Hodgkin's lymphoma (HL) is an uncommon neoplasm with an estimated number of 65,950 cases globally; its incidence varies significantly by age, sex, ethnicity, geographic location and socioeconomic status, being more frequent in young people. It is classified as either nodular lymphocyte predominant Hodgkin's lymphoma (NLPHL) or classical Hodgkin's lymphoma (CHL).1 The CHL accounts for 95% of all HL cases and can be further subdivided into four histological subtypes.2
The biology of HL depends on the interaction of the few pathognomonic, morphologically abnormal germinal B giant cells (Reed-Sternberg cells), with the surrounding reactive infiltrate composed of T-cells, histiocytes, eosinophils, and plasma cells. The ability of Reed-Sternberg cells to survive during negative selection in the germinal center depends, partially, on deregulated NFkB expression, closely related to the involvement of the Epstein-Barr Virus (EBV).3 The increase in NFkB transcription factor levels enhances the expression of several cytokines, including interleukin 6 (IL-6).4,5 As such, increased IL-6 levels at diagnosis may then be associated with a worse prognosis6 and with the presence of B symptoms and bulky disease.7
We evaluated all consecutive patients with CHL who were treated in the Department of Hematology at Hospital Mário Covas in Santo André, Brazil according to the WHO criteria to assess if IL-6 could relate to prognosis and should be studied as a prognostic marker, either at diagnosis or at mid-treatment evaluation (interim). Patients where included from March 2012 to July 2015, either at diagnosis or at relapse. Results of the interim PET scans obtained after two cycles of chemotherapy (I-PET2) were classified according to the criteria of Deauville.8 For our study, we considered a negative PET as 0–3 and positive as 4–5 on the Deauville scale. Peripheral blood samples from patients were taken before the administration of chemotherapy and before the fourth treatment cycle (interim). We stored samples in a freezer at −3°C and measured the IL-6 levels with a competitive chemiluminescent immunometric assay. Luminescence was performed using the Siemens Immulite 1000 (https://www.healthcare.siemens.com/immunoassay/systems/immulite-1000immunoassay-system/assays), following good practices in clinical analyses.
Statistical analysis was performed through overall survival (OS) and progression-free survival (PFS), using the Kaplan–Meier method. The time of PFS was estimated as the time from diagnosis to clinical progression, relapse or death. Data analysis was performed with the statistical program NCSS (www.ncss.com), with a significance level of 5%.
Of the 21 patients recruited, 18 underwent IL-6 sampling in the planned schedule. There were 12 women (58.2%), whose median age was 28 years, ranging from 17 to 68 years. A total of 66.7% presented in stages III and IV. Using the prognostic score (IPS), we classified eight patients (38.1%) as having an unfavorable diagnosis and 13 (61.9%) as having a favorable prognosis. Most patients (66.7%) received the first line of treatment with the Doxorubicin, Bleomycin, Vinblastine and Dacarbazine (ABVD) regimen (Table 1). Two patients died during the study. The median follow-up was 23 months.
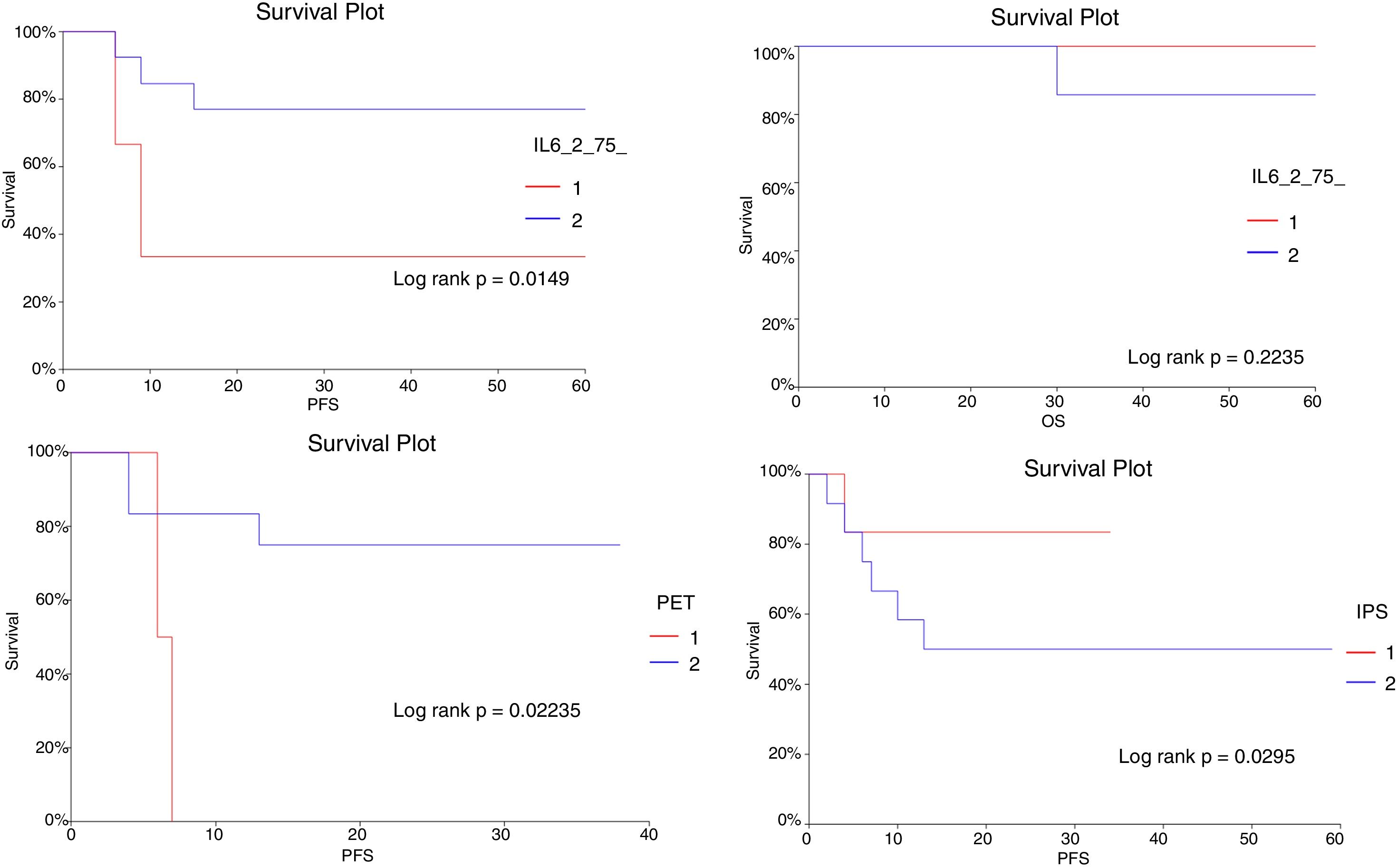
No significant correlations where observed between IL-6 levels, at either diagnosis or after treatment, with IPS, I-PET results or OS. Interestingly, the IL-6 levels at diagnosis were not correlated with the PFS. The interim IL-6 level significantly correlated with the PFS (p<0.0001). Patients with IL-6 higher than 8.05pg/mL after treatment had significantly lower PFS rates, compared to those with lower IL-6 levels (log rank p=0.0149) (Figure 1). Two patients had iodine-124 positron emission tomography (I-PET) delayed to the 3rd cycle, due to logistic issues.
Kaplan–Meier curves of the PFS and OS with IL-6, IPS and I-PET. IL_6_75_: Interim IL-6 (collected after the third chemotherapy cycle); I-PET: Interim PET scan (done after the third chemotherapy cycle); and IPS: International Prognostic Index. We classified the IPS into the favorable and unfavorable groups. In the unfavorable group, we included both intermediary and high-risk groups.
The evaluation of response parameters at intermediary points in the CHL treatment, such as I-PET,9 seems to stratify patients into different prognostic subgroups for whom diverse therapeutic strategies can be considered.
To the best of our knowledge, our study is the first to show that lower Il-6 concentrations in the middle of treatment (Interim IL-6) correlated with a superior disease-free survival (DFS) in patients with CHL treated with chemotherapy. The small sample of patients precluded the use of multivariate analysis. Nevertheless, because we observed no significant associations between IL-6 after treatment and I-PET, it is possible that interim IL-6 could potentially add to I-PET for patient prognostic stratification after the start of therapy.
Conflicts of interestThe authors declare no conflicts of interest.