Hereditary pyropoikilocytosis (HPP) is a rare recessively-inherited hemolytic anemia, related to hereditary elliptocytosis (HE). The HE red blood cells (RBCs) are partially deficient in spectrin or protein 4.1R and contain a mutant α or β-spectrin that is defective in terms of spectrin self-association.1 In HPP, homozygous or compound heterozygous allele mutations in the genes that underlie the spectrin–protein 4.1R-actin complex weaken the “horizontal” cytoskeletal associations, causing decreased mechanical stability and deformability of erythrocytes, that results in striking microcytosis, with a mean corpuscular volume (MVC) typically less than 60fL, poikilocytosis with micro-spherocytosis and increased fragmentation and thermal instability of the RBCs.2
HPP is typically diagnosed in patients with a family history of HE, but presents with increased severity, variable splenomegaly, frequently with transfusion requirement in infancy or, rarely, long-term transfusion dependence.3
However, coexistence of membranopathies with other red cell disorders impose a diagnostic challenge that requires a thorough analysis of clinical, morphologic and laboratory findings, in order to differentiate and elucidate the conditions involved.
Here, we describe the clinical and laboratory changes in a patient with co-inheritance of beta-thalassemia minor and HPP.
Case reportA 5-year-old boy was referred to our institute due to a moderate microcytic and hypochromic anemia, with hemoglobin levels ranging from 8.0 to 8.5g/dL that were persistent and not responsive to iron supplementation. The main hypothesis of the referring physician was beta-thalassemia minor because the patient's mother had been diagnosed with this condition several years prior to this. Clinical examination revealed mild pallor, jaundice and frontal bossing; the liver and spleen were felt 2cm below the right and left costal margin, respectively.
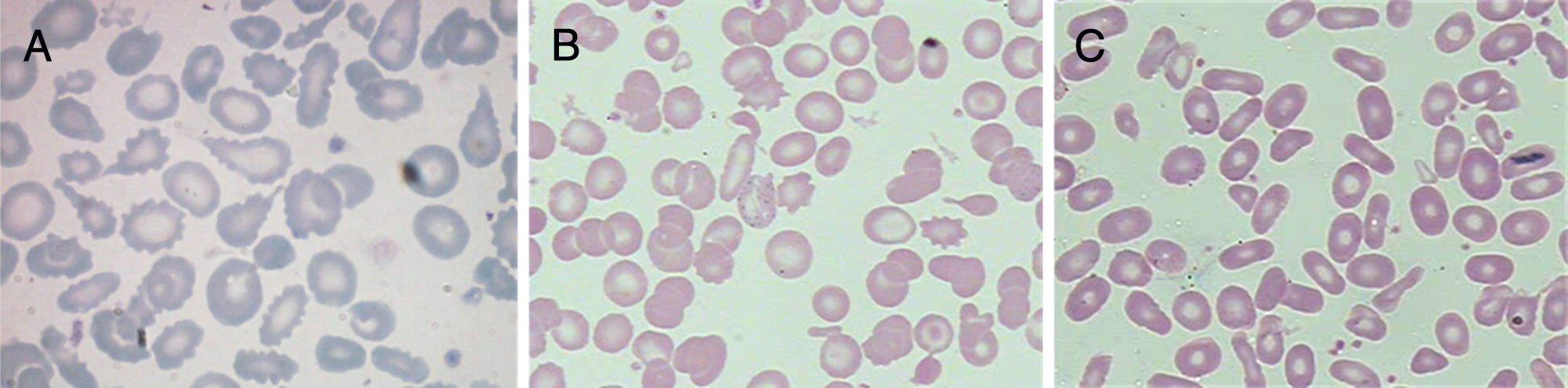
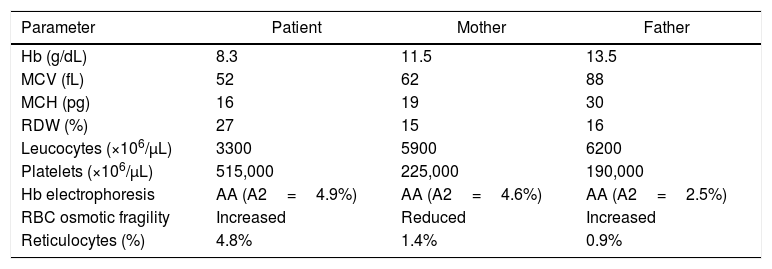
A more comprehensive investigation was made of the patient and his parents. The main results are summarized in Table 1. Figure 1 displays the morphological abnormalities of the RBCs in their peripheral blood smears.
Laboratory characteristics of the family members.
| Parameter | Patient | Mother | Father |
|---|---|---|---|
| Hb (g/dL) | 8.3 | 11.5 | 13.5 |
| MCV (fL) | 52 | 62 | 88 |
| MCH (pg) | 16 | 19 | 30 |
| RDW (%) | 27 | 15 | 16 |
| Leucocytes (×106/μL) | 3300 | 5900 | 6200 |
| Platelets (×106/μL) | 515,000 | 225,000 | 190,000 |
| Hb electrophoresis | AA (A2=4.9%) | AA (A2=4.6%) | AA (A2=2.5%) |
| RBC osmotic fragility | Increased | Reduced | Increased |
| Reticulocytes (%) | 4.8% | 1.4% | 0.9% |
Hb=hemoglobin; MCV=mean corpuscular volume; MCH=mean corpuscular hemoglobin; RDW=red blood cell distribution width; RBC=red blood cells. There is no expansion for "AA".
Taken together, the erythrogram, fragility test, Hb electrophoresis and RBC morphology were suggestive of coinheritance of beta-thalassemia minor and HPP in the patient, whereas in his parents, the results were indicative of beta-thalassemia minor (mother) and HE (father).
Further investigations in the patient revealed heterozygosis for the CD39 mutation, which is associated with the beta-thalassemia minor phenotype.4 The thermal sensitivity test showed increased RBC fragmentation at 45°C. Furthermore, the thrombocytosis was found to be spurious due to contamination of the platelet channel by microcytes with less than 30fL, and the hemolysis markers were increased (indirect bilirubin=2.2mg/dL and reticulocytes=4.8%).
To date, at the age of 14, his condition has been following a relatively benign and stable course with supportive measures, without the need for specific therapy, such as hydroxyurea and splenectomy. During the follow-up, the patient was transfused on only two occasions. The first one, at the age of 8, due to an aplastic crisis caused by a Parvovirus B19 infection, and at the age of 9, when he presented with infectious mononucleosis, requiring also therapy with intravenous immunoglobulin.
DiscussionIn our patient, the diagnosis of beta-thalassemia minor was, as expected, rather straightforward; however, the clinical severity along with marked morphologic abnormalities in the peripheral blood smear raised the suspicion of another inherited red blood cell disorder with overlapping features, in relation to beta-thalassemia.
Family investigation can add substantial information in this situation, as up to one-third of HPP patients have a sibling or parent with HE.5 Unlike hereditary spherocytosis, that asymptomatic parents can produce offspring with typical signs and symptoms of this disease, in a majority of HE/HPP cases, at least one parent is expected to have an HE phenotype.6 In our report, detailed family investigation revealed heterogeneous clinical and laboratorial presentations among the affected patient and his parents and helped to determine the discrimination pattern of two concurrent red cell disorders.
In general, an appropriate diagnosis of HE and HPP can often be made with screening tests, careful family investigation, along with blood count results, reticulocyte count, red cell morphology, and chemistry results.5 Although not available in our case, specialist tests, such as the SDS–polyacrylamide gel electrophoresis of erythrocyte membrane proteins, monovalent cation flux measurement and molecular analysis of membrane protein genes can be requested for further investigation or for suspected cases with unremarkable clinical and laboratory features.6,7
Coinheritance of beta-thalassemia and hereditary spherocytosis is known to occur in the Mediterranean countries.6 However, little is known about the prevalence of co-inheritance of other membranopathies with hemoglobinopathies. In our case, the patient presented an elevated hemoglobin A2 level, which indicates the inheritance of beta-thalassemia minor. However, the higher intensity of anemia with striking microcytosis, distinguishing poikilocytosis and RBC fragmentation, along with increased RBC fragility and clinical features of chronic and significant hemolysis, were compatible with HPP. In our patient, this condition could be the result of compound heterozygosis for a mutant α or β-spectrin from his father and, presumably, an αLELY allele from his mother, which is an α-spectrin low expression polymorphic allele found in 20–30% of the population, which can be silent in heterozygous individuals.3,6
In conclusion, clinical and laboratory physicians must be aware of the possibility of coinheritance of RBC disorders in patients with a divergent clinical course and, in the context of HPP, be able to recognize the morphologic and laboratory abnormalities of this potentially severe anemia.
Conflicts of interestThe authors declare no conflicts of interest.