Chronic myeloid leukemia (CML) is an indolent neoplasm which is known for its invariable progression from a chronic to blastic phase. However, CML primarily presenting with megakaryocytic blast crisis (MKBC) in a young male without any preceding chronic phase is of extremely rare occurrence. CML in transformation carries a dismal prognosis and imposes a major therapeutic challenge. Accumulations of certain mutations and chromosomal aberrations derive a case of CML from a relatively benign chronic phase to blast phase which is characterized by presence of >20% blasts in the peripheral blood or bone marrowor an extramedullary accumulation of blast cells, or large foci or clusters of blasts in the bone marrow biopsy (WHO 2016). These criteria must be supported by the presence of Philadelphia (Ph) chromosome resulting from reciprocal translocation of chromosomes 9 and 22.1,2 Phenotype most commonly seen in blast crisis is of the myeloid lineage followed by lymphoid and erythroid. MKBC is extremely uncommon, comprising of <3% of CML in transformation.2 We report an unusual case of a young man who presented for the first time with splenomegaly and presence of blasts in the peripheral blood and bone marrow, clinically masquerading as acute leukemia.
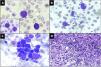
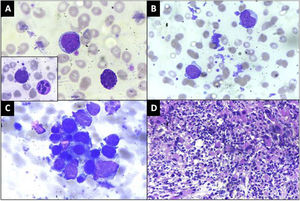
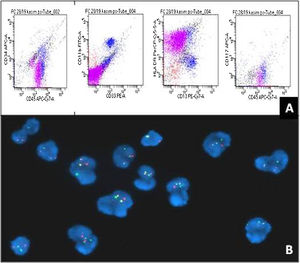
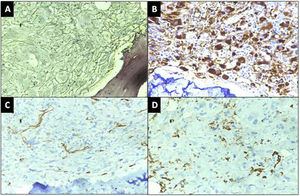
Case reportA 22-year-old male presented with complaints of progressive fatigue, abdominal discomfort and weight loss for one month. He had no history of cough, fever, mucocutaneous bleeding and tuberculosis contact. His family history was unremarkable. On general examination, he had significant pallor, mild hepatomegaly and a moderate splenomegaly extending 6 cm below the left costal margin. His liver and renal function tests were within normal limits. Ultrasound abdomen revealed hepato-splenomegaly Routine hemogram revealed haemoglobin of 5.6 g/dL, white blood cell (WBC) count of 1.34 × 109/L and a platelet count of 165 × 109/L. The peripheral blood smear showed moderate anisopoikilocytosis in the red blood cells (RBC) with predominantly microcytic hypochromic cells along with a fair number of dacrocytes and nucleated RBCs (8/100WBC). The leukocyte differential count was blast 36%, myelocytes 4%, metamyelocytes 2%, neutrophils 36%, lymphocytes 16%, monocytes 2%, eosinophils 1% and basophils 3%. Blasts were large in size with high nucleocytoplasmic ratio, fine chromatin, 2–3 nucleoli and moderate basophilic cytoplasm with cytoplasmic blebs at places. Auer rods were not seen. Dysplastic changes were seen in around 10% neutrophils in the form of hypolobation and hypogranulation. Few large platelets and megakaryocytic fragments were also noted (Fig. 1A, B). Cytochemical staining with Myeloperoxidase (MPO) was negative in the blasts. Based on these findings, a provisional diagnosis of acute leukemia was considered and the patient was planned for bone marrow aspiration (BMA), flowcytometry (FCM) and cytogenetic studies. BMA smears were aparticulate and diluted with peripheral blood, however blasts cells with similar morphology were noted along with myeloid and erythroid precursors (Fig. 1C). On, FCM, the blasts were dim positive for CD45, with CD38 heterogenous, HLA-DR heterogenous and CD13 dim expression. CD34, CD33, CD117 and cytoplasmic MPO were negative in these cells (Fig. 3A). Bone marrow biopsy (BMB) revealed predominantly fibrosis with marked proliferation of large atypical cells with megakaryocytic differentiation along with numerous micromegakaryocytes. There was clustering and streaming of megakaryocytes with interspersed small pockets of erythroid and myeloid cells (Fig. 1D). Reticulin staining revealed grade III fibrosis (Fig. 2A). An immunohistochemistry (IHC) panel was applied and the large atypical cells as well as the megakaryocytes expressed CD61 thereby confirming the megakaryocytic phenotype of the blasts (Fig. 2B). CD34, CD117 and MPO were negative in the atypical cells (Fig. 2C, D). A Fluorescence In-Situ Hybridization (FISH) test was performed on peripheral blood and it yielded >90% Ph-positive cells (Fig. 3B).This was followed by a quantitative reverse transcriptase polymerase chain reaction (RT-PCR) which confirmed the presence of p210 BCR-ABL-1 transcript.An overall final diagnosis of CML in MKBC was rendered. Patient was subsequently put on a 7 + 3 regimen of cytarabine and doxorubicin in combination with Dasatinib 140 mg daily. A hemogram repeated after two months showed a striking response to therapy with a WBC count of 2400/µL and a differential count of neutrophil 43%, lymphocyte 55%, eosinophil 1% and monocyte 1%, thereby suggestive of hematological remission. There was regression of spleen and FISH for BCR-ABL-1 was negative confirming a complete cytogenetic response. Patient was advised to continue dasatinib and was further planned for allogeneic hematopoietic stem cell transplantation (HSCT). The patient however did not turn up for follow-up since then citing his financial restraints as the reason.
A: Peripheral blood smear showing a blast and a basophil. Red Blood cells in the background show moderate anisopoikilocytosis. Inset: A dysplastic hypersegmented neutrophil along with a blast (×1000, Giemsa), B: Blasts with cytoplasmic blebs and megakaryocytic fragments, (×1000, Giemsa) C: Bone marrow aspirate smear showing a cluster of dysplastic megakaryocytes and blasts (×1000, Giemsa), D: Trephine biopsy showing streaming and clusters of dysplastic megakaryocytes and megakaryoblasts (×400, Hematoxylin & eosin).
CML in blast crisis may show magakaryoblastic phenotype which poses a diagnostic challenge in differentiating it from de novo Acute Megakaryoblastic Leukemia (AMKL) which is also a rare diagnosis.3–7 The two entities are difficult to differentiate morphologically alone especially when no prior history of CML is available. Contrary to the usual occurrence of CML in the fourth decade in the Indian population, our case involved a young patient.8 Splenomegaly, leukocytosis and basophilia, which are consistent features of CML were also present in our patient. Persistence or increase in any of these signifies disease progression.1,2 In contrast, none of the three features are seen in de novo AMKL (Table 1).2,3,6,7 The megakaryoblasts are usually medium to large in size (12–18 µm) with a round to indented nucleus, fine reticular chromatin, one to three nucleoli and basophilic cytoplasm which may show distinct blebs or pseudopod formation.2,3 Circulating micromegakaryocytes were not seen but megakaryocytic fragments, dysplastic large platelets and hypogranular neutrophils which are a feature of AMKL, were conspicuous in the present case. The dysplastic features seen in the neutrophils as well as the erythroid precursors on BMA in our case can also be seen in AML with myelodysplasia-related changes (AML-MDC), however dysplasia in AML-MDC must be present in minimum 50% of the cells in at least 2 lineages, supplemented by the signature cytogenetic abnormalities.2 Hence AML-MDC was excluded in this case.
Differential diagnosis of CML in Megakaryocytic Blast Crisis.
| CML-MKBC | De novo AMKL | Acute Panmyelosis with MF | Primary MF in Blast transformation | Ph-Positive AML | |
|---|---|---|---|---|---|
| Leukocytosis | Present | Leukopenia | Pancytopenia | Present | Present |
| Splenomegaly | Present | Absent | Absent | Present | Less frequent |
| Basophilia | Usually present | Absent | Absent | Absent | Absent |
| CD34 positive blasts | Usually absent | Usually absent | Present | Present | Present |
| BCR-ABL transript | Present (p210) | Absent | Absent | Absent | Present (p190) |
CML Chronic Myeloid Leukemia, MKBC Megakaryocytic Blast Crisis, AMKL Acute Megakaryoblastic Leukemia, MF Myelofibrosis, Ph Philadelphia, AML Acute Myeloid Leukemia.
BMA of our patient yielded a diluted marrow probably due to myelofibrosis which is a frequent finding in both denovo AMKL and MKBC.3–7 Besides fibrosis, bone marrow biopsy in both conditions shows varying proportions of blasts and maturing dysplastic megakaryocytes. Confirmation of megakaryocytes and megakaryoblasts requires expression of platelet-specific antigens such as CD41, CD42, CD61 or Factor VIII on IHC or FCM. Megakaryoblasts are often negative for CD34 and MPO, however markers like CD13 or CD33 may be positive, as in our case.2,3,5,7 A leukoerythroblastic blood picture along with presence of >20% blasts, myelofibrosis and presence of clusters of abnormal megakaryocytes in the BMB implicate consideration of AMKL, acute panmyelosis with myelofibrosis, and primary myelofibrosis in blast transformation as the possible differentials besides a diagnosis of CML with blast crisis.2,7 Features such as leukocyte count, splenomegaly, basophilia and CD34 positive blasts in the marrow provide a valuable clue in differentiating between these close mimicking entities (Table 1). However, demonstration of BCR-ABL fusion protein or Ph chromosome proves pivotal in arriving at a diagnosis as in our case. Here, another neoplasm namely, Ph-positive AML (WHO 2016) needs to be addressed.2 Literature states that presence of left shifted myeloid maturation, splenomegaly and basophilia in the peripheral blood favour a diagnosis of CML in blast crisis rather than Ph-positive AML9 (Table 1).
CML-MKBC carries a poor prognosis with usually low expected survival rate.1,10 Combination of chemotherapy and tyrosine kinase inhibitors (TKI) has proven beneficial in improving the disease outcome to the extent of achieving a haematological and cytogenetic remission as in our patient.10 However, a molecular response needs to be monitored in such patients by a qualitative RT-PCR, which could not be done in our case. Also, despite a good initial response, TKI therapy does not appear to offer long-term survival benefits in advanced phase CML. In such cases, allogeneic HSCT is the treatment of choice, especially once a chronic phase is achieved.11 Unfortunately, HSCT remains beyond the availability of many patientsdue to the high cost involved, as was the case with our patient.
ConclusionMegakaryocytic blast crisis has a characteristically low incidence and may prove to be a diagnostic challenge for the clinicians and the pathologists especially in previously undetected CML cases. The case report highlights the importance of differentiating blast transformation of CML from other close differentials with the use of an appropriate IHC panel and cytogenetic studies, as there are therapeutic implications.
Conflicts of interestThe authors declare no conflicts of interest.