homozygous or heterozygous peaks corresponding to alleles not shared by patient and donor.13,14
Statistical analysisThe results of the quantitative variables were described as medians, and minimum and maximum values, and qualitative variables by their frequencies and percentages. Comparisons of two groups of patients (chimerism 51–90% and >90% donor cells) in regard to age, interval between diagnosis and BMT, number of infused cells, neutrophil count, platelet count and hemoglobin level were performed using Student's t-test for independent samples or the nonparametric Mann–Whitney test. While comparisons of three groups of patients (chimerism levels ≤50%, 51–90% and >90%) were by the variance analysis model (ANOVA) with a factor or the nonparametric test of Kruskal–Wallis. The condition of normality of variables was evaluated using the Kolmogorov–Smirnov test. Association analysis between chimerism levels and gender was achieved by Fisher's exact test for two chimerism levels and the chi-square test for three levels; associations between recipient/donor sex matching and three chimerism levels were investigated by the chi-square test. Multivariate analysis included variables with p-values <0.25 in the univariate analysis, and employed an adjusted logistic regression model. The significance of variables was assessed by the Wald test. p-Values <0.05 were considered statistically significant. Data were analyzed using Statistica v.10.0 software (Stat Soft, Oklahoma, USA).
ResultsUnivariate analysisConditioning regimen and chimerismThe percentage of donor derived cells was >90% in 24 (43.6%), 51–90% in 11 (20.0%) and ≤50% in 20 (36.4%) patients in the group treated with CY alone. While in the group that received CY plus BUS, 44 (89.8%) achieved chimerism >90% and five (10.2%) from 51–90% donor cells. These results showed a significant association between the conditioning regimen and chimerism level (p-value<0.001). The follow up of chimerism status showed that patients with ≤50% donor cells had progressive reductions of donor derived cells with predominance of their own cells, and they were considered as having autologous recovery (AR) in this study.
Pre-transplant characteristics and chimerismThere was no significant association between chimerism levels and patients’ age (p-value=0.241), gender (p-value=0.953) or interval between diagnosis and transplant (p-value=0.448) in the group conditioned with CY alone and none of these pre-transplant variables had any effect on chimerism levels in the CY+BUS Group (p-value=0.394, p-value=0.306 and p-value=0.211, respectively).
Transplant characteristics and chimerismOf all the transplant variables, only the number of infused cells had a significant association with the chimerism levels in the CY alone group, i.e., the higher the number of cells infused the greater the degree of chimerism (p-value=0.013). No association was seen between chimeric status and donor age (p-value=0.518) or gender (p-value=0.901). In the group conditioned with CY+BUS, no significant difference was found between chimerism levels (51–90% and >90% of donor cells) and donor age (p-value=0.219), gender (p-value=1.0) or number of infused cells (p-value=0.357). Patient and donor sex matching did not have an impact on chimerism levels when both conditioning groups were analyzed together (p-value=0.740; n=104) or on the CY alone group (p-value=0.938; n=55). The CY+BUS group was not analyzed separately due to small numbers of patients at the ≤50% and 51–90% chimerism levels in the four sex matching categories.
Post-transplant characteristics and chimerismPatients conditioned with CY alone who had higher chimerism levels showed higher neutrophil (p-value=0.003) and platelet counts (p-value<0.001) (Table 4). In the CY+BUS group, patients with chimerism >90% of donor cells had neutrophil (p-value=0.051) and platelet counts (p-value=0.051) slightly higher than those with chimerism between 51% and 90%. Hemoglobin was not associated with levels of chimerism in either of the conditioning groups.
Associations between chimerism levels and post-transplant variables in patients conditioned with CY alone.
| Variable | Chimerism | p-Value | ||
|---|---|---|---|---|
| ≤50% (n=20) | 51–90% (n=11) | >90% (n=24) | ||
| Neutrophil count (×103/μL) – median (range) | 2.2 (1.0–5.9) | 2.3 (1.5–6.4) | 3.3 (2.3–8.2) | 0.003b |
| Hemoglobin (g/dL) – median (range) | 15.1 (9.2–17.3) | 15.2 (11.7–17.0) | 15.1 (13.3–19.2) | 0.597a |
| Platelet count (×103/μL) – median (range) | 143 (12–249) | 163 (131–315) | 218 (131–315) | <0.001b |
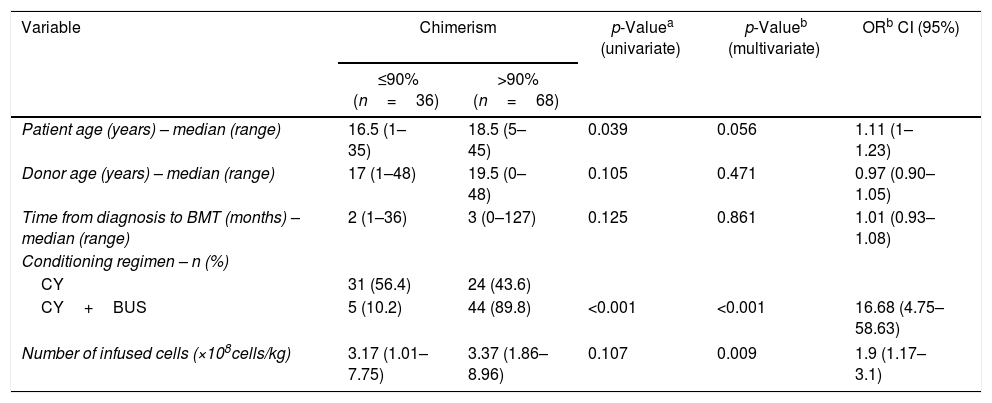
Pre-transplant and transplant variables included in the multivariate analysis were those previously associated with chimerism level >90% of donor cells in the univariate analysis (Table 5). Results revealed associations of the CY+BUS conditioning (p-value<0.001) as well as higher number of infused cells (p-value=0.009) with levels of chimerism >90%.
Influence of pre-transplant and transplant variables on the chimerism levels by multivariate analysis.
| Variable | Chimerism | p-Valuea (univariate) | p-Valueb (multivariate) | ORb CI (95%) | |
|---|---|---|---|---|---|
| ≤90% (n=36) | >90% (n=68) | ||||
| Patient age (years) – median (range) | 16.5 (1–35) | 18.5 (5–45) | 0.039 | 0.056 | 1.11 (1–1.23) |
| Donor age (years) – median (range) | 17 (1–48) | 19.5 (0–48) | 0.105 | 0.471 | 0.97 (0.90–1.05) |
| Time from diagnosis to BMT (months) – median (range) | 2 (1–36) | 3 (0–127) | 0.125 | 0.861 | 1.01 (0.93–1.08) |
| Conditioning regimen – n (%) | |||||
| CY | 31 (56.4) | 24 (43.6) | |||
| CY+BUS | 5 (10.2) | 44 (89.8) | <0.001 | <0.001 | 16.68 (4.75–58.63) |
| Number of infused cells (×108cells/kg) | 3.17 (1.01–7.75) | 3.37 (1.86–8.96) | 0.107 | 0.009 | 1.9 (1.17–3.1) |
CI: confidence interval; OR: odds ratio; BMT: bone marrow transplant; CY: cyclophosphamide; BUS: busulfan.
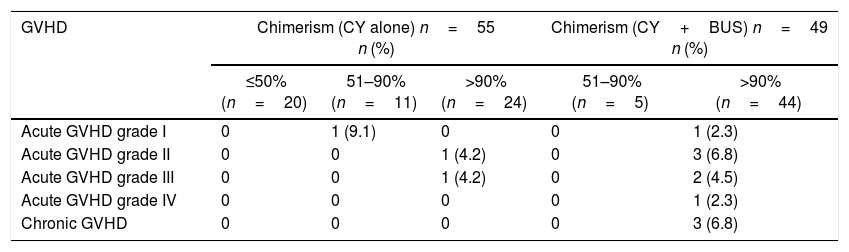
In this study, 5.4% (3/55) of patients in the CY alone group and 14.3% (7/49) in the CY+BUS Group developed acute graft-versus-host disease (GVHD). Chronic GVHD was present in 6.1% (3/49) of patients conditioned with CY+BUS and in none of the patients who received CY alone (Table 6).
GVHD in patients conditioned with CY alone and CY+BUS.
| GVHD | Chimerism (CY alone) n=55 n (%) | Chimerism (CY+BUS) n=49 n (%) | |||
|---|---|---|---|---|---|
| ≤50% (n=20) | 51–90% (n=11) | >90% (n=24) | 51–90% (n=5) | >90% (n=44) | |
| Acute GVHD grade I | 0 | 1 (9.1) | 0 | 0 | 1 (2.3) |
| Acute GVHD grade II | 0 | 0 | 1 (4.2) | 0 | 3 (6.8) |
| Acute GVHD grade III | 0 | 0 | 1 (4.2) | 0 | 2 (4.5) |
| Acute GVHD grade IV | 0 | 0 | 0 | 0 | 1 (2.3) |
| Chronic GVHD | 0 | 0 | 0 | 0 | 3 (6.8) |
GVHD: graft-versus-host disease; CY: cyclophosphamide; BUS: busulfan.
This retrospective study investigated the influence of two different conditioning regimens on the chimeric status of SAA patients who received bone marrow grafts from HLA-identical siblings. Chimerism tests were performed on samples collected at ≥18 months post-transplant. Results showed that the type of conditioning influenced the level of chimerism since nearly 90% of patients treated with CY+BUS reached levels >90% of donor cells, while only 44% of those who received CY alone reached this level of chimerism. Despite the observation by Lawler et al.15 of a higher frequency of full donor chimerism in patients conditioned with CY alone compared to other regimens, this association was very weak.
Variations in chimerism levels after HSCT may reflect differences related to the sensitivity of the methods and the type of conditioning. Results of this study are similar to others that also reported presence of higher percentages of donor cells in myeloablative regimens.16,17 Frequencies of chimerism between 51% and 90% donor cells were twice as high in patients who received CY alone (20%) compared to those treated with CY+BUS (10%). Our result is similar to Gomez et al.,18 Jillella et al.19 and Shamshad20 who also found higher frequencies of mixed chimerism in patients with non-myeloablative conditioning.
The frequency of AR in the current study was much higher than that reported in the literature. A multicenter analysis of SAA patients by Piccin et al.21 and a study by Pieroni et al.22 found AR frequencies of 4.2% and 7.0%, respectively. These lower frequencies could be due to different conditioning regimens and/or different methods used to analyze chimerism levels. Piccin et al.21 also showed that AR patients have a better prognosis than those with successful engraftment and significantly better than patients with graft failure. The major concern with AR is related to the susceptibility of autologous cells to clonal evolution that may result in difficult-to-manage hematological malignancies. The absence of AR in the CY+BUS group is consistent with results of previous studies18,19 and emphasizes that myeloablative conditioning commonly eradicates the patient's cells providing full donor chimerism. Piccin et al.21 found that the time interval between diagnosis and transplant was longer in the AR group compared to patients with stable engraftment but this was not observed in the present study. It is important to point out that all patients evaluated for chimerism status were alive at the time of investigation and the occurrence of late rejection or death related to transplantation were extremely rare events 18 months after transplant.
Pre-transplant characteristics such as patient age and sex did not influence chimerism levels in either of the conditioning groups. This result is similar to those found by Lawler et al.15 and Piccin et al.,21 although their analyses were not done separately according to the conditioning regimen.
Gorin et al.23 showed that infusions of greater number of cells remains an important factor in graft outcome and they point out that every effort must be made to achieve cell numbers >2.5×108kg−1. In the current study, the number of infused cells was associated with chimerism levels only in the CY alone group, where those who received fewer cells had chimerism ≤50%. Multivariate analysis revealed an association between higher numbers of infused cells and chimerism >90%, independently of the conditioning regimen. Other transplant features such as donor age and sex did not influence chimerism levels, with similar results being reported by Lawler et al.15
Post-transplant outcome was assessed by hematological recovery, based on the latest available blood count after 18 months of HSCT. Positive associations between higher neutrophil and platelet counts with greater chimerism levels were observed only in the CY alone group. Hematological recovery data in the literature refer mostly to the period immediately following transplantation; studies investigating associations of hematological parameters with long-term chimerism were not found.
Data from this study showed a higher frequency of acute (7/44=15.9%) and chronic (3/44=6.8%) GVHD in patients conditioned with CY+BUS who had chimerism >90% compared to those conditioned with CY alone whose frequencies of acute and chronic GVHD were 8.4% (2/24) and 0%, respectively. At HC-UFPR, it was observed that the rejection rate was close to 50% in patients who received ≥16 transfusions and were conditioned with CY alone. In order to reduce rejection the institution started to use conditioning with CY plus BUS. The association of these drugs reduced the rejection rate to approximately 15%. From 1993 onward, polytransfused patients (≥16 transfusions) who transplanted with a related donor received the CY plus BUS and no longer CY alone. The overall survival at ten years for patients with ≥16 transfusions who received CY alone was around 50%; this increased to approximately 70% with the CY+BUS combination (unpublished data).
Early referral of individuals diagnosed with SAA to specialized centers is crucial to reduce the number of transfusions as this is a risk factor for rejection and requires more aggressive conditioning regimens. On comparing polytransfused patients with those who received few transfusions, the rejection rate remains high for the latter group despite the use of less immunogenic blood products in recent years. This could be because new therapeutic approaches that are supposed to reduce rejection are not always available in small centers. Patients who are referred to reference centers have better chances of receiving this kind of treatment.
We acknowledge some limitations of this study. In addition to being retrospective, there is a lack of information about chimerism status on leukocyte subpopulations since the VNTR/STR assays were performed on whole blood. It is well known that post-transplant engraftment monitoring of cell subsets increases the sensitivity of the assay to detect donor cells.
ConclusionChimeric status of patients under long-term monitoring showed differences according to the intensity of conditioning, where the CY+BUS myeloablative regimen favors complete chimerism and CY alone the establishment of mixed chimerism. The observation that conditioning with CY alone favors AR in SAA patients corroborates the use of immunosuppressants to treat this disease, and encourages the search for novel agents with lower toxicity and higher efficacy that can regenerate bone marrow. Information about patient post-transplant chimeric status taken together with other criteria may contribute to timely therapeutic interventions in order to prevent graft rejection and to improve the hematopoietic function.
Conflicts of interestThe authors declare no conflicts of interest.
We wish to thank Roseli Rocio Silva for her technical assistance and the data manager Heliz Regina Neves.