Thrombotic thrombocytopenic purpura (TTP) is one of microangiopathic haemolytic disorder characterized by the fragmented erythrocytes and thrombocytopenia in the peripheral blood, secondary to disease of small blood vessels.1 The reported incidence is 3.7 per million Torok et al., 1995. Prompt recognition and treatment are vital, as delayed treatment have been shown to adversely affect outcome.2 In this study, we report a case TTP who presented with recurrent thrombocytopenia with a brief literature review.
Case reportA 35-year-old female admitted to the emergency departement with complaints of irrelevant speech, headache, numbness and reduced sensations on in the right side of the body. There was no history of seizures, loss of consciousness or trauma. She had a known case of hypothyroidism. She had past medical history of recurrent hospital admissions with thrombocytopenia in the range of 40,000–50,000/uL for the past two years and was treated with steroids. Steroid treatment helped in the increase of platelets, but the level of platelets was not always static and she often required hospital admission for plasma transfusion. She informed a past history of 2 abortions, the last being 1 week prior to this date, with a gestational age of 7 weeks. There was no evidence of retained products of conception. At that time, she was found to have a low platelet count and was diagnosed with immune thrombocytopenia (ITP). On examination, she was conscious, restless, irritable and afebrile. She was anemic, not cyanotic or jaundiced, and presented no clubbing. Her systemic examination did not reveal any significant findings.
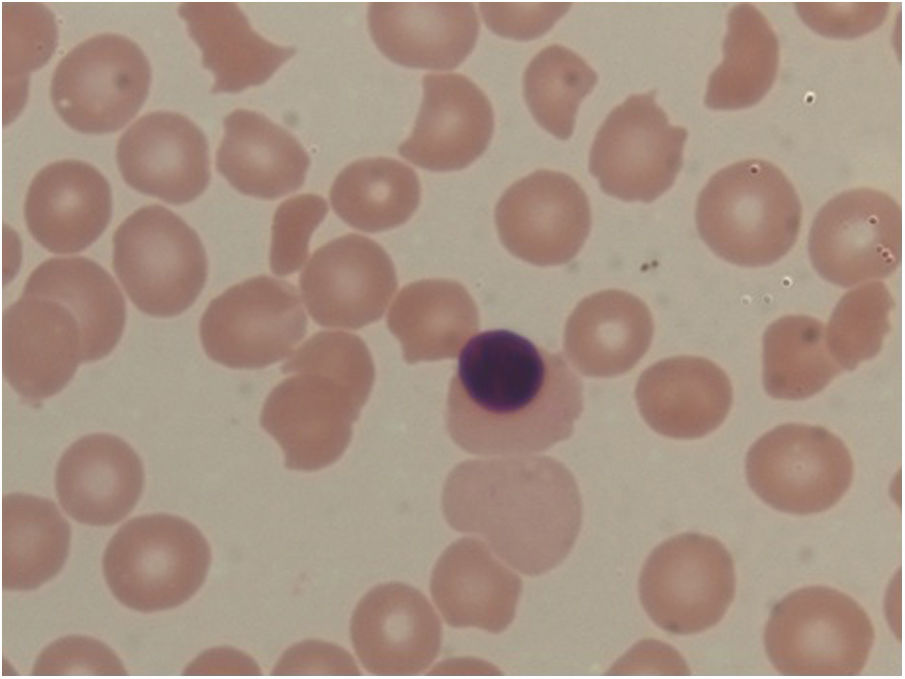
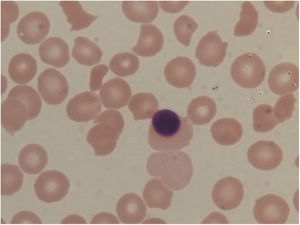
Laboratory investigationsShe was investigated with basic routine investigations and for neurologic symptoms. Complete blood count showed a hemoglobin level of 9.1g/L a total WBC count of 17,300 cells/cu.mm (84% neutrophils) a and platelet count of 23,000 cells/cu.mm. The reticulocyte count was 8%. Red cell indices were normal. The erythrocyte sedimentation rate (ESR) was elevated to 64mmHg. The urine examination was normal. The coagulation profile included the prothrombin time (PT) of 12.6 (C-12.3s), the international normalized ratio (INR) of 1.03 and the activated partial thromboplastin time (APTT) of 24.2 (C-30s). The fibrinogen 358.5 (250−520mg/dl) was normal. The lupus anticoagulant was negative. The peripheral blood smear examination revealed normocytic normochromic anemia with increased schistocytes (9.2%), polychromatophilic red blood cells and thrombocytopenia, suggesting microangiopathic hemolytic anemia (Figure 1).
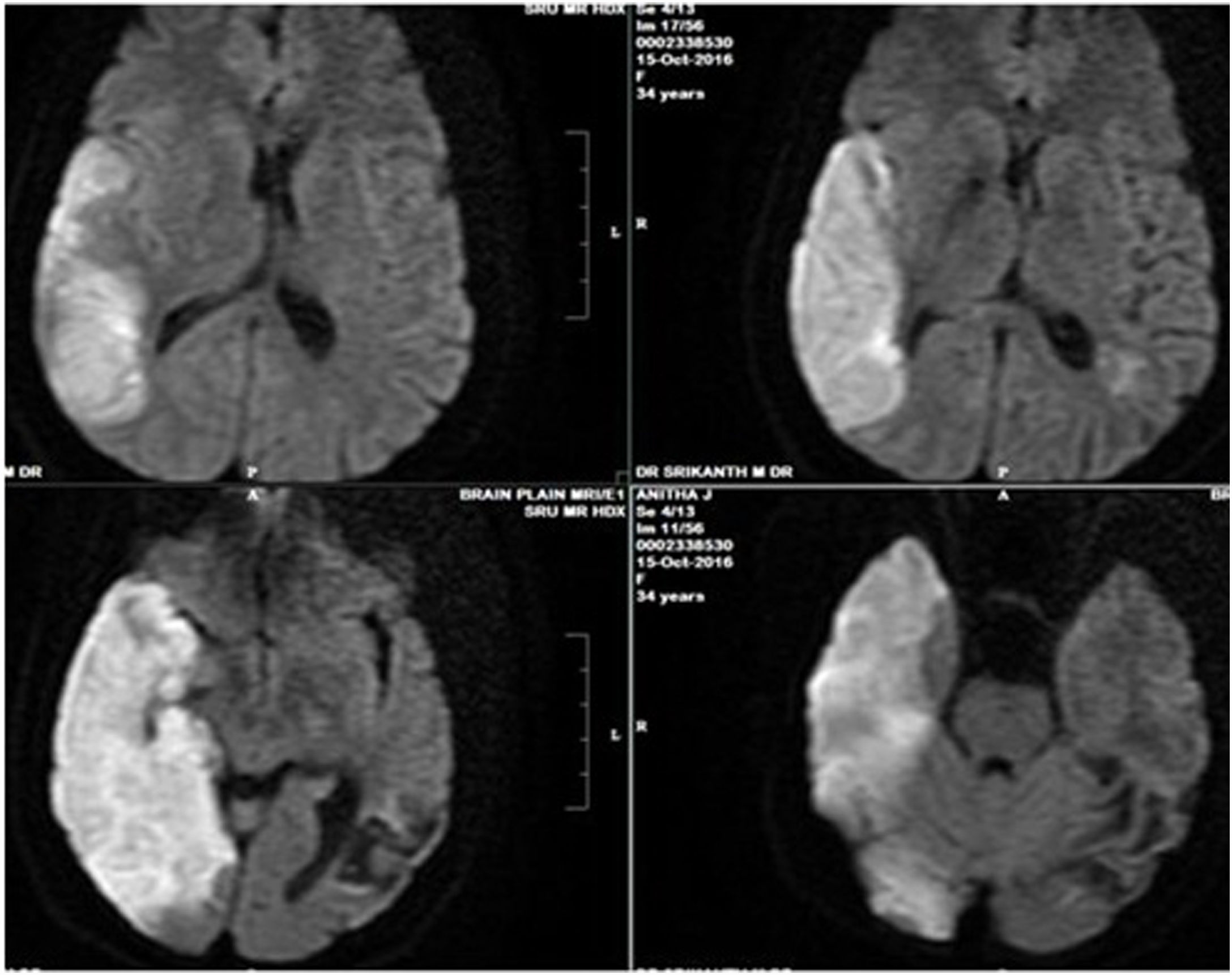
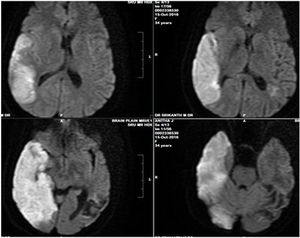
Biochemical investigations revealed elevated total bilirubin levels of 3.13 gm/dL and indirect bilirubin of 2.4gm/DL. The serum lactate dehydrogenase (LDH) was elevated, at 806 U/L. Liver enzymes, such as serum glutamic-oxaloacetic transaminase (SGOT), serum glutamic-oxaloacetic transaminase (SGPT) and alkaline phosphatase were within normal limits. The renal function tests, thyroid profile, lipid profile, blood sugar levels were normal. The antinuclear antibody (ANA), double-stranded (dsDNA), rheumatoid factor, direct coombs test, paraproteins and antiphospholipid antibody (APLA) immunoglobulins (Ig) G and M were normal. The brain MRI with venogram showed an acute non-hemorrhagic infarct in the left parietooccipital region and the computed tomography (CT) venogram showed a subacute infarct in the left middle cerebral artery (MCA) territory (Figure 2). The abdominal ultrasonography (USG) and the chest and abdomen CTs were unremarkable. With the history and laboratory investigations, it was diagnosed as thrombotic thrombocytopenic purpura (TTP). A blood sample was sent for a disintegrin-like metalloproteinase with thrombospondin with type I repeats (ADAMTS-13) levels and inhibitors testing, using a collagen-binding assay, as described by Gerritsen et al.4 The results showed that the ADAM TS-13 levels were undetectable and there were no inhibitors in the sample.
Follow-up and hospital stayThe patient was treated with plasma exchange. As she was intolerant to this and demonstrated moderate allergic reactions, including rashes, it was discontinued and treatment with fresh frozen plasma (FFP) at 10ml/kg body weight, totaling 6 FFPs, 2 units of packed cells, steroids and folic acid was started. After starting the FFP treatment, the platelet count gradually increased and the LDH, reticulocyte and schistocyte counts started to fall. At the time of discharge, which was the seventh day after admission, the patient had a schistocyte count of 0.3% and a normal serum LDH level, reticulocyte count, platelet count, and hemoglobin level.
Three weeks after the first admission, the patient was readmitted to the emergency department with complaints of blurred vision. The hemoglobin was 8g/L, The platelet count was reduced to 60,000, the serum LDH was increased to 792IU and the serum bilirubin was also elevated. The peripheral smear showed schistocytes at 11.5% and reticulocytosis at 5%. She was treated with FFP and supportive management. She was discharged after achieving a normal hemoglobin level and platelet count and the disappearance of schistocytes from the peripheral smear. She was again readmitted after two weeks with ocular disturbances and left upper limb weakness. Her laboratory investigations revealed a low hemoglobin level, a low platelet count, schistocytes at 3% and high LDH and elevated serum bilirubin levels. She was treated with FFP and packed cell transfusion.
DiscussionThe TTP is a disorder characterized by excessive platelet aggregation resulting in the occlusion of the microvasculature, the consumption of platelets, the red blood cell hemolytic crisis and thrombocytopenia.3 The TTP is a classic pentad of features, namely fever, thrombocytopenia, microangiopathic hemolytic anemia, neurologic abnormalities and renal impairment.4 The clinical diagnosis of TTP is extremely difficult, as it has a varied presentation and a tendency to mimic several disorders.5
Schistocytes and shear stressThe characteristic feature in the peripheral blood is the presence of schistocytes. A schistocyte is a broken, torn or sheared red blood cell. The red blood cell fragments after exposure to excessive environmental stress or shearing force within the vasculature.6 Following the variation in the physicochemical properties of red blood cell surfaces, specifically both fibrinogen and globin, a histone can increase the adhesiveness of erythrocytes to fibrin.7 The red blood cells are arrested by fine fibrin strands that are formed during thrombosis and get deformed by the fluid shear. As the flow rate increases, the cells show the morphological changes.8
Enumeration of schistocytesThe International Council for Standardization in Hematology (ICSH) has recommendations for the identification of schistocytes by specific positive morphologic criteria and enumeration of schistocytes. The schistocyte count should be considered clinically meaningful if schistocytes are the main abnormality in the blood cell smear. The morphological indication of schistocytes above 1% is indicative of the diagnosis of thrombotic microangiopathic anemia in adults.9 Schistocytes equal to, or over, 50% of the normal count always comprise less than 0.5% of the red cell population.10 Fragmented red cells enumeration by automated blood cell counters are also a useful complement to microscopy, providing rapid results with a high negative predictive value. There is a good correlation between automated and manual fragmented red cell (FRC) count percentage (r=0.8, p<0.0001, overestimation by 0.82%), but there is lack of correlation in the presence of severe microcytosis, as the analyzers consider the size and not the shape.11
Congenital TTP Upshaw Schaulmann Syndrome is a disease of neonates, infancy, and childhood. The patients present with severe jaundice, hemiparesis, and seizures. Pregnancy is a common precipitant in adults, as in this case report. Fever, infection, alcohol and vaccination are other precipitants. Congenital TTP is characterized by <5% ADAMTS-13 activity and absence of antibodies for homozygous or heterozygous defective ADAMTS13 genes. Acquired TTP also occurs in adults, more frequently among females, usually in the fourth decade, with ADAMTS-13 activity <10% due to an autoantibody that binds the ADAMTS-13 association with autoimmune disease.12
Pathogenesis of TTPThe ADAMTS-13 is a metalloprotease that limits platelet aggregation by cleavage of the Tyr1605-Met1606 bond at the central A2 subunit of the von Willebrand factor (VWF).12 A deficiency in ADAMTS-13 leads to the accumulation of large multimers of VWF and thrombosis. The red blood cells become fragmented between the fibrin strands and cause hemolysis. Johann et al. have described that ultra-large von Willebrand factor multimers (UL-VWFM) were found in the plasma of patients with TTP. The UL-VWF multimers are more efficient in binding GpIb/IX/V and Gp IIb/IIIa receptors and are responsible for the platelet aggregation and thrombogenic symptoms of TTP.13 Paul coppo et al. have demonstrated by immunohistochemistry that the TTP thrombi revealed an abundance of the VWF factor and little fibrin.14 According to Furman et al., the 30 patients diagnosed with TTP and 6 patients with familial TTP lacked all ADAMTS-13 activity; of the 24 patients with non-familial TTP, 20 had severe deficiency (<5% of the normal activity) and 4 had a moderate deficiency (5–25% of the normal activity). Of the 23 patients diagnosed with the hemolytic-uremic syndrome (HUS), 21 patients had normal activity.15
Management of TTPManagement of TTP necessitates a multidisciplinary team comprised by a hematologist, blood bank personnel, a pathologist, a biochemist, coagulation laboratory personnel, a neurologist and an intensivist.16 The British Journal of Haematology (BJH) guidelines state that a plasma exchange at 1.5 plasma volume ×3 should be immediately initiated, followed by 1 plasma volume/day until the conditions are stabilized.17 The patient is considered to be in remission if the platelet count is above 50,000/uL 30 days after the last plasma exchange. Relapsed patients are treated with daily plasma exchange, steroids, and Rituximab.18 With regard to the choice of the replacement fluid, there is great variability. A cryosupernatant is claimed to be preferable, as it lacks the large VWF multimers. However, studies by Zeigler et al. have proved that there is not much difference in the outcome, whether a cryosupernatant or fresh frozen plasma is used.19 An earlier BJH research paper observed that there is no advantage of cryosupernatant (CSP) over FFP as the choice of replacement fluid in the management of TTP.20
ConclusionThrombotic thrombocytopenic purpura is a hematological emergency and a diagnostic challenge. The key factor in patient survival is timely diagnosis and treatment. Since the presenting symptoms vary significantly, and as it mimics more common disorders, the outcome depends largely on the prompt recognition and knowledge of this rare and rapidly fatal disease. The management of TTP again stresses the importance of the multidisciplinary team in the diagnosis and management of hematological diseases.
Ethics approval and consent to participateThe study was approved by the institutional ethics committee “Ramachandra Institute of Higher Education and Research SRIHER” and informed constant were obtained from each individual participant, who was informed of the study follow-up before enrolling in the study.
Consent to publishThis study was approved by the Institutional Ethical Commitee (IEC), which granted us permission to publish the report.
Availability of data and materialsAll associated data are available in this manuscript, no further data beyond what is presented being available.
FundingNone.
Author contributionsAn equal contribution was made by both the authors.
Conflicts of interestThe authors declare no conflicts of interest.