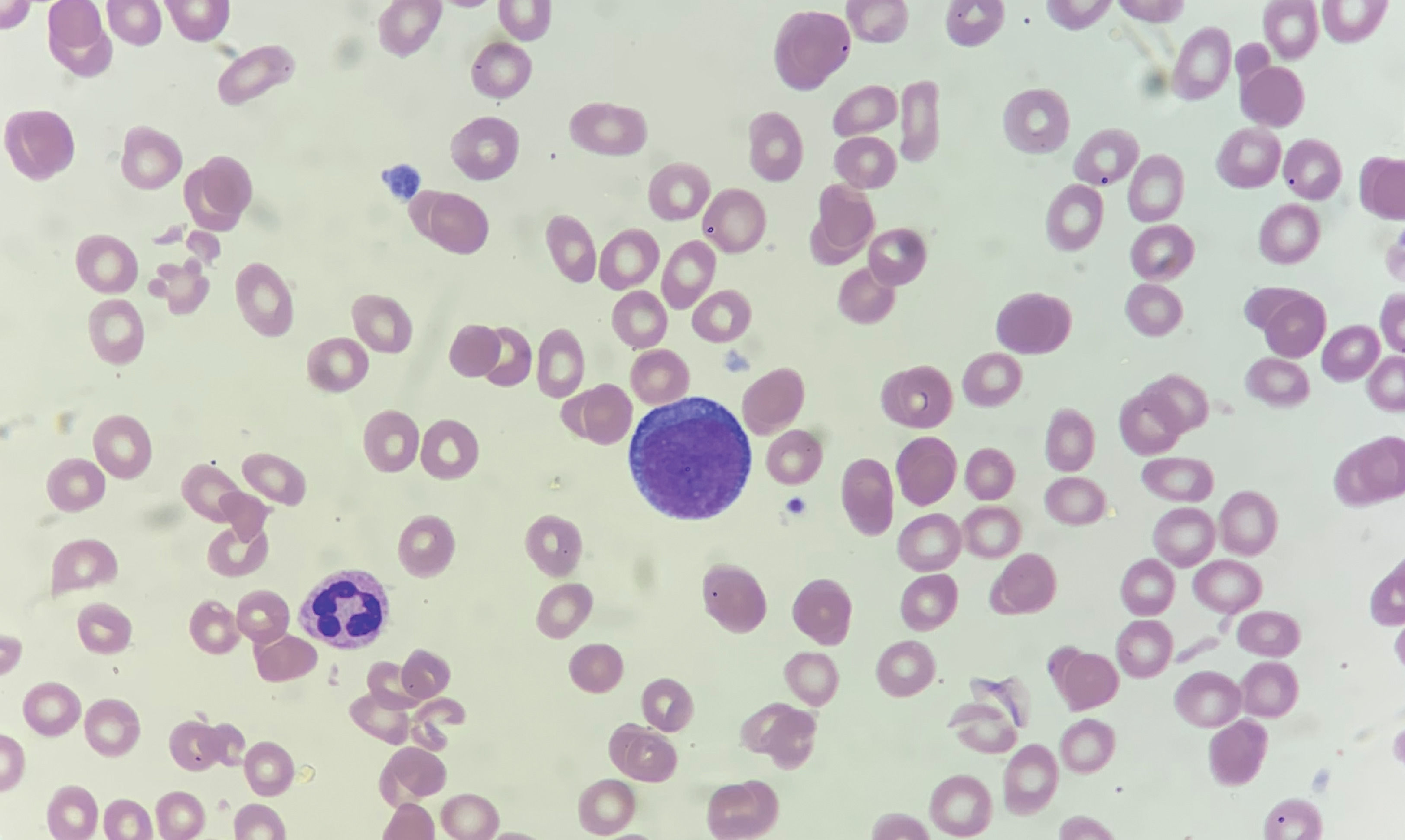
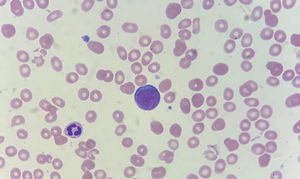
A 80-years-old woman presented with 6-weeks history of fatigue and shortness of breath. Hemoglobin was 9.7 g/dL, platelets were 79 × 10³ /μL, leukocytes were 4.06 × 10³ /μL and B12 vitamin level was <200 pg/ml (reference value 197 - 771). After administration of cyanocobalamin her B12 vitamin level improved, but the anemia got progressively worse (hemoglobin was 3.7 g/dl) and reticulocyte count did not improve. A peripheral blood smear showed proerythroblasts (Figures 1, 2). Bone marrow aspirates and biopsy showed hypercellularity, being mainly erythroid progenitors, constituting >80% of bone marrow cell count with >30% proerythroblast without a significant myeloblastic component. The erythroid progenitors exhibited CD45+, CD34-, CD71+ and CD117+ by flow cytometry, and E-cadherin+, CD71+ and TP53+ by immunohistochemical staining. Fluorescence in situ hybridization analysis showed deletion 5q and loss of TP53. Karyotype showed deletion 5q, and derivates chromosomes 17 (included loss of TP53 gen) and 19. Next-generation sequencing panel detected potentially pathogenic variants in TET2 (VAF 28%) and TP53 (VAF 40%) genes. The findings were diagnostic of pure erythroblastic leukemia (PEL). Our patient was treated with azacytidine and blood transfusion support with a poor response over five months of follow-up.
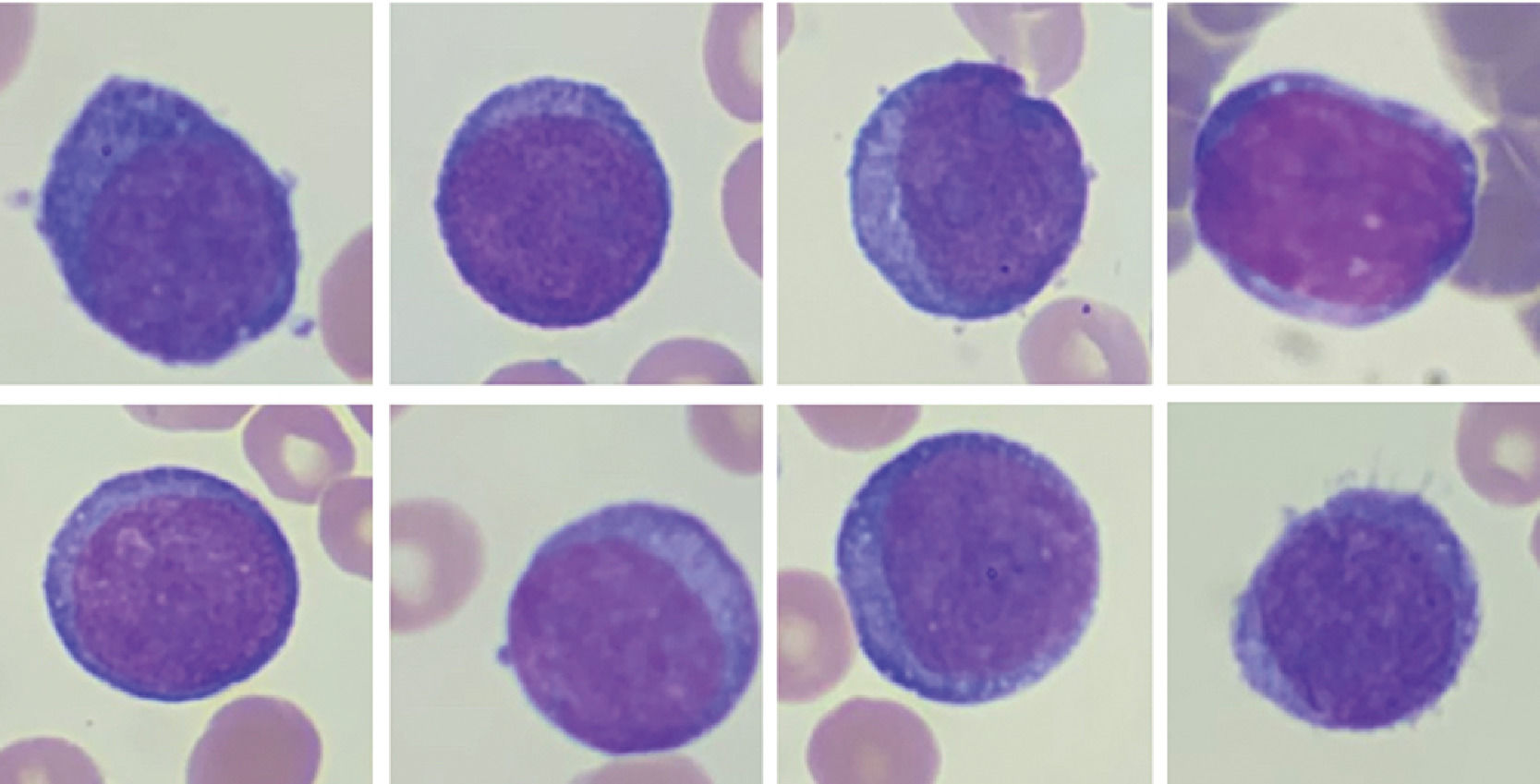
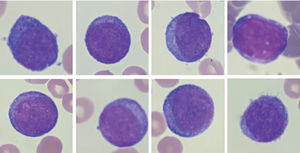
Peripheral blood smear. Proerythroblasts; panel A-H: erythroid progenitors having large irregular nuclei, dispersed chromatin, some with prominent nucleoli, deeply basophilic cytoplasm, and high nuclear to cytoplasmic ratios. Wright stain; 100X objective, original magnification X1000.
PEL is a rare type of acute leukemia that represents less than 1% of all cases of acute myeloid leukemia (previously called M6, by the French-American-British cooperative group).1,2 PEL may be therapy-related, preceded by a myelodysplastic syndrome or develop de novo.2 PEL is defined in the 2016 WHO classification system, as a neoplastic proliferation of erythroid progenitors constituting > 80% of bone marrow cellularity with ≥30% proerythroblasts without a significant myeloblastic component.3 Reactive erythroid hyperplasia is a well-known morphologic mimic of PEL in many diverse clinical situations due to erythroid hyperplasia as non-neoplastic (eg, megaloblastic anemia) and neoplastic entities. Although clinical presentation, laboratory, cytogenetic and molecular studies may ultimately resolve the differential diagnosis (eg, P53 mutation, complex karyotype).4 PEL have a clinically aggressive course associated with a poor prognosis.1
Data availability statementData sharing not applicable to this article as no datasets were gener-ated or analysed during the current study.