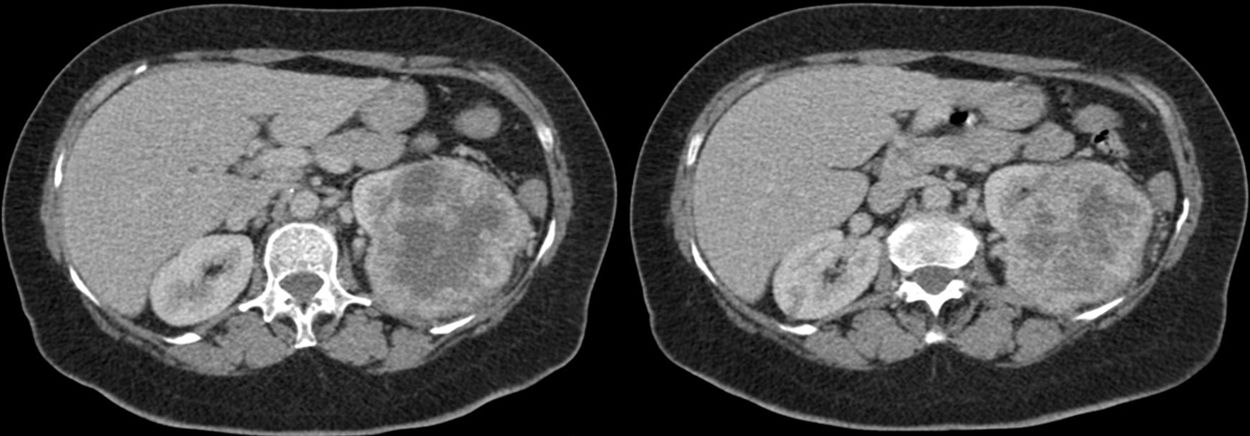
A 65-year-old white female presented with dyspnea caused by ordinary physical activity for about 6 months, associated with episodes of headache, dizziness, mild paresthesia in hands and feet, and difficult-to-control arterial hypertension. Physical examination revealed a large mass in the left hypochondrium and flank. She started periodical phlebotomies as treatment for symptomatic polycythemia about 4 months ago. Laboratory tests showed red blood cell count of 8.15million/mm3, hemoglobin 17.2g/dL, hematocrit 57.9%, reticulocytes 2.2%, serum iron 53mcg/dL, ferritin 65.1ng/mL, LDH 646U/L, urea 25.8mg/dL, creatinine 0.7mg/dL, erythropoietin 82.5mIU/mL (reference range: 4.3–29.0mUI/mL) and negative JAK2 V617F mutation. Computed tomography (CT) scan of the abdomen revealed a left kidney with lobulated contours and heterogeneous density, with a large contrast-capturing heterogeneous nodule with central necrosis area, at upper pole with 10.3×8.5cm (Figure 1). The patient underwent left kidney nephrectomy with histopathological diagnosis of clear cell renal cell carcinoma (CCRCC). After surgery, the patient progressed with resolution of polycythemia and normalization of red blood count.
Paraneoplastic polycythemia associated with excessive erythropoietin (EPO) production is rare, occurring in less than 1–5% of renal cell carcinomas.1 The relationship between CCRCC and secondary polycythemia has been gradually clarified. This neoplasm is usually associated with genetic alterations, and 70% associated with Von Hippel–Lindau (VHL) gene mutations. The usually hypoxic tumor microenvironment causes cells to acquire adaptive modifications such as increased expression of hypoxia-inducible transcription factor (HIF), which is composed of an α and β subunits. HIF-α level are determined by intracellular oxygen tension, and under normal conditions, functional VHL will decode a protein responsible for its rapid inactivation. Mutations of the VHL gene will compromise this process, causing the accumulation of intracellular HIF-α, activating elements responsive to hypoxia, leading to the DNA transcription of molecules that promote angiogenesis, cell proliferation, drug resistance and tumor progression. One of these hypoxia-induced molecules is EPO, which acts at the cellular level to stimulate erythropoiesis.1,2
Although unusual, secondary EPO-producing kidney neoplasms should always be considered in the differential diagnosis of polycythemia. JAK2 V617F testing and abdominal imaging should be performed to avoid a misdiagnosis.
Conflicts of interestThe authors declare no conflicts of interest.