Acute promyelocytic leukemia is a subtype of acute myeloid leukemia, characterized by the presence of neoplastic promyelocytes, due to the reciprocal balanced translocation between chromosomes 15 and 17. Currently, with the use of agents that act directly on this molecular change, such as all-trans retinoic acid and arsenic trioxide, APL has shifted from a highly mortal to a curable disease. However, some cases are still at high risk of death, especially early death, and acquiring a better understanding of the clinical and biological factors involving APL is needed to correctly identify and treat such cases. The early suspected diagnosis and prompt initiation of the target therapy are important for better response rates. The follow-up and outcomes, using real-life data from 44 consecutive APL patients, were studied between 2001 and 2013. The overall survival rate was 82.7% and early death was 16%. Almost all patient deaths were due to severe bleeding, which was confirmed by multivariate analysis, as the most important prognostic factor leading to death. A better understanding the pathogenesis of the hemorrhagic complications in APL is needed, as well as the risk factors associated with early death in APL patients, as this has become synonymous with overall mortality.
Acute promyelocytic leukemia (APL) is a distinct subtype of acute myeloid leukemia (AML), most common in young adults, characterized by the accumulation of neoplastic promyelocytes in the bone marrow or/and peripheral blood.1 Cytogenetically, APL is identified by a balanced reciprocal translocation between chromosomes 15 and 17, which results in the fusion between the promyelocytic leukemia (PML) gene and retinoic acid receptor-α (RARα) gene, that can be identified by modern molecular techniques, such as the reverse transcription polymerase chain reaction (RT-PCR) for t(15;17).2 Immunophenotypically, APL cells share certain features with normal promyelocytic cells, however, APL cells express abnormally low levels of CD15 and weak or variable CD117.3,4
Clinically, APL represents a medical emergency, with a high rate of early mortality often due to a hemorrhage from typical coagulopathy associated with APL (TC-APL), low platelet count and/or hyperfibrinolysis.5,6 The prompt identification of APL, rapid target treatment and major support measures to avoid severe bleeding are important for good response rates.1
The introduction of all-trans retinoic acid (ATRA), in 1987, changed the treatment of APL.7,8 The use of ATRA, combined with anthracycline-based chemotherapy for induction and anthracycline-based chemotherapy, with or without cytarabine arabinoside (Ara-C) in consolidation, depending on the risk level, is still considered the standard-care treatment for APL.5,9–11 Arsenic trioxide (ATO) has been shown to be highly effective in APL and was initially tested in patients with relapse after ATRA and chemotherapy, but recent studies demonstrated the success of chemotherapy-free therapy (CFT) with ATO plus ATRA in first-line therapy in low- and intermediate-risk groups.12–14
The incidence of APL in Latin America is slightly high (20–25%), compared to North American and Northern European countries (10–15%), and some particularities of this disease must be studied.15,16 Some cases are still at high risk of death, especially early death (ED) and a better understanding of the clinical and biologic factors involving APL is needed to correctly identify and treat these cases.5,17,18 This retrospective study reported the clinical features and outcomes of forty-four consecutive newly diagnosed young adult APL patients, diagnosed between January 2001 and July 2013.
Material and methodsPatients and diagnostic proceduresForty-four consecutive newly diagnosed young adult APL patients, diagnosed between January 2001 and July 2013 and treated at the Hematology Service of the Hospital Universitário of the Universidade Federal de Santa Catarina (HU-UFSC), were included in this study, according to the criteria of the research ethics committee of human beings (CAAE 03704812.7.0000.0121). Informed consent was obtained from all patients or guardians to be included in the study. The APL diagnosis was based on clinical features, morphological evaluation, immunophenotype, cytogenetic and molecular genetic studies, according to the criteria of the World Health Organization (WHO) Classification of Tumors of Hematopoietic and Lymphoid Tissue.3 The diagnostics were confirmed by the RT-PCR, as reported by the BIOMED-1 Program.19 The type of PML-RARα fusion products (PML break at bcr1, bcr2 and bcr3) were determined from the size of the RT-PCR product. Patients were stratified into three risk groups, according to the Programa para el Tratamiento de Hemopatías Malignas (PETHEMA) system: high risk (HR) with leucocytes >10 × 109/l; intermediate risk (IR) with platelets ≤40 × 109/l and leucocytes ≤10 × 109/l, and; low risk (LR) with platelets >40 × 109/l and leucocytes ≤10 × 109/l. Neoplastic cell immunophenotype analysis was performed in all cases.4 All patients underwent complete laboratory tests for serology, biochemistry and hematology studies. Laboratory coagulation disturbances were the prolonged prothrombin time (PT) and activated partial thromboplastin time (aTTP). The ED was defined as death within 30 days of the APL diagnosis.
Treatment dataTherapy was initiated as soon as possible after a presumptive APL diagnosis. Of the total of 44 patients, 41 promptly received ATRA (until complete remission (CR)), the majority in the emergency room. Idarrubicin (IDA), daunorrubicin (DNR) or mitoxantrone (MTZ) and anthracycline on days 2, 4, 6, and 8, plus ATRA, were administered as induction therapy. After achieving CR, there occurred the administration of 3–4 courses of post-remission chemotherapy, containing MTZ, DNR or IDA with 45 mg/m2 of ATRA for 15 days in each cycle. In the high-risk group, an intermediate dose 1,000 mg/m2 on days 1, 2, 3 and 4 of Ara-C was added. Maintenance was performed for two years with 45 mg/m2 of ATRA for 15 days every three months and 15 mg/m2 of weekly methotrexate and 50 mg/m2/day of mercaptopurine, with careful toxicity control. The treatment of three patients was suspended before 24 months due to high toxicity. The hematological relapses were treated with a new course of chemotherapy (7 + 3). Given the advances in molecular techniques and the access to ATO, the four patients who had molecular relapse received ATO for 30 consecutive induction doses and 20 consolidation doses, while three patients underwent autologous hematopoietic stem cell transplantation after the ATO cycles. Three patients did not initiate chemotherapy because of ED. The European LeukemiaNet guidelines for supportive measures for APL patients were followed.5 Platelets were transfused to maintain platelet counts above 30−50 × 109/l and cryoprecipitate was administered to maintain the fibrinogen level above 150 mg/dl.
Statistical analysisThe chi-square and Fisher’s exact tests were used to analyze the association between nominal variables and the numeric variables were used in the Mann-Whitney U test. The overall survival (OS), disease-free survival (DFS), and event-free survival (EFS) were calculated using the Kaplan–Meier method. The OS was defined as the time from the initiation of induction therapy to death from any cause; those alive or lost to follow-up were censored on the date when last known to be alive. The DFS was defined as the time from CR to disease relapse or death from any cause, whichever occurred first. Patients who were alive without disease relapse were censored at the time last seen alive and disease-free. The EFS was defined as the time from the initiation of induction therapy to disease relapse or death from any cause, whichever occurred first. The log-rank test was used to compare the Kaplan-Meier curves. The variables for the Cox multivariate analysis for death included several categorized variables: presence of infection, coagulation disturbance, severe bleeding, hemoglobin <10 g/dl, platelets ≤40 × 109/l and neutrophils <500/mm3, expression of CD34 by immunophenotyping, additional chromosome abnormalities (ACAs), hypogranular APL and relapse. All statistical analyses were performed using the software MedCalc 19.0.6 A p ≤ 0.05 was considered statistically significant.
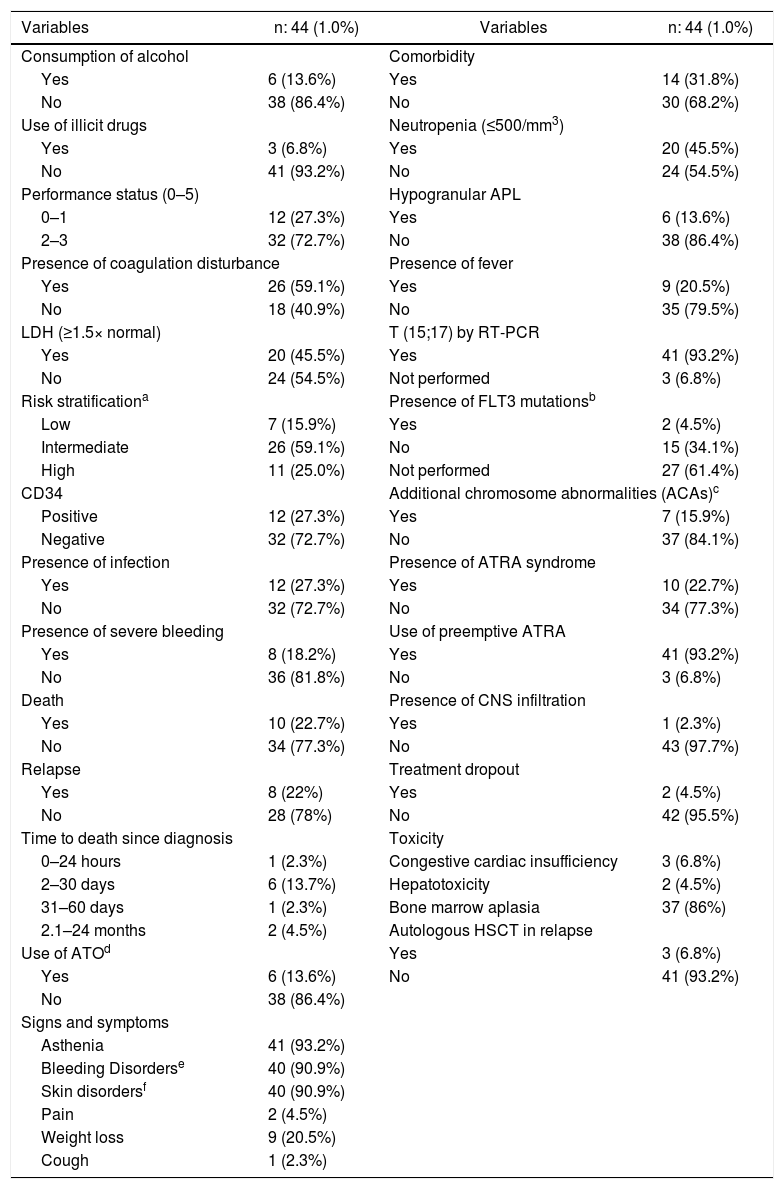
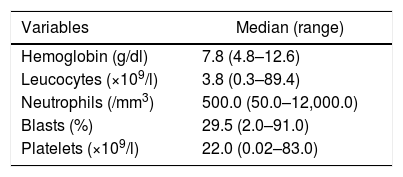
ResultsPatient characteristicsThe Hospital Universitário of the Universidade Federal de Santa Catarina is a public hospital, reference in acute leukemias, which covers a population of around 3 million, with 15 hospital beds, isolation area, emergency care and adult intensive care units. At the institution where the study was performed, APL represented 25% of all AML (188 cases) diagnoses during the study period. The main clinical and laboratory features of the 44 patients are summarized in Tables 1 and 2. Of the 44 patients, 24 were male (54.5%) and 20 were female (45.5%). The median age was 34 years (range 15–58 years old). All patients were human immunodeficiency virus (HIV) negative and one patient was positive for syphilis and hepatitis B and C. Seven (15.9%) patients were included in the low-risk group, 26 (59.1%) in the intermediate-risk group and 11 (25 %) in the high-risk group. Laboratory coagulopathy was present in 26 (59.1%) patients. More than 90% had some type of bleeding disorder, but severe bleeding, which occurs in the lungs, abdomen and central nervous system (CNS), was present in eight (18.2%) patients. One patient (2.3%) has deep venous thrombosis (DVT), associated with hemorrhage. Seventeen patients underwent FLT3 mutation analysis and two (4.5%) were positive.
Characteristics of the acute promyelocytic leukemia patients.
| Variables | n: 44 (1.0%) | Variables | n: 44 (1.0%) |
|---|---|---|---|
| Consumption of alcohol | Comorbidity | ||
| Yes | 6 (13.6%) | Yes | 14 (31.8%) |
| No | 38 (86.4%) | No | 30 (68.2%) |
| Use of illicit drugs | Neutropenia (≤500/mm3) | ||
| Yes | 3 (6.8%) | Yes | 20 (45.5%) |
| No | 41 (93.2%) | No | 24 (54.5%) |
| Performance status (0–5) | Hypogranular APL | ||
| 0–1 | 12 (27.3%) | Yes | 6 (13.6%) |
| 2–3 | 32 (72.7%) | No | 38 (86.4%) |
| Presence of coagulation disturbance | Presence of fever | ||
| Yes | 26 (59.1%) | Yes | 9 (20.5%) |
| No | 18 (40.9%) | No | 35 (79.5%) |
| LDH (≥1.5× normal) | T (15;17) by RT-PCR | ||
| Yes | 20 (45.5%) | Yes | 41 (93.2%) |
| No | 24 (54.5%) | Not performed | 3 (6.8%) |
| Risk stratificationa | Presence of FLT3 mutationsb | ||
| Low | 7 (15.9%) | Yes | 2 (4.5%) |
| Intermediate | 26 (59.1%) | No | 15 (34.1%) |
| High | 11 (25.0%) | Not performed | 27 (61.4%) |
| CD34 | Additional chromosome abnormalities (ACAs)c | ||
| Positive | 12 (27.3%) | Yes | 7 (15.9%) |
| Negative | 32 (72.7%) | No | 37 (84.1%) |
| Presence of infection | Presence of ATRA syndrome | ||
| Yes | 12 (27.3%) | Yes | 10 (22.7%) |
| No | 32 (72.7%) | No | 34 (77.3%) |
| Presence of severe bleeding | Use of preemptive ATRA | ||
| Yes | 8 (18.2%) | Yes | 41 (93.2%) |
| No | 36 (81.8%) | No | 3 (6.8%) |
| Death | Presence of CNS infiltration | ||
| Yes | 10 (22.7%) | Yes | 1 (2.3%) |
| No | 34 (77.3%) | No | 43 (97.7%) |
| Relapse | Treatment dropout | ||
| Yes | 8 (22%) | Yes | 2 (4.5%) |
| No | 28 (78%) | No | 42 (95.5%) |
| Time to death since diagnosis | Toxicity | ||
| 0–24 hours | 1 (2.3%) | Congestive cardiac insufficiency | 3 (6.8%) |
| 2–30 days | 6 (13.7%) | Hepatotoxicity | 2 (4.5%) |
| 31–60 days | 1 (2.3%) | Bone marrow aplasia | 37 (86%) |
| 2.1–24 months | 2 (4.5%) | Autologous HSCT in relapse | |
| Use of ATOd | Yes | 3 (6.8%) | |
| Yes | 6 (13.6%) | No | 41 (93.2%) |
| No | 38 (86.4%) | ||
| Signs and symptoms | |||
| Asthenia | 41 (93.2%) | ||
| Bleeding Disorderse | 40 (90.9%) | ||
| Skin disordersf | 40 (90.9%) | ||
| Pain | 2 (4.5%) | ||
| Weight loss | 9 (20.5%) | ||
| Cough | 1 (2.3%) | ||
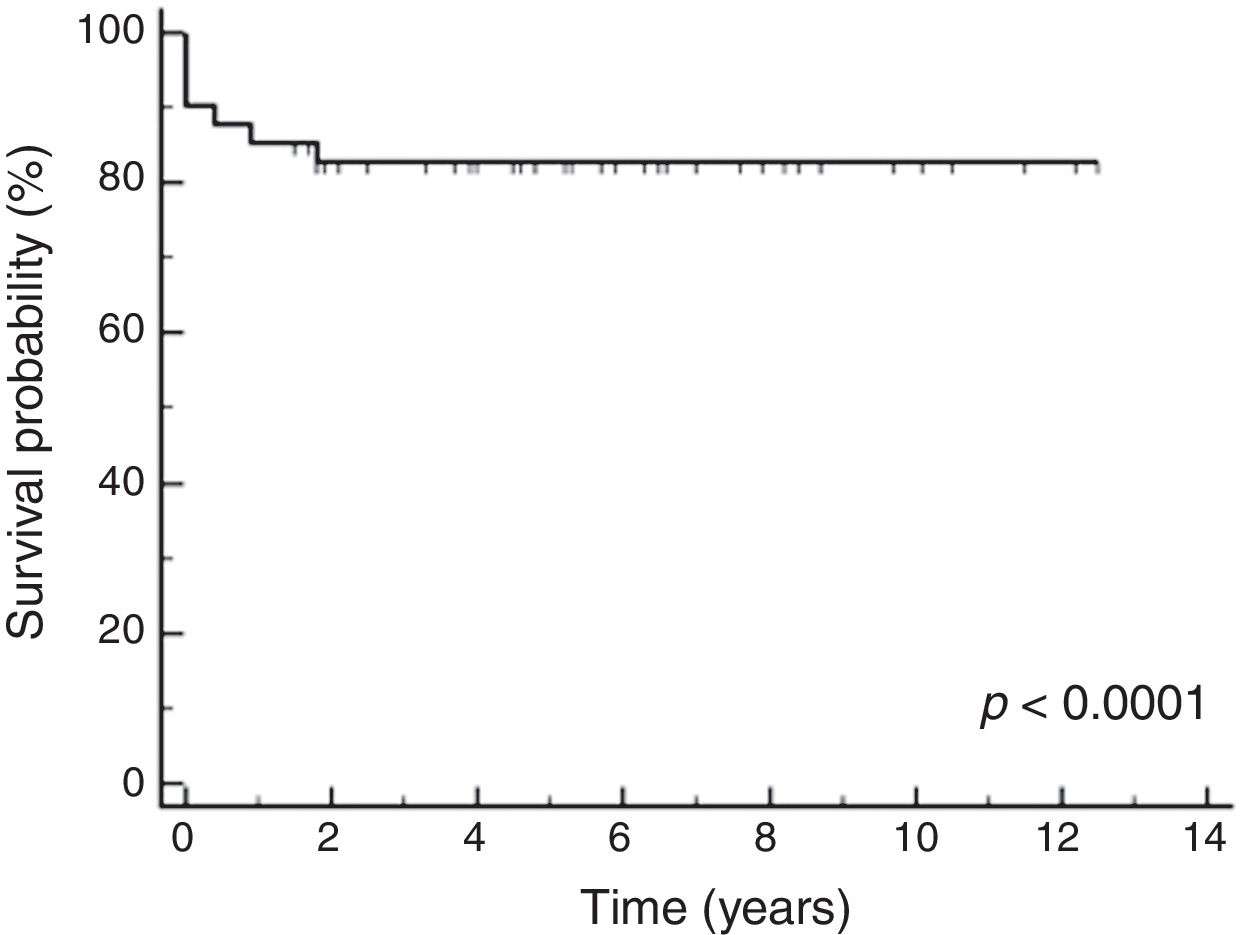
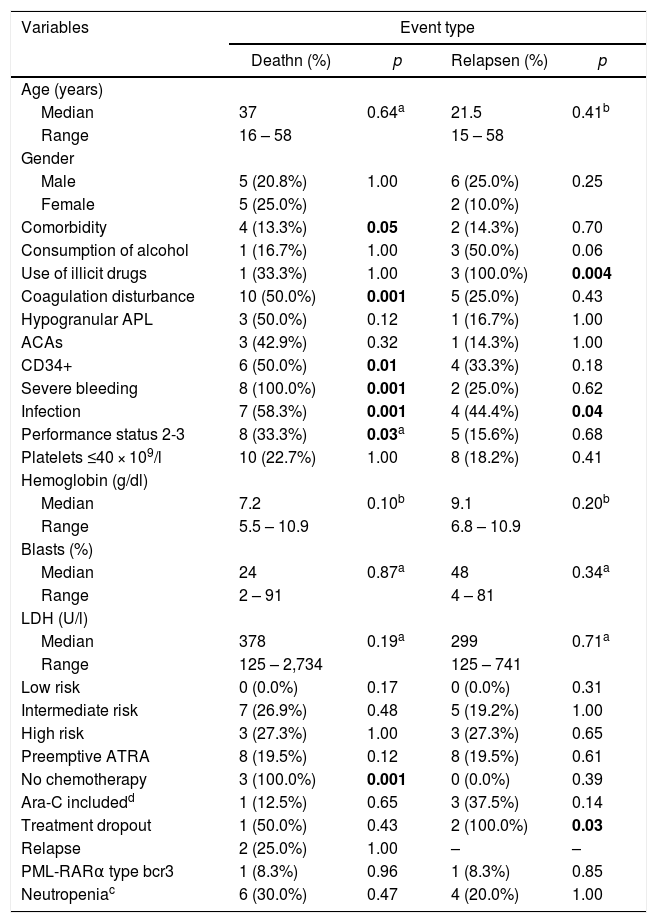
The median follow-up time was 150 months 4,575 days. The 5-year OS, DFS, and EFS were 82.7% (95% confidence interval (CI95%): 76.7%–88.7%), 62.0% (CI95%: 53.0%–71.0%) and 73.2% (CI95%: 65.4%–81.0%), respectively, which can be seen in Fig. 1.
Of the 41 patients treated, 36 (87.8% (CI95%: 81.8%–93.8%)) responded to treatment. Of the initial 44 patients, ten (22.7%) patients died: one (2.3%) due to treatment dropout, one (2.3%) due to hematologic and extramedullary relapse (spleen) and eight (18.2%), as a result of severe bleeding (seven had ED and one died after induction therapy). The major cause of death was hemorrhage, especially in the central nervous system. One patient that had early death had the following concomitant factors: high risk, hypogranular APL and the FLT3-ITD mutation. All patients who did not have a coagulation disturbance were alive by the end of the study. Eight (22%) patients relapsed: three (8.3%) had a hematologic relapse, one (2.7%) had a hematologic and extramedullary relapse (spleen) and four had only a molecular relapse (11%). Of the patients who relapsed, two were due to treatment dropout. All relapsed patients who used ATO were alive and in control of the disease by the end of the study. Table 3 shows the association between variables and death or relapse.
Association between prognostic factors and death or relapse.
| Variables | Event type | |||
|---|---|---|---|---|
| Deathn (%) | p | Relapsen (%) | p | |
| Age (years) | ||||
| Median | 37 | 0.64a | 21.5 | 0.41b |
| Range | 16 – 58 | 15 – 58 | ||
| Gender | ||||
| Male | 5 (20.8%) | 1.00 | 6 (25.0%) | 0.25 |
| Female | 5 (25.0%) | 2 (10.0%) | ||
| Comorbidity | 4 (13.3%) | 0.05 | 2 (14.3%) | 0.70 |
| Consumption of alcohol | 1 (16.7%) | 1.00 | 3 (50.0%) | 0.06 |
| Use of illicit drugs | 1 (33.3%) | 1.00 | 3 (100.0%) | 0.004 |
| Coagulation disturbance | 10 (50.0%) | 0.001 | 5 (25.0%) | 0.43 |
| Hypogranular APL | 3 (50.0%) | 0.12 | 1 (16.7%) | 1.00 |
| ACAs | 3 (42.9%) | 0.32 | 1 (14.3%) | 1.00 |
| CD34+ | 6 (50.0%) | 0.01 | 4 (33.3%) | 0.18 |
| Severe bleeding | 8 (100.0%) | 0.001 | 2 (25.0%) | 0.62 |
| Infection | 7 (58.3%) | 0.001 | 4 (44.4%) | 0.04 |
| Performance status 2-3 | 8 (33.3%) | 0.03a | 5 (15.6%) | 0.68 |
| Platelets ≤40 × 109/l | 10 (22.7%) | 1.00 | 8 (18.2%) | 0.41 |
| Hemoglobin (g/dl) | ||||
| Median | 7.2 | 0.10b | 9.1 | 0.20b |
| Range | 5.5 – 10.9 | 6.8 – 10.9 | ||
| Blasts (%) | ||||
| Median | 24 | 0.87a | 48 | 0.34a |
| Range | 2 – 91 | 4 – 81 | ||
| LDH (U/l) | ||||
| Median | 378 | 0.19a | 299 | 0.71a |
| Range | 125 – 2,734 | 125 – 741 | ||
| Low risk | 0 (0.0%) | 0.17 | 0 (0.0%) | 0.31 |
| Intermediate risk | 7 (26.9%) | 0.48 | 5 (19.2%) | 1.00 |
| High risk | 3 (27.3%) | 1.00 | 3 (27.3%) | 0.65 |
| Preemptive ATRA | 8 (19.5%) | 0.12 | 8 (19.5%) | 0.61 |
| No chemotherapy | 3 (100.0%) | 0.001 | 0 (0.0%) | 0.39 |
| Ara-C includedd | 1 (12.5%) | 0.65 | 3 (37.5%) | 0.14 |
| Treatment dropout | 1 (50.0%) | 0.43 | 2 (100.0%) | 0.03 |
| Relapse | 2 (25.0%) | 1.00 | – | – |
| PML-RARα type bcr3 | 1 (8.3%) | 0.96 | 1 (8.3%) | 0.85 |
| Neutropeniac | 6 (30.0%) | 0.47 | 4 (20.0%) | 1.00 |
The chi-square test, Fisher’s exact test or Mann–Whitney U test showed that the presence of a coagulation disturbance, severe bleeding, infection, high performance status (the highest was 2–3), ACAs, CD34+ by immunophenotyping and comorbidities, as well as the absence of chemotherapy initiation, was related to the occurrence of death (p ≤ 0.05). In addition, prior use of drugs, infection at presentation and treatment dropout were shown to be related to relapse (Table 3).
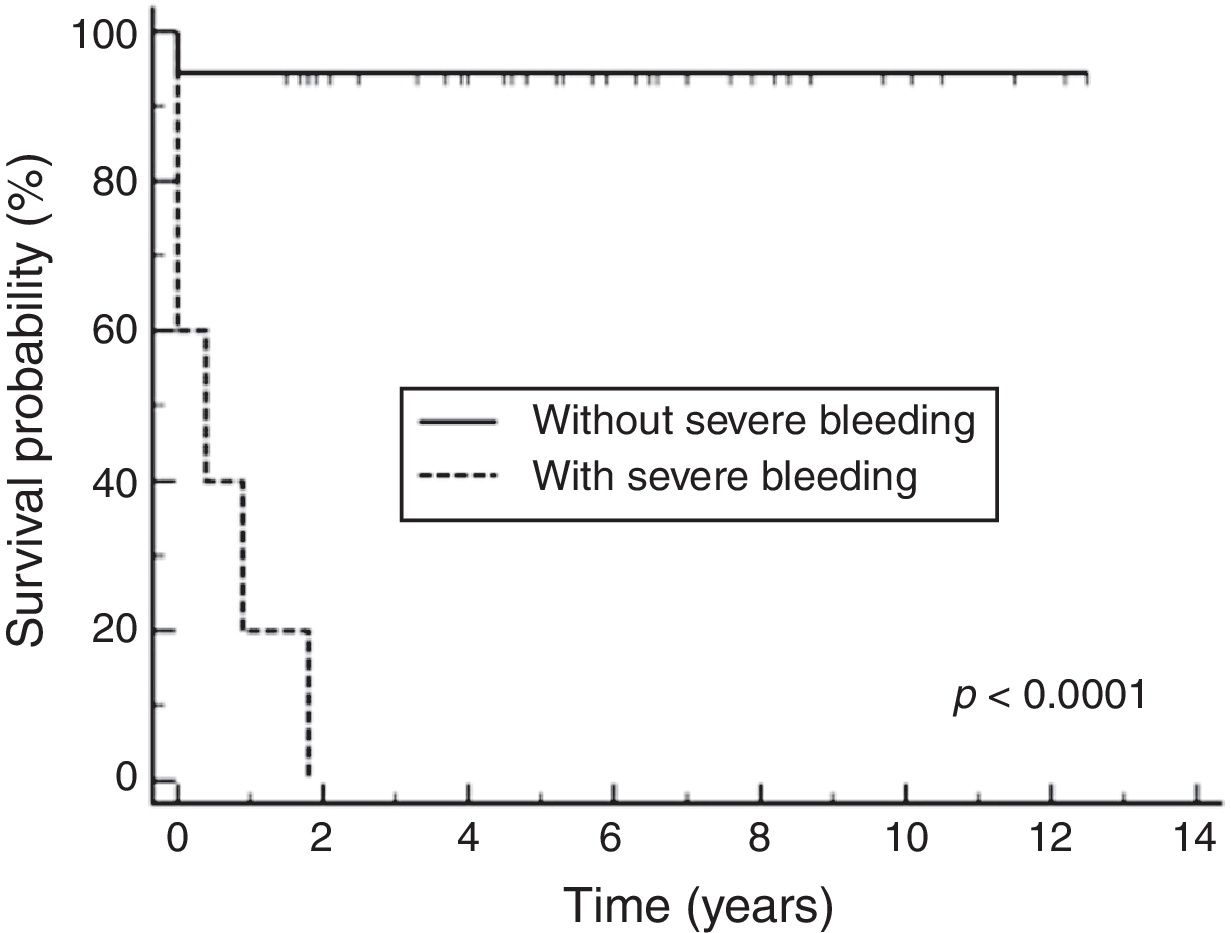
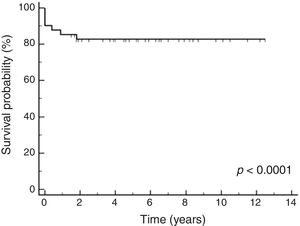
Among prognostic factors included in the Cox multivariate analysis, only severe bleeding significantly influenced the survival probability (p < 0.0001), with a relative risk of 36.10 (CI95%: 7.29–178.72). The Cox multivariate analysis showed a survival probability of 89% (CI95%: 83%–95%).
According to the Kaplan-Meier curves, the mean survival time of the patients who did not have severe bleeding was 4,309 days CI95%: 3968 to 4650 and of those who had severe bleeding was 149 days CI95%: 0–306), as shown in Fig. 2.
DiscussionMany efforts have been made to identify prognostic markers for death in APL and make it a curable disease.1,5 The white blood cell count is a well-known important prognostic factor, described by the Programa para el Estudio de la Terapéutica en Hemopatía Maligna (PETHEMA) and Gruppo Italiano Malattie e Matologiche dell'Adulto (GIMEMA) cooperative groups in 2000, which provides guidance for treatment.20 Even as a retrospective study with a limited number of patients, the data confirmed the slightly higher incidence of APL in the Latin American population and reflected the results of other studies.
The population of the present study includes young adult patients, the majority of whom were at intermediate and high risk (84.1%), and 59.1% had a laboratory coagulopathy at diagnosis, which compromised the outcome. APL is frequently associated with TC-APL, which leads to a high mortality rate, especially early mortality.5 It is known that the prolongation of PT and aPTT, associated with thrombocytopenia and leukocytosis, are risk factors for severe bleeding and possible death. Patients at high risk for bleeding should be managed more aggressively.21,22
Many articles are trying to find new prognostic factors, such as the hemoglobin count, blast count,23 expression of CD34 by immunophenotyping,24 presence of FLT3 mutations,6,25 short bcr3 type PML-RARα fusion gene,26 ACAs,27 severe bleeding, or hypogranular APL,28 and to discover whether these new factors indeed have any influence on APL.
The association of reciprocal translocation t (15;17) with other ACAs has been established in many studies, but this has not happened for any independent influence on the outcome.25 The incidence is around 10–25 % of ACAs in APL.25 The FLT3-ITD occurs in 12–38 % of all APL patients and the FLT3-D835, in 2–20%.6,23 The FLT3-ITD and D835 mutations occurred in 17 patients and two (4.5%) of them were positive for FLT3-ITD (one had hypogranular APL, bcr3 transcript, was at high risk, and suffered early death). The statistical analysis was difficult because of the low number of patients. Usually, FLT3 was reported to worsen prognosis and to be associated with leukocytosis and the hypogranular subtype.6 However, more studies need to be carried out with the FLT3 mutations and ATO therapy as first line to observe the responses.
CD34 is a type I transmembrane glycophosphoprotein expressed on hematopoietic precursor cells in the early stages of differentiation. In APL, CD34 expression is clinically related to leukocytosis and hypogranular APL, but there is no clear association with the outcome.24 In this study, the CD34 expression was observed in 12 (27.3%) patients (Table 1). A simple analysis showed an association with death and CD34 expression, but this was not confirmed in a multivariable logistic regression analysis, reducing the power of this prognostic factor, however, the low number of patients in this study may have influenced this analysis.
APL therapy has improved dramatically over the past three decades, leading to a significant increase in the OS.29 This study presented a good OS rate (82.7%); it was carried out in a developing country by a teaching hospital fully funded by public resources. The early use of ATRA is a key factor contributing to a positive outcome and the medication may be administered as early as in the emergency room, even before a definitive diagnosis. The role of properly trained general practitioners and emergency physicians, as well as laboratory staff, is crucial for APL cases to be quickly recognized.
The introduction of the use of ATO for relapsed cases at the center where this study was performed, in 2008, changed the evolution of these patients with better response rates. As seen in many papers, the use of ATO has been a new target for therapy in APL.17,24
The International Consortium on Acute Promyelocytic Leukemia (IC-APL) was established in 2005 as an initiative of the International Members Committee of the American Society of Hematology (ASH). Its purpose was to create a network of institutions in developing countries for the exchange of experience and data and to receive support from well-established cooperative groups in the Unites States and Europe. After IC-APL was created, life expectancy of APL patients increased by 30%, which shows that this cooperation was essential for the increase in positive outcomes.18 However, early death, late diagnosis and relapse remain problems that need to be identified and avoided.17 In an American study by Park30 2011 with 1,400 APL patients, the overall ED was 17.3% between 1992 and 2007, even after the introduction of ATRA therapy, which corroborates the high ED rates (16.0%) in the present study. As can be seen in the Cox multivariate regression, the factor significantly related to death was severe bleeding, which was responsible for almost all deaths.
Furthermore, recognizing APL as a medical emergency that requires specific and simultaneous actions, including rapid and aggressive supportive care for bleeding and coagulopathy, early diagnosis with new molecular techniques, such as anti-PML mAb, prompt initiation of ATRA, transferal of patients to experienced medical centers when the disease is first suspected and the identification of high-risk patients to optimize treatment are key measures in improving the outcomes.
In conclusion, the present study demonstrates the reality of APL in the 20th century in a developing country and a public university hospital. Our results have shown that the rapid laboratory diagnosis of patients and immediate initiation of treatment with ATRA directly influence their survival. However, some cases had hemorrhagic complications that may be associated with the difficulty of patients to access the hospital, difficulty in recognizing early symptoms and difficulty in advanced or blood support.
The presence of severe bleeding is still the most important impact factor for death and its early recognition at patient presentation is very important and might be a useful tool in the identification of high-risk patients. However, more studies, continuous networking programs and basic research in developing countries must continue to be performed.
Conflicts of interestThe authors declare no conflicts of interest.
Santos-Silva M.C. is recipient of a Research Fellowship from CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico - Brazil).