Mantle cell lymphoma (MCL) accounts for 3–10% of Non-Hodgkin's Lymphoma (NHL) and generally affects patients who are approximately 60 years old. It is genetically marked by t(11;14)(q13;q32), which deregulates cyclin D1. As a consequence, the whole cellular cycle becomes deregulated, offering possibilities for multiple mutations and generating a complex genetic landscape, multidrug resistance and disease refractoriness.1–5 Therefore, besides the fact that approximately 10–20% of patients have an indolent course, it is the worst prognostic B-cell lymphoma.
Over the last decade, the incorporation of rituximab in all chemotherapeutic regimens, widespread adoption of cytarabine-based therapy and autologous Peripheral Blood Stem Cell Transplantation (PBSC) in younger and fit patients has become an evidence-based standard of care.6 However, MCL is still regarded as incurable, with a reported median overall survival (OS) of 3–4 years, in its aggressive form. Recently, the development of new drugs, such as Bendamustine and Ibrutinib, and their incorporation into the MCL treatment schedules has improved its median overall survival,2,3 placing the MCL treatment into a new scenario.6–10 Nevertheless, as Rituximab, Bendamustine and Ibrutinib are not available to most patients in many developing countries around the world including Brazil in its public health system, access to this new scenario has been limited. Therefore, many Brazilian MCL patients afflicted with its aggressive form usually do not benefit from the newer disease-altering therapies.
In this study, we show how this situation impacts patient survival in one of the major general hospitals in Brazil.
Cases and methodsIn a retrospective study, we evaluated non-indolent MCL patients treated at the Hematology and Oncology Service at Santa Casa of Sao Paulo (SCSP), Sao Paulo, Brazil, between January 1, 2003 and May 31, 2015, according to the WHO-2008 diagnosis criteria. The cytology, histology and immunohistochemistry analyses were performed by two cytohematopathologists. In case of diagnosis disagreement, a third professional stood by to settle the issue.
Age, gender, Ann Arbor stage, International Prognosis Mantle Cell Lymphoma Index (MIPI), extranodal commitment, bulk disease – defined as a tumor mass larger than 7.5cm – and treatment data were considered in all patients. Unfortunately, as we had not performed the test, we did not have Ki-67 for most patients, so we decided we could not use it as a prognostic factor.
Patients were divided into two groups according to age: younger than 65 years old [young subgroup (Y)] and 65 years or older [elderly subgroup (E)]. They were also divided into 3 groups according to treatment: the “standard” chemo group (SCG), the “non-standard” chemo group (NSCG) and the palliative group.
We called SCG the one nearest to the standard treatment practice. An SCG patient younger than 65yo received AraC-HD with or without Rituximab, followed by autologous PBSC, or received anthracycline-based chemotherapy with Rituximab, but without AraC-HD and without autologous PBSC, if the patient was 65yo or older. On the very opposite side were bed-confined patients who did not have the performance status to receive chemotherapy of any kind. All other patients receiving any other chemotherapy without autologous PBSC were considered as NSCG (Table 1).
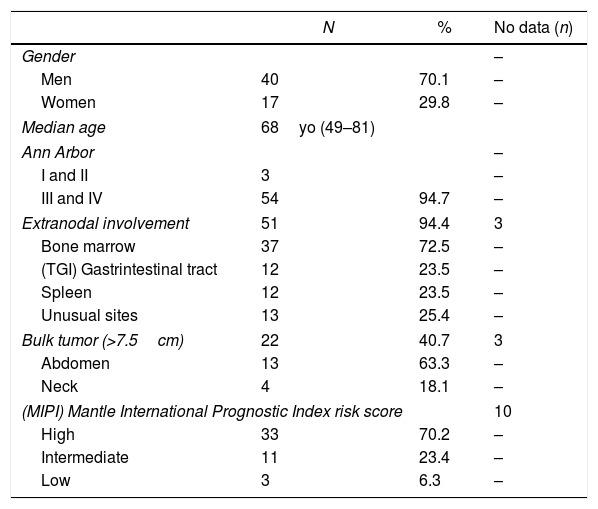
General characteristics of (MCL) Mantle Cell Lymphoma patients treated at the Hematology and Oncology Service at Santa Casa of Sao Paulo (SCSP), Sao Paulo, Brazil, between January 1, 2003 and May 31, 2015.
| N | % | No data (n) | |
|---|---|---|---|
| Gender | – | ||
| Men | 40 | 70.1 | – |
| Women | 17 | 29.8 | – |
| Median age | 68yo (49–81) | ||
| Ann Arbor | – | ||
| I and II | 3 | – | |
| III and IV | 54 | 94.7 | – |
| Extranodal involvement | 51 | 94.4 | 3 |
| Bone marrow | 37 | 72.5 | – |
| (TGI) Gastrintestinal tract | 12 | 23.5 | – |
| Spleen | 12 | 23.5 | – |
| Unusual sites | 13 | 25.4 | – |
| Bulk tumor (>7.5cm) | 22 | 40.7 | 3 |
| Abdomen | 13 | 63.3 | – |
| Neck | 4 | 18.1 | – |
| (MIPI) Mantle International Prognostic Index risk score | 10 | ||
| High | 33 | 70.2 | – |
| Intermediate | 11 | 23.4 | – |
| Low | 3 | 6.3 | – |
Treatment response was analyzed after complete chemotherapy treatment schedule, according to a CT scan or PET–CT scan and bone marrow biopsy. Based on these, responses were defined as Confirmed/Unconfirmed (cCR/uCR), partial (PR) or refractory (RR), according to Cheson et al.11 (Figure 1).
We considered the follow-up (Fu) as the time between the diagnosis and death or the last visit, overall survival (OS) as the time between the diagnosis and death and time to next treatment (TTNT) as the time between the finish of the first treatment and the beginning of a new one. Overall survival and time to next treatment were calculated according to the Kaplan–Meier method and differences in subgroups were analyzed by the log-rank test. Age, gender, Ann Arbor stage and MIPI index were submitted to univariate analysis. All p-values were two-tailed and a confidence interval of 95% was used. Statistical analysis was performed with the SPSS2.0 and Microsoft excel 2010 version.
The study was approved by the local ethics committee of Santa Casa of Sao Paulo, Sao Paulo, and conducted in accordance with the Declaration of Helsinki.
ResultsSixty-four patients were diagnosed with MCL in its non-indolent form between January 1, 2003 and May 31, 2015. The median age at diagnosis was 67 years old and 36 patients (63.1%) were 65 or older and MCL was more than twice as common among men with a male/female ratio of 2.3:1.0. We excluded 7 patients from the study because we found incomplete medical records. Therefore, we analyzed 57 patients.
Clinical presentation and prognostic factorsThe general patient characteristics are shown in Table 1. Ann Arbor stage IV was observed in 46 patients (80.7%), 3 patients (5.2%) presented localized disease (Ann Arbor stage I or II without systemic symptoms), and 8 patients (14.0%) were not staged. Extranodal involvement was observed in 51 patients (89.5%) and the most frequently involved sites were bone marrow, gastrointestinal tract and spleen. We also observed lungs, pleura, eyes, nasopharynx and oropharynx involvement. Moreover, in 26 patients (45.6%) we observed at least two simultaneously involved extranodal sites. This data did not show a statistically significant difference between the younger and elderly subgroups, however when we considered the presence of bulk disease, we observed it more frequently in the younger group (61.9%) than in the elderly one (38.5%).
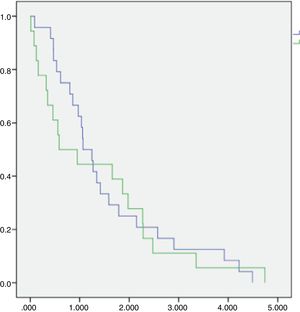
Ten patients (17.6%) did not have data to calculate the MIPI score, so we performed it for 47 (82.4%), of whom 33 (70.2%) were classified as high-risk, 11 (23.4%), as intermediate-risk and 3 (6.4%), as low-risk. The Ki-67 was not performed for most of the cases, so we did not calculate the biological-MIPI. Comparing the high-risk and intermediate-risk MIPI groups, the Kaplan–Meier OS curves showed a trend favoring intermediate-risk survival (Figure 2). Because we had only 3 low-risk patients, the comparison between this MIPI risk group and others was not statistically possible.
Treatment modalitiesFrom 57 patients, we analyzed 44 patients submitted to systemic therapy. Thirteen patients were excluded because of frailty, and were submitted to palliative treatment, or because of death before systemic therapy was begun.
From 44 analyzed patients, 26 (59.1%) were in the SCG. In this group, 17 (65.4%) were younger than 65 years old and 9 (34.6%) were 65 or older. After induction chemotherapy, we observed cCR/uCR in 10 and PR in 9. (We did not get treatment response information for 7 patients.) From 19 patients, 10 cCR/uCR younger patients were submitted to high-dose chemotherapy followed by autologous PBSC transplantation. Subsequently, 6 remained in cCR/uCR and we lost the follow-up for 4 (Figure 1). Concerning the 7 PR patients, 2 refused the autologous PBSC, 3 did not get enough stem cells to perform the treatment and 4 presented progressive disease immediately before apheresis.
In the remaining patients submitted to chemotherapy, 18 belonged to the NSCG and all but 1 were 65 years old or over. After treatment, only 4 were classified as cCR/uCR (Figure 1).
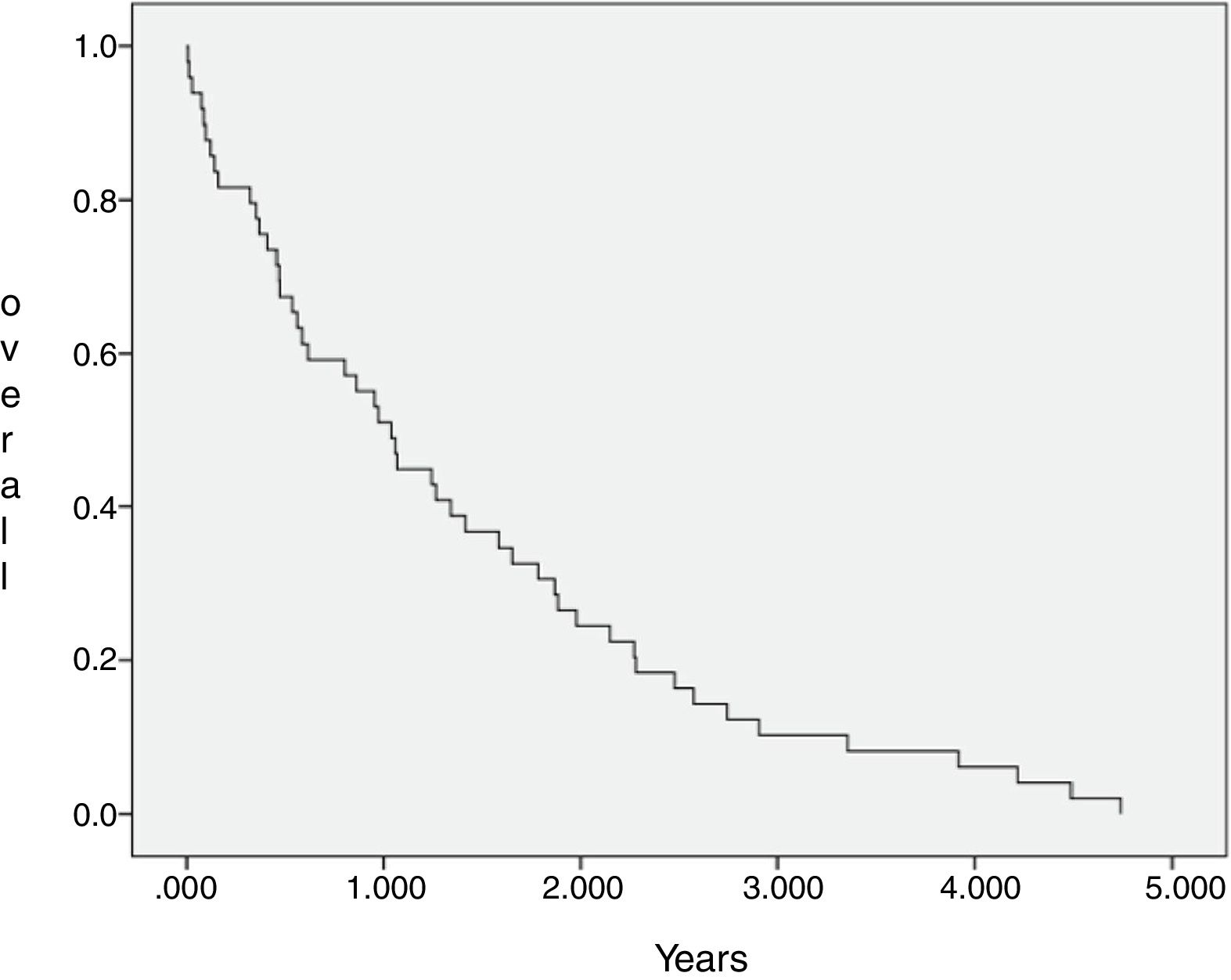
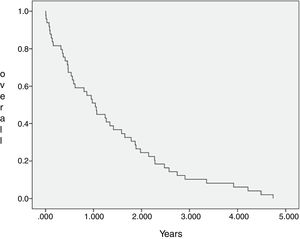
The median OS of all 44 patients submitted to chemotherapy was 12.9 months (IC 95=8–17.8 months), with 2 and 3 years estimated overall survival of 24.2% and 9.7%, respectively. The SCG and NSCG overall survival were 12 months and 9.6 months, respectively, and these data were not statistically significant (Figures 2 and 3).
DiscussionThe B-cell lymphoma with the worst prognosis is MCL. It is clinically characterized by frequent relapses and early refractoriness. Therefore, the search for new approaches with new treatment options has received attention over the last few years. This has been translated into different managements: Rituximab inclusion in induction and maintenance schedules, high-dose Cytarabine inclusion in first-line treatment for younger fit patients,7,8,12–17 Bendamustine in the elderly population15 and new drugs, such as NF-κβ and Bruton-kinase inhibitors, such as Bortezomib and Ibrutinib, respectively, which resulted in an increasing response rate and overall survival.18–22 However, in the public health system in Brazil, most of these new approaches are not available.
Our aim was to analyze non-indolent MCL patients in a cohort study at a single public hospital in Brazil. Thus, we evaluated the MCL patients treated at the Hematology and Oncology Service at Santa Casa of São Paulo, SP, Brazil, between January 1, 2003 and May 31, 2015.
In a retrospective cohort study, we analyzed 57 patients. The epidemiologic aspects showed that our population median age was 68 years old, with a 2.3:1.0 men to women ratio. These data are similar to those which are usually found.2,3 In clinical aspects, almost 95% of our patients were Ann Arbor stage III or IV and their extranodal involvement was also near 95%. These data are also similar to the literature, in which approximately 90% of patients already presented with Ann Arbor stage IV and bone marrow involvement at diagnosis, even in developed countries.4,9,13,14 Concerning the prognostic index in our cohort, more than 70.0% had a high MIPI score and only 6.3% had a low-risk MIPI score, so our cohort had a higher MIPI score than those in developed countries, which is approximately 40%. In addition, our overall survival curves for intermediate and high-risk MIPI scores were very inferior to those found by Hoster et al. These authors observed 51 months overall survival for an intermediate MIPI score and 29 months for a high MIPI score, while our data showed 23 months and 10 months overall survival, respectively. Moreover, our data showed a median overall survival inferior to 13 months and a 3-year estimated overall survival inferior to 10% for the whole group. Hence, there are two salient points: first, is why such a high-index MIPI score proportion and second, why such a low overall survival.
The real world showed by Abrahamsson et al.16 differs from ours. Patients that use the public health system in Brazil often belong to lower social, cultural and economic brackets. This fact influences nutritional status and disease understanding, both of which impact treatment response. This fact is already observed in other diseases.22 However, there are some points that do not depend on the patient's sociocultural or economic status, but rather on the health system itself. Hence, we believe our results reflect a double problem in the Brazilian public health system. Besides the fact that we did not evaluate the time between the patient's initial symptoms and diagnosis and time between the diagnosis and initial treatment for MCL, the high-risk MIPI scores proportion might point to the patient's difficulties in arriving at a Hematology-Oncology service capable of diagnosing and treating MCL. Nevertheless, once a patient arrives at an appropriate center, we observe that the correct diagnosis does not necessarily mean proper treatment, as the public health system in Brazil does not make available the most commonly used drugs in modern chemotherapy schedules. We only used Rituximab to treat MCL patients for a short period of time when it was permitted. Subsequently, the MCL patients in the Brazilian public health system in general cannot use Rituximab either for induction or for maintenance, as established by the Ministry of Health Ordnance.23 Neither can it be used for purging “in vivo”. We think these facts explain the poor results in the auto-PBSC group. On the other hand, Bendamustine, Bortezomib or Ibrutinib unavailability limits therapeutic options and helps to explain the poor results in the non-auto PBSC group.
This combined situation impacts our results, as not only patients are frequently overly high-scored in the prognosis, but also fewer patients will achieve complete first remission and if they do, relapse tends to come earlier, as there are few disposable drugs. Even if a patient achieves a second remission this one will depend on treatments that show more toxicity and/or inferior results than the advisable ones. We observe a uCR/cCR rate inferior to 50% and an estimated overall survival in 3 years near to 12% in the SCG, which is inferior to that of the MCL2 trial study that showed not only how important high-dose Cytarabine is, but also the importance of the addition of Rituximab to the chemotherapy schedule.17 In the NSCG, our results were even worse, as we could not use Rituximab in induction and/or maintenance, and we observed a 3-year estimated overall survival around 7%. In comparison, the SCG and NSCG overall survival results were not statistically significant, which means that with the disposable chemotherapy armamentarium, whatever efforts we make, the results are quite the same, regardless of patient fitness or age (Figure 3).
We hope in the near future patients will have a faster access to expert services and benefit from newer and better approaches already used in many countries. An earlier and economically viable treatment with a better approach will result in fewer relapses and a better quality of life.
Conflicts of interestThe authors declare no conflicts of interest.


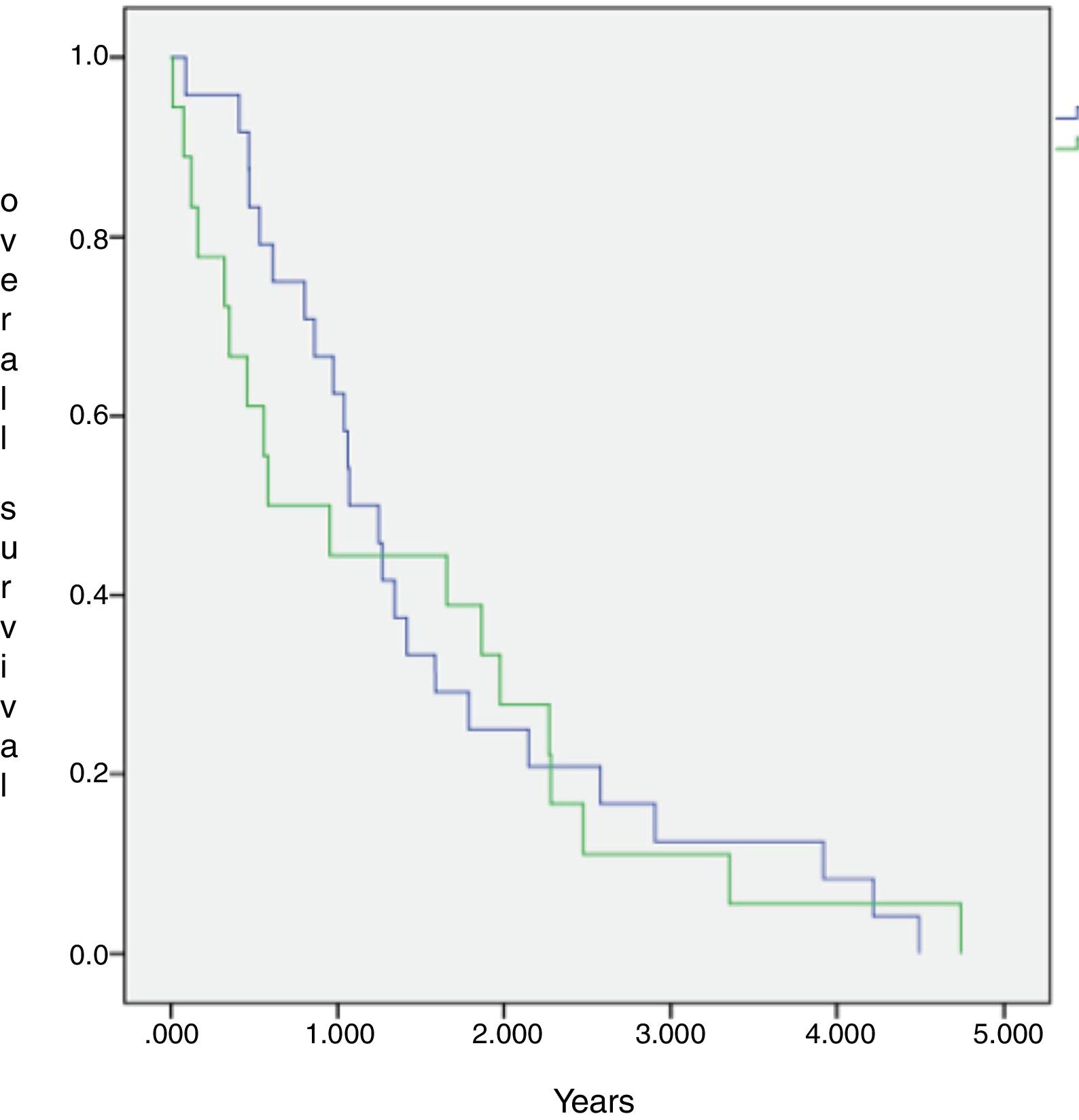
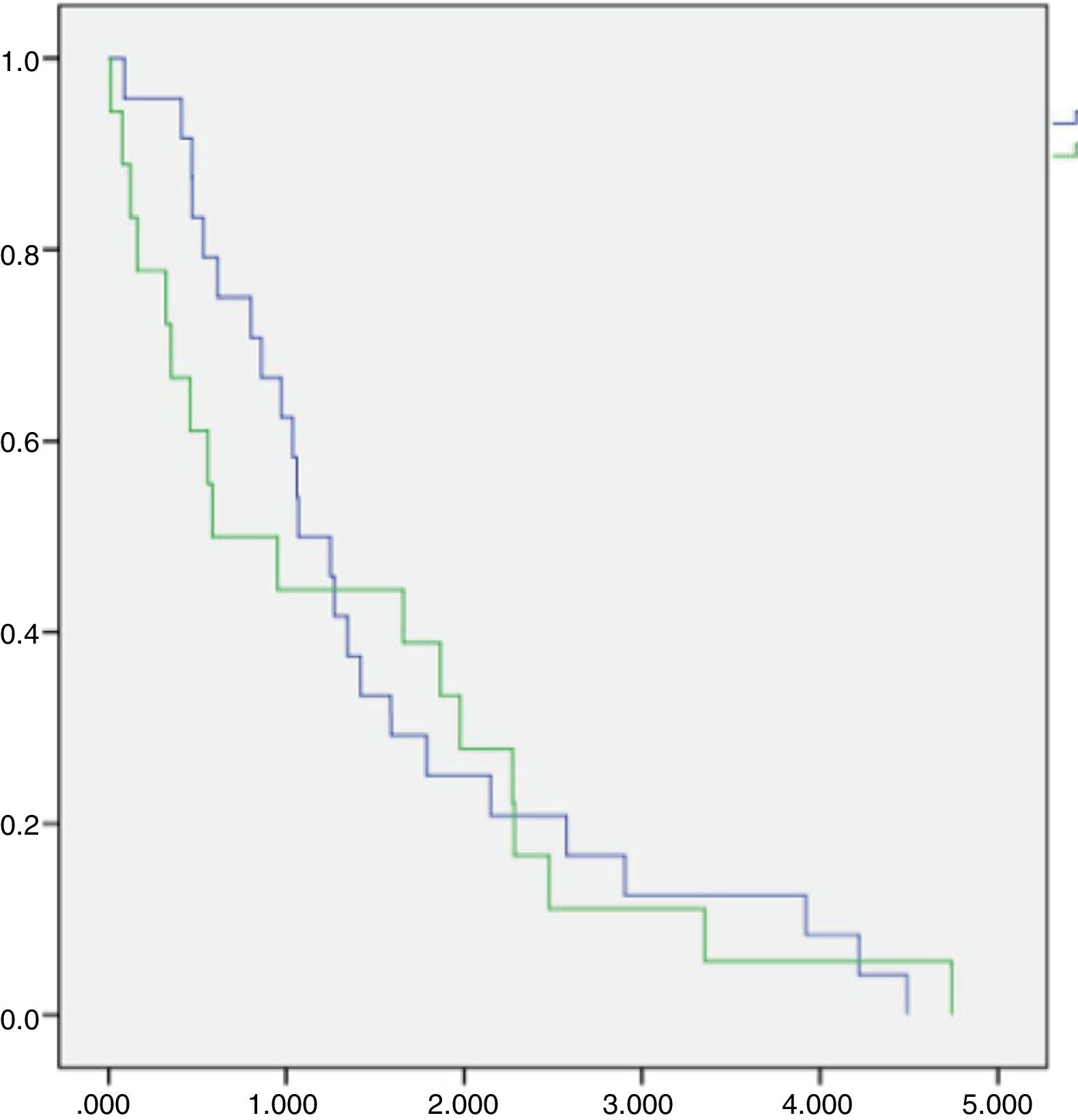
![Overall survival of fifty-seven MCL patients followed at Santa Casa of Sao Paulo between January 1, 2003 and May 31, 2015, according to chemotherapy treatment groups [standard (blue curve)×non-standard (green curve)]. Overall survival of fifty-seven MCL patients followed at Santa Casa of Sao Paulo between January 1, 2003 and May 31, 2015, according to chemotherapy treatment groups [standard (blue curve)×non-standard (green curve)].](https://static.elsevier.es/multimedia/25311379/0000004200000001/v2_202003250636/S2531137919300471/v2_202003250636/en/main.assets/thumbnail/gr2.jpeg?xkr=ue/ImdikoIMrsJoerZ+w93OM6WmS6o9DeZl+SVh74uo=)